NEX SCIENCE PRACTICE QUESTIONS
Designed to reinforce core scientific knowledge through realistic, exam-style questions that reflect nursing program entry standards.
Topics Covered
Cellular structure and function
Organ system interactions
Chemical reactions and solutions
Basic physics principles in health science
Data interpretation
Foundational microbiology
00:00
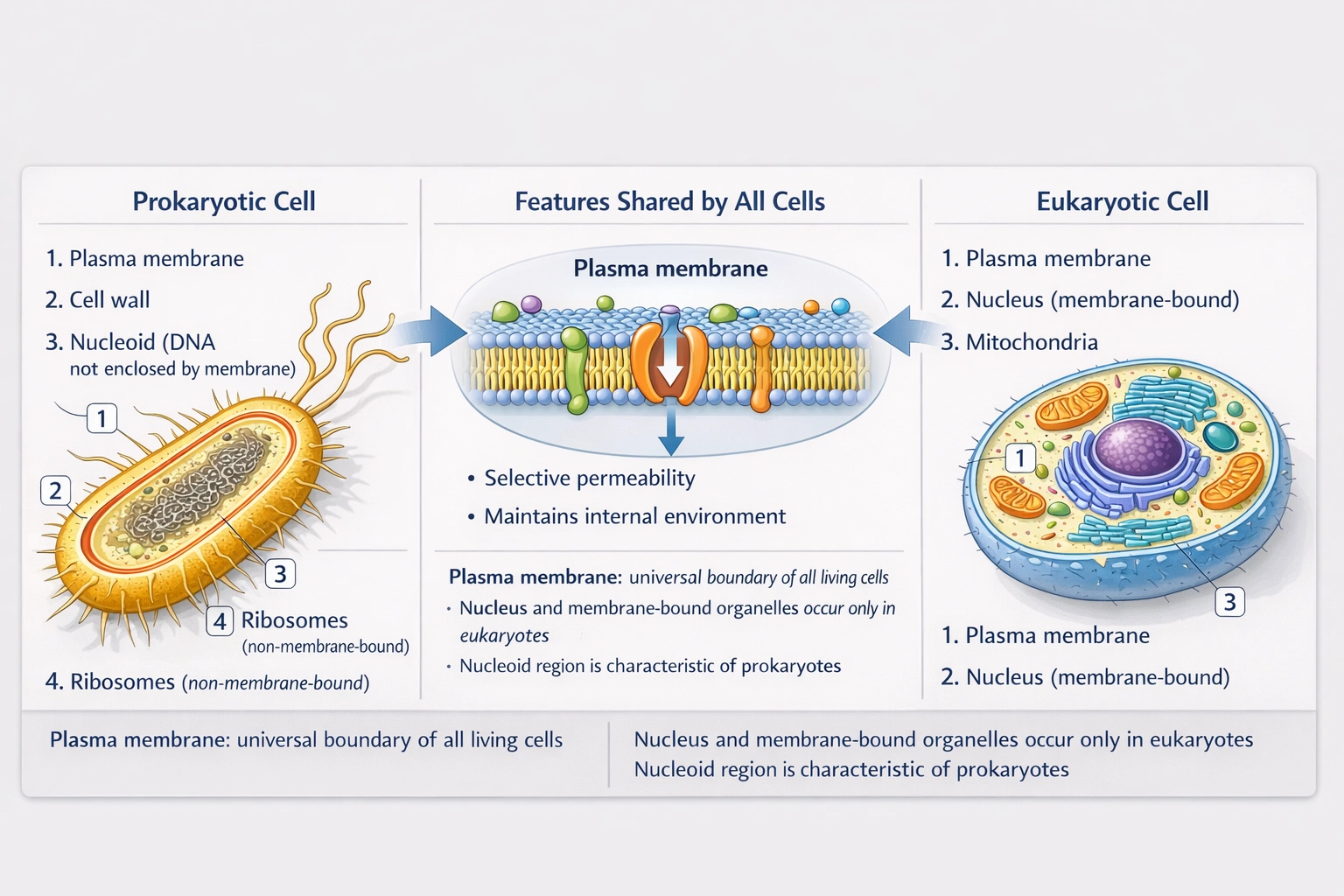
What is ONE feature that both prokaryotes and eukaryotes have in common?
A.
A plasma membrane
B. A nucleus enclosed by a membrane
C. Organelles
D. A nucleoid
Rationale
Both prokaryotic and eukaryotic cells possess a plasma membrane composed of a phospholipid bilayer with embedded proteins that regulates material exchange, maintains cellular integrity, and establishes electrochemical gradients essential for life processes.
The plasma membrane represents a universal cellular feature predating the evolutionary divergence of prokaryotes and eukaryotes approximately 2 billion years ago—serving as the fundamental boundary separating intracellular from extracellular environments across all domains of life.
A) A plasma membrane
All living cells require a selectively permeable barrier to maintain homeostasis, concentrate metabolites, and generate proton motive force for ATP synthesis. Both prokaryotes (bacteria, archaea) and eukaryotes construct plasma membranes from amphipathic phospholipids forming bilayers with hydrophobic tails inward and hydrophilic heads outward. Embedded proteins facilitate transport, signaling, and energy transduction in both cell types. This universal feature reflects common ancestry and fundamental biophysical requirements for cellular life—no known organism exists without a plasma membrane boundary.
B) A nucleus enclosed by a membrane
Membrane-bound nuclei exist exclusively in eukaryotes, defining the domain Eukarya. Prokaryotes lack nuclear membranes; their DNA resides in a nucleoid region directly exposed to cytoplasm. This distinction represents the primary taxonomic difference between prokaryotic and eukaryotic organization—making nuclear enclosure a eukaryotic synapomorphy rather than shared feature.
C) Organelles
"Organelles" requires precise definition. Membrane-bound organelles (mitochondria, ER, Golgi, lysosomes) exist only in eukaryotes. Prokaryotes possess non-membrane-bound functional structures (ribosomes, carboxysomes, magnetosomes) sometimes called "organelles" in broad terminology, but standard biological usage reserves "organelle" for membrane-enclosed compartments. Since the question likely intends classical organelle definition (membrane-bound), this feature isn't universally shared. Even with broad definition, ribosomes represent the shared subcellular structure—but option A (plasma membrane) is more fundamentally universal.
D) A nucleoid
Nucleoids—irregularly shaped DNA-protein complexes without surrounding membranes—characterize prokaryotic chromosome organization. Eukaryotes package DNA within membrane-bound nuclei using histone proteins to form chromatin, not nucleoids. While both store genetic material, the structural organization differs fundamentally: nucleoid (prokaryotic) versus nucleus (eukaryotic). This represents a distinguishing rather than shared feature.
Conclusion:
The plasma membrane constitutes life's universal boundary structure—essential for maintaining the disequilibrium between intracellular and extracellular environments that defines living systems. While prokaryotes and eukaryotes diverged dramatically in internal organization (nuclear enclosure, organelle complexity), both retain this fundamental phospholipid bilayer boundary inherited from the last universal common ancestor (LUCA). Option A correctly identifies this shared essential feature, distinguishing it from domain-specific characteristics like nuclear membranes (B), membrane-bound organelles (C), or nucleoid organization (D). Understanding this commonality underscores unity of life principles while recognizing evolutionary innovations that enabled eukaryotic complexity.
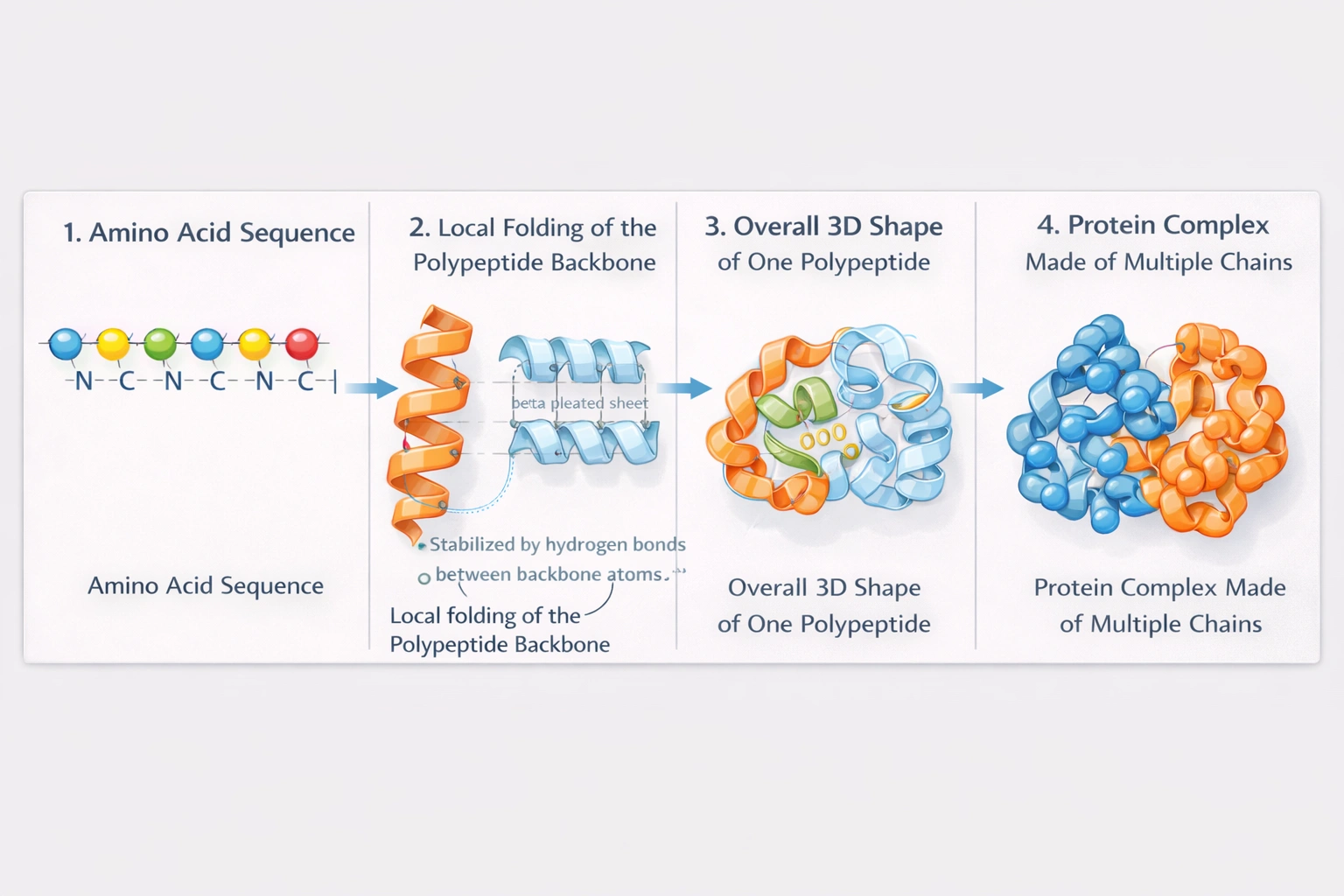
Which level of protein structure is defined by the folds and coils of the protein's polypeptide backbone?
A.
Primary
B. Secondary
C. Tertiary
D. Quaternary
Rationale
Secondary structure encompasses local folding patterns stabilized by hydrogen bonds between backbone amide and carbonyl groups—primarily alpha-helices (coils) and beta-pleated sheets (folds)—without involvement of amino acid side chains.
Secondary structure represents the first level of protein folding beyond linear sequence, arising from hydrogen bonding between backbone atoms at regular intervals that create repetitive structural motifs essential for higher-order folding and functional domains.
A) Primary
Primary structure refers exclusively to the linear sequence of amino acids linked by peptide bonds—determined by genetic code and covalent bonding. It contains no folding or three-dimensional arrangement; it is simply the polypeptide chain's chemical formula. Primary structure dictates all higher folding levels but itself remains unfolded and linear.
B) Secondary
Secondary structure forms when hydrogen bonds develop between backbone carbonyl oxygen (C=O) of one amino acid and amide hydrogen (N-H) of another—typically four residues away in alpha-helices (right-handed coils with 3.6 residues per turn) or between adjacent strands in beta-sheets (parallel or antiparallel folds). These interactions involve only backbone atoms, not R-groups. Alpha-helices and beta-sheets represent the two predominant secondary structures, with turns and loops connecting them. This level defines the characteristic coils and folds referenced in the question.
C) Tertiary
Tertiary structure describes the overall three-dimensional folding of a single polypeptide chain, stabilized by interactions among amino acid side chains (R-groups): hydrophobic interactions, disulfide bridges, hydrogen bonds, ionic bonds, and van der Waals forces. It incorporates secondary structural elements into globular or fibrous arrangements but extends beyond backbone folding to include side chain positioning and long-range interactions.
D) Quaternary
Quaternary structure involves assembly of multiple independent polypeptide chains (subunits) into a functional protein complex—stabilized by the same forces as tertiary structure but operating between separate chains. Examples include hemoglobin (four subunits) and DNA polymerase (multiple subunits). This level requires multiple polypeptides and doesn't describe folding within a single chain's backbone.
Conclusion:
Protein structural hierarchy progresses from linear sequence (primary) through local backbone folding (secondary) to global chain conformation (tertiary) and multi-subunit assembly (quaternary). Secondary structure uniquely defines the repetitive coils (alpha-helices) and folds (beta-sheets) arising from backbone hydrogen bonding—distinct from primary sequence linearity, tertiary side chain interactions, or quaternary subunit assembly. Option B correctly identifies secondary structure as the level specifically characterized by polypeptide backbone folding patterns fundamental to protein architecture and function. Understanding this hierarchy proves essential for predicting protein behavior, designing drugs that target specific structural motifs, and interpreting mutations that disrupt folding at particular hierarchical levels.
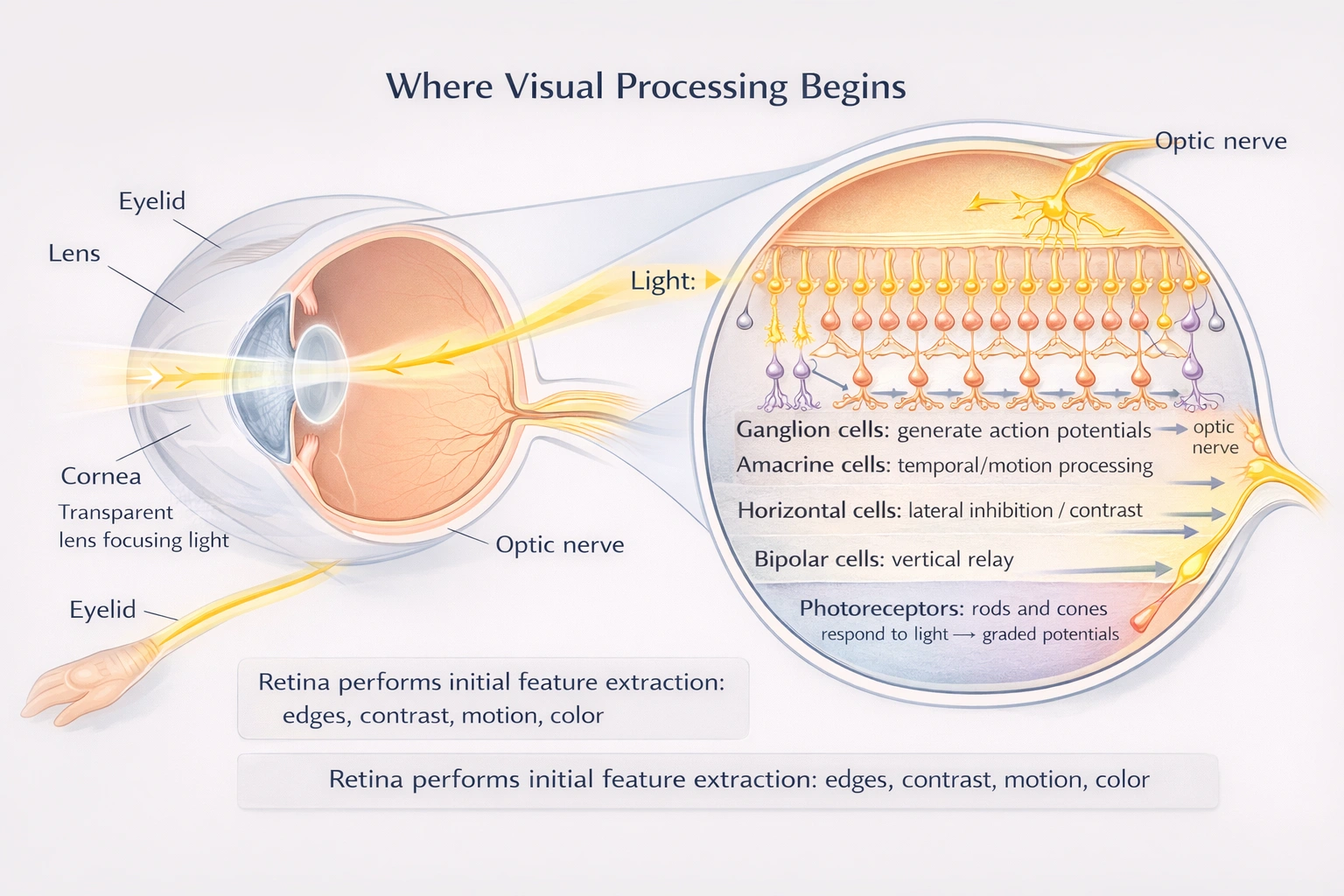
In which part of the eye does visual processing begin?
A.
Cornea
B. Optic nerve
C. Retina
D. Eyelid
Rationale
Visual processing initiates in the retina where photoreceptors (rods and cones) transduce light energy into neural signals, followed by initial processing through bipolar, horizontal, amacrine, and ganglion cells before transmission via the optic nerve.
The retina functions as neural tissue extending from the diencephalon—not merely a passive light receptor—performing sophisticated preprocessing including contrast enhancement, edge detection, and adaptation before signals reach the brain.
A) Cornea
The cornea serves as a transparent refractive structure focusing light onto the retina but contains no neural elements for signal transduction or processing. It functions purely as an optical component (providing ~70% of eye's refractive power) without photoreceptors, neurons, or synaptic connections—making it incapable of initiating visual processing.
B) Optic nerve
The optic nerve (cranial nerve II) transmits processed visual information from retinal ganglion cells to the brain but performs no processing itself. Composed of ganglion cell axons and glial cells without synapses, it functions as a transmission cable—not a processing center. Damage causes signal transmission failure but doesn't alter processing mechanisms.
C) Retina
The retina contains multiple neural layers executing initial visual processing:
• Photoreceptors (rods/cones) convert photons to graded potentials via photopigment bleaching
• Bipolar cells relay signals vertically while implementing center-surround receptive fields
• Horizontal cells mediate lateral inhibition between photoreceptors/bipolars for contrast enhancement
• Amacrine cells process temporal aspects and motion detection in inner plexiform layer
• Ganglion cells integrate inputs and generate action potentials encoding features like edges, motion direction, and color opponency
This multilayered processing extracts fundamental visual features before signals leave the eye—demonstrating the retina's role as the true initiation site of visual processing rather than a simple camera film.
D) Eyelid
Eyelids provide mechanical protection, distribute tears for corneal lubrication, and regulate light entry through blinking—but contain no visual processing structures. Their sensory innervation (trigeminal nerve) detects touch/pain but doesn't contribute to image formation or visual signal processing.
Conclusion:
Visual processing begins with phototransduction in retinal photoreceptors, immediately followed by neural computations within retinal circuitry that extract essential features before signals reach the brain. The retina's sophisticated preprocessing—demonstrated by phenomena like Mach bands (enhanced edge contrast from lateral inhibition) and adaptation to ambient light levels—establishes it as the true starting point of visual processing. Option C correctly identifies the retina's dual role as both transducer and initial processor, distinguishing it from purely optical structures (cornea), transmission pathways (optic nerve), or protective mechanisms (eyelids). This understanding proves essential for diagnosing visual disorders—retinal diseases (macular degeneration) impair processing initiation, while optic nerve damage (glaucoma) disrupts transmission of already-processed signals.
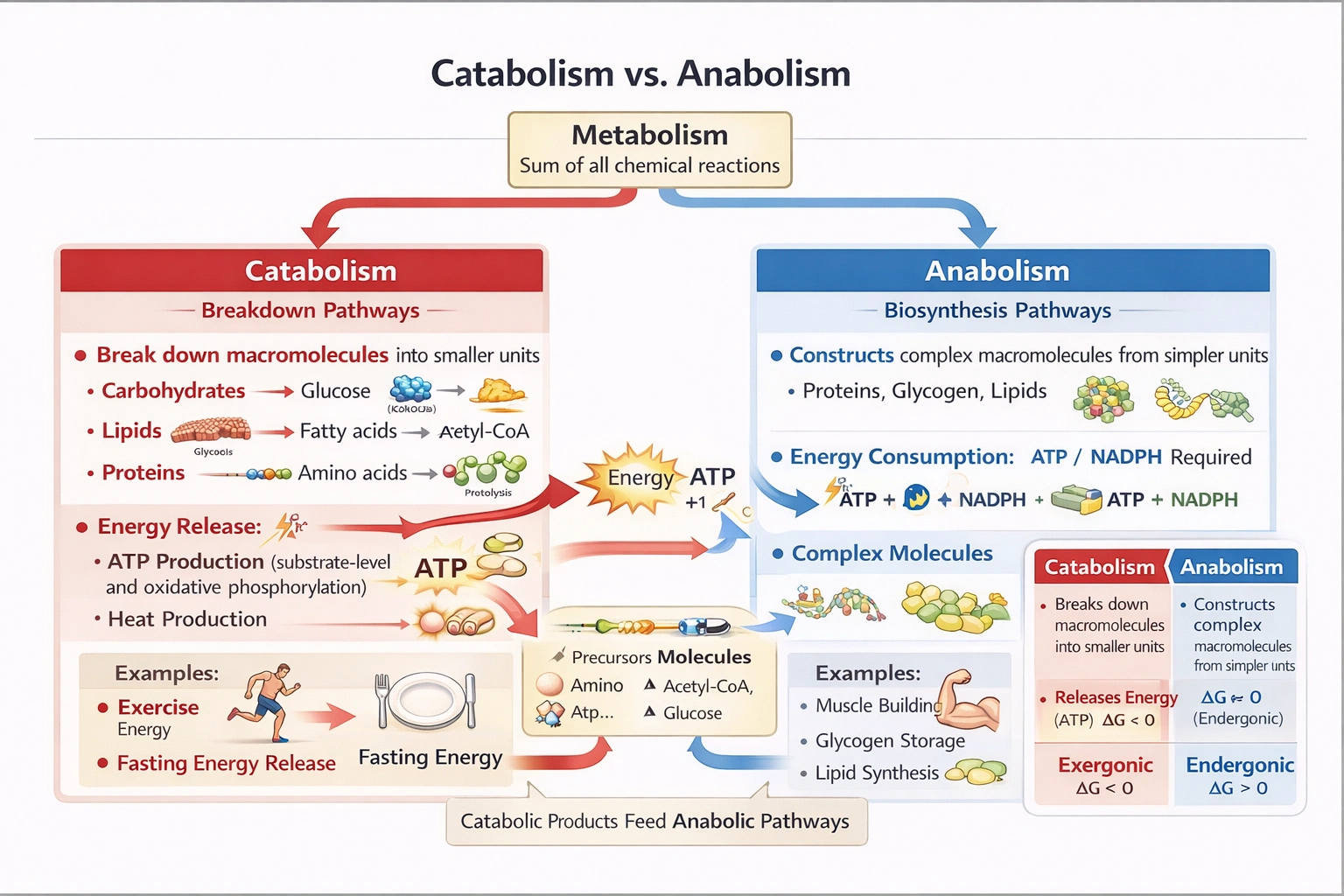
The process of breaking large molecules into smaller molecules to provide energy is known as which of the following?
A.
Metabolism
B. Bioenergetics
C. Anabolism
D. Catabolism
Rationale
Catabolism encompasses enzymatic breakdown of complex macromolecules (carbohydrates, lipids, proteins) into simpler units with concomitant energy release—either captured as ATP or dissipated as heat—fueling cellular work and anabolic processes.
Catabolic pathways follow thermodynamic principles where bond energy stored in complex molecules exceeds that in breakdown products, yielding negative ΔG (exergonic reactions) that drive ATP synthesis through substrate-level phosphorylation or oxidative phosphorylation.
A) Metabolism
Metabolism represents the sum total of all chemical reactions in an organism—encompassing both catabolism (breakdown) and anabolism (synthesis). While catabolism constitutes a metabolic subset, metabolism itself isn't specific to energy-yielding breakdown processes. Using "metabolism" to describe only catabolism misrepresents its comprehensive scope including energy-consuming biosynthetic pathways.
B) Bioenergetics
Bioenergetics studies energy flow and transformation in living systems—analyzing thermodynamic principles governing metabolic reactions. It's a scientific discipline examining energy relationships, not a specific metabolic process. Confusing the field of study with the process itself represents a category error—similar to calling "respiration" the study of breathing rather than the physiological process.
C) Anabolism
Anabolism describes energy-consuming biosynthetic pathways building complex molecules from simpler precursors (e.g., protein synthesis from amino acids, glycogen formation from glucose). These endergonic reactions require ATP hydrolysis or reducing power (NADPH) input—opposite to energy-yielding catabolic processes. Anabolism and catabolism represent complementary metabolic arms maintaining dynamic equilibrium.
D) Catabolism
Catabolism includes:
• Glycolysis: Glucose → 2 pyruvate + 2 ATP (net) + 2 NADH
• Beta-oxidation: Fatty acids → acetyl-CoA + NADH + FADH₂
• Proteolysis: Proteins → amino acids → entry into Krebs cycle via various intermediates
• Oxidative phosphorylation: NADH/FADH₂ oxidation driving proton gradient for ATP synthesis
These pathways release energy by breaking high-energy bonds in macromolecules, with approximately 40% captured as ATP and 60% dissipated as heat—essential for maintaining body temperature and powering cellular work. Catabolism provides both energy currency (ATP) and precursor molecules for anabolic pathways.
Conclusion:
Catabolism specifically defines energy-yielding molecular breakdown processes fundamental to cellular energetics—distinct from metabolism's comprehensive scope (A), bioenergetics' analytical framework (B), or anabolism's energy-consuming synthesis (C). Option D precisely identifies the process described, reflecting a cornerstone concept in biochemistry where catabolic pathways supply the thermodynamic driving force for life's energy requirements. Understanding this distinction proves essential for comprehending metabolic disorders (e.g., diabetes involves catabolic/anabolic imbalance), nutritional science (caloric value reflects catabolic energy yield), and exercise physiology (intensity determines catabolic substrate utilization—carbohydrates vs. fats).
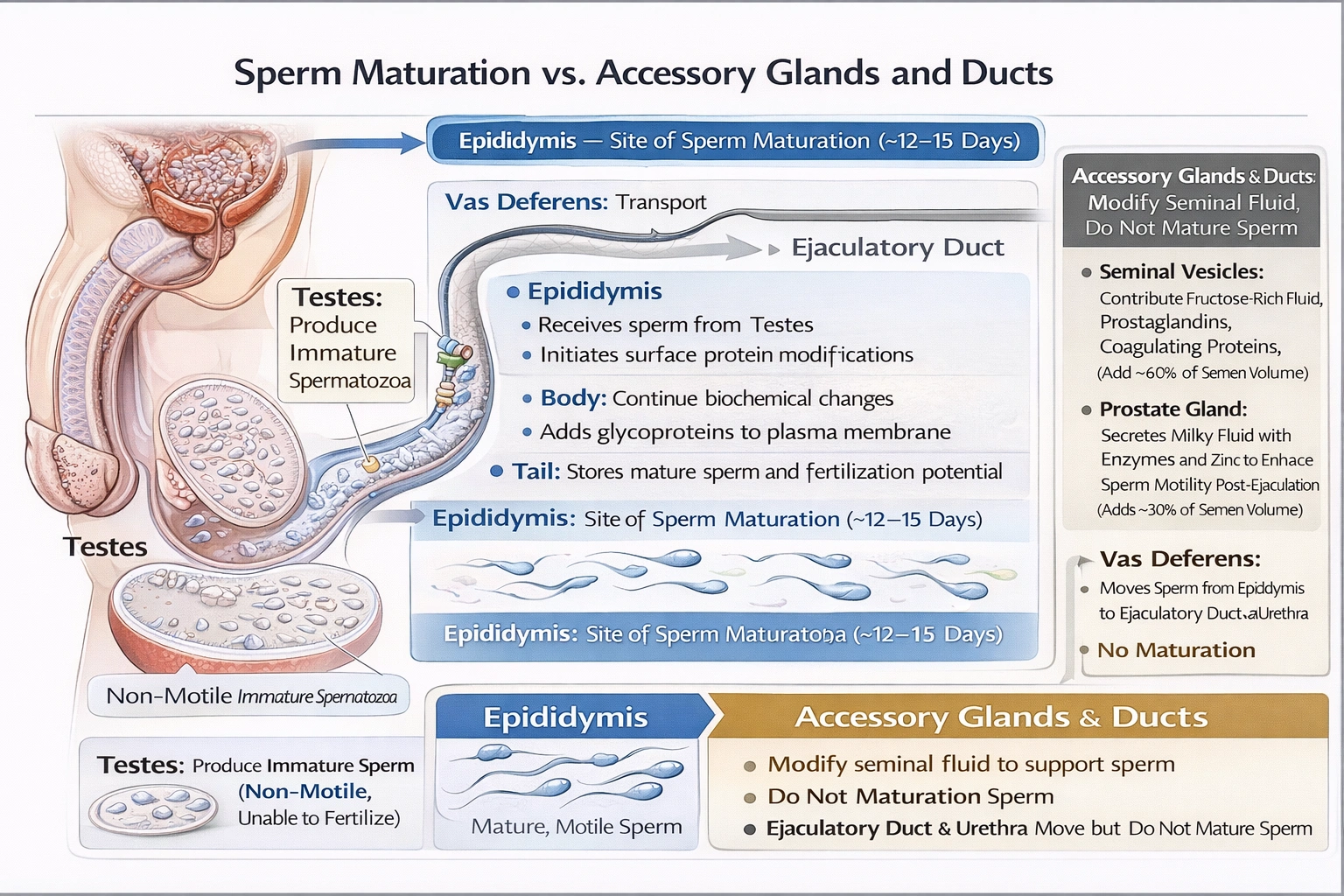
Where does sperm maturation take place in the male reproductive system?
A.
Seminal vesicles
B. Prostate gland
C. Epididymis
D. Vas Deferens
Rationale
Sperm maturation occurs within the epididymis—a highly coiled duct where immature, non-motile spermatozoa from the testes undergo biochemical and physiological changes over approximately 12–15 days to acquire motility capacity and fertilization potential.
The epididymis provides a specialized microenvironment with precise pH, ion concentrations, and secreted proteins that modify sperm surface properties, compact chromatin further, and prime flagellar function—transforming testicular sperm into functionally mature gametes capable of progressive motility and zona pellucida penetration.
A) Seminal vesicles
Seminal vesicles contribute 60–70% of semen volume—secreting a viscous, fructose-rich fluid containing prostaglandins, ascorbic acid, and coagulating proteins that nourish sperm and facilitate uterine transport. They don't participate in sperm maturation; their secretions mix with already-mature sperm during emission but provide no maturation environment. Seminal vesicle fluid lacks the prolonged contact time and specific molecular factors required for sperm maturation processes.
B) Prostate gland
The prostate secretes a milky, slightly acidic fluid containing proteolytic enzymes (PSA), citric acid, and zinc that initially coagulate then subsequently liquefy semen—enhancing sperm motility post-ejaculation. Like seminal vesicles, the prostate contributes to seminal fluid composition but doesn't provide the extended residence time (days) required for maturation. Prostatic secretions contact sperm only briefly during emission/ejaculation—insufficient for maturation processes requiring sustained epididymal exposure.
C) Epididymis
The epididymis consists of three regions with sequential maturation functions:
• Head (caput): Receives immotile sperm from efferent ductules; initiates surface protein modification
• Body (corpus): Continues biochemical changes including glycoprotein addition to plasma membrane
• Tail (cauda): Stores mature sperm (up to several weeks) while maintaining viability through low pH (6.5) and low oxygen tension
Key maturation changes include:
• Acquisition of progressive motility potential (activated only upon ejaculation by seminal fluid components)
• Membrane remodeling enabling zona pellucida recognition/binding
• Cytoplasmic droplet migration and eventual shedding
• Chromatin stabilization through disulfide bond formation in protamines
Sperm removed directly from testes cannot fertilize ova; only epididymal transit confers fertilization competence.
D) Vas Deferens
The vas deferens (ductus deferens) functions as a muscular transport conduit moving mature sperm from the epididymis tail to the ejaculatory duct during emission—via peristaltic contractions. It provides no secretory environment for maturation; sperm transit takes minutes to hours—far too brief for the days-long maturation process. The vas deferens stores some sperm in its ampulla but doesn't modify their functional properties beyond temporary storage.
Conclusion:
Sperm maturation requires prolonged exposure (12–15 days) to the epididymis' specialized luminal environment—making it the exclusive site where testicular sperm acquire motility potential and fertilization capacity. Accessory glands (seminal vesicles, prostate) contribute to seminal fluid but don't participate in maturation; the vas deferens serves only as a transport conduit. Option C correctly identifies the epididymis as the maturation site—a critical distinction for understanding male infertility (epididymal obstruction causes obstructive azoospermia) and developing male contraceptives targeting epididymal function. Without epididymal transit, sperm remain functionally immature despite normal testicular production.
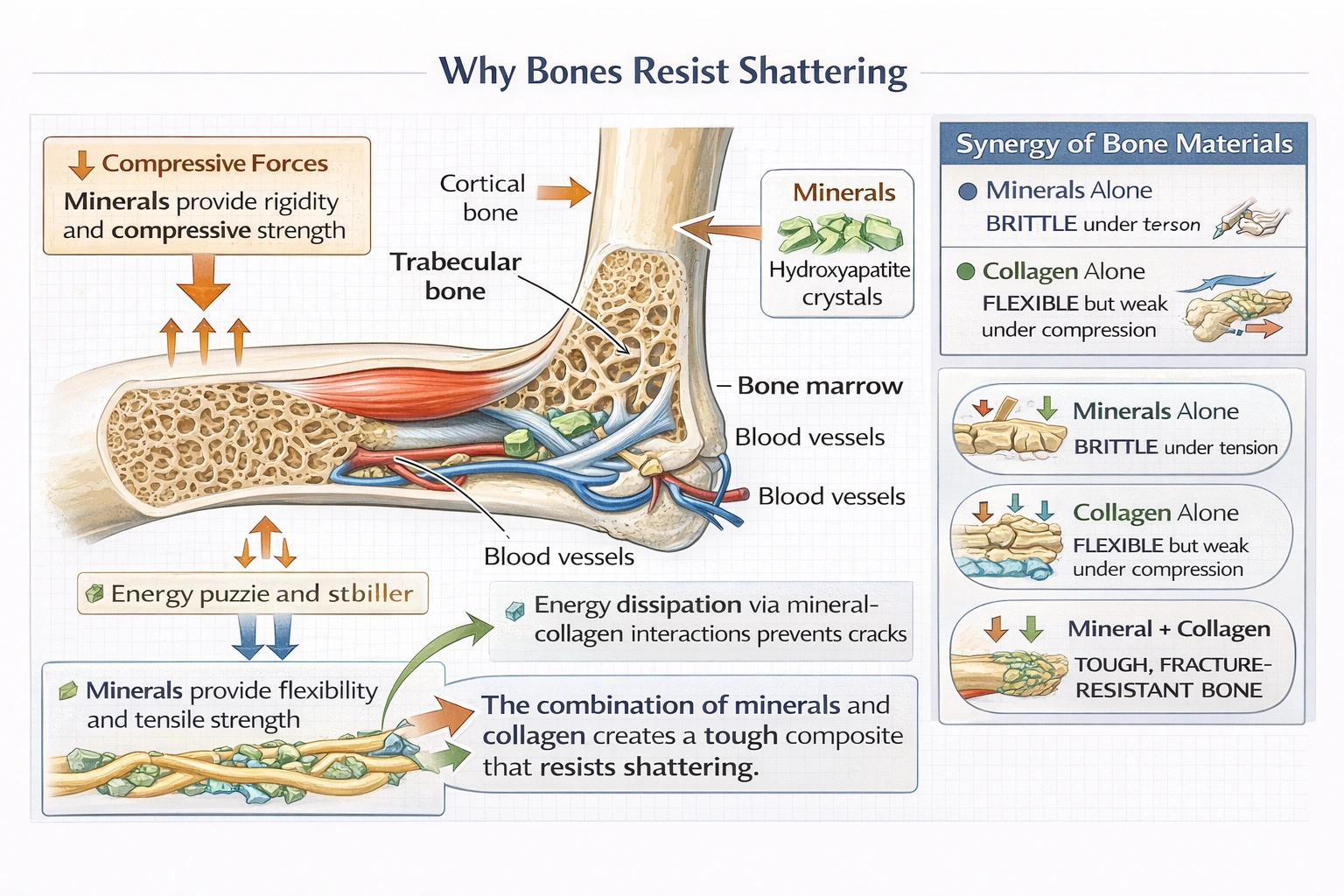
What makes bone resistant to shattering?
A.
The calcium salts deposited in the bone
B. The collagen fibers
C. The bone marrow and network of blood vessels
D. The intricate balance of minerals and collagen fibers
Rationale
Bone's resistance to shattering arises from the composite material structure combining brittle hydroxyapatite mineral crystals (providing compressive strength and rigidity) with flexible type I collagen fibers (providing tensile strength and elasticity)—creating a biomaterial stronger than either component alone through synergistic interaction.
This mineral-collagen composite follows principles of composite material engineering where a brittle matrix reinforced with flexible fibers achieves optimal mechanical properties—resisting both compression-induced crushing and tension-induced fracture through complementary material behaviors.
A) The calcium salts deposited in the bone
Hydroxyapatite crystals [Caâ‚â‚€(POâ‚„)₆(OH)â‚‚] provide exceptional compressive strength (resistance to squeezing forces) and rigidity—enabling bones to support body weight without deforming. However, mineralized tissue alone is brittle and shatters easily under tension or bending forces (like chalk or untempered ceramic). Demineralized bone (treated with acid) becomes rubbery and flexible but cannot resist compression—demonstrating minerals alone cannot prevent shattering under all loading conditions.
B) The collagen fibers
Type I collagen fibrils provide tensile strength (resistance to pulling forces) and elasticity—allowing bone to absorb energy through slight deformation before fracturing. Collagen's triple-helix structure with covalent cross-links creates flexible fibers that resist crack propagation. However, collagen alone lacks rigidity—demineralized bone bends excessively under load without maintaining structural integrity. Pure collagen cannot prevent crushing under compressive loads despite excellent shatter resistance in tension.
C) The bone marrow and network of blood vessels
Bone marrow (hematopoietic or fatty tissue) and vasculature provide metabolic support, nutrient delivery, and blood cell production—but contribute negligibly to mechanical strength. Marrow occupies medullary cavities and trabecular spaces without reinforcing the cortical bone matrix where mechanical loads are borne. Vascular channels actually create stress concentrators that could theoretically weaken bone if not compensated by surrounding matrix strength. These components support bone vitality but not fracture resistance directly.
D) The intricate balance of minerals and collagen fibers
Bone's exceptional fracture resistance emerges from the nanoscale integration of hydroxyapatite platelets (50–70% by weight) embedded within and between collagen fibrils:
• Minerals resist compressive forces and prevent excessive deformation
• Collagen resists tensile forces and arrests crack propagation through fiber bridging
• Interface between components enables stress transfer while allowing slight sliding that dissipates energy
This composite structure gives bone a toughness (energy required to fracture) approximately 10× greater than pure hydroxyapatite and 100× greater than pure collagen—demonstrating true synergy. Aging or disease disrupting this balance causes pathology: osteoporosis reduces mineral density increasing crush susceptibility; osteogenesis imperfecta produces defective collagen causing brittle bones prone to shattering despite normal mineralization.
Conclusion:
Bone's shatter resistance depends fundamentally on the composite material properties arising from mineral-collagen integration—not either component alone. Option D correctly identifies this synergistic balance as the source of bone's remarkable mechanical performance—combining the compressive strength of ceramics with the tensile toughness of polymers in a naturally engineered composite. Options A and B describe essential but insufficient individual components; option C describes metabolic support structures irrelevant to mechanical properties. Understanding this composite nature proves essential for treating bone diseases (bisphosphonates target mineralization; collagen disorders require different approaches) and developing biomimetic materials for orthopedic implants that replicate bone's fracture-resistant architecture.
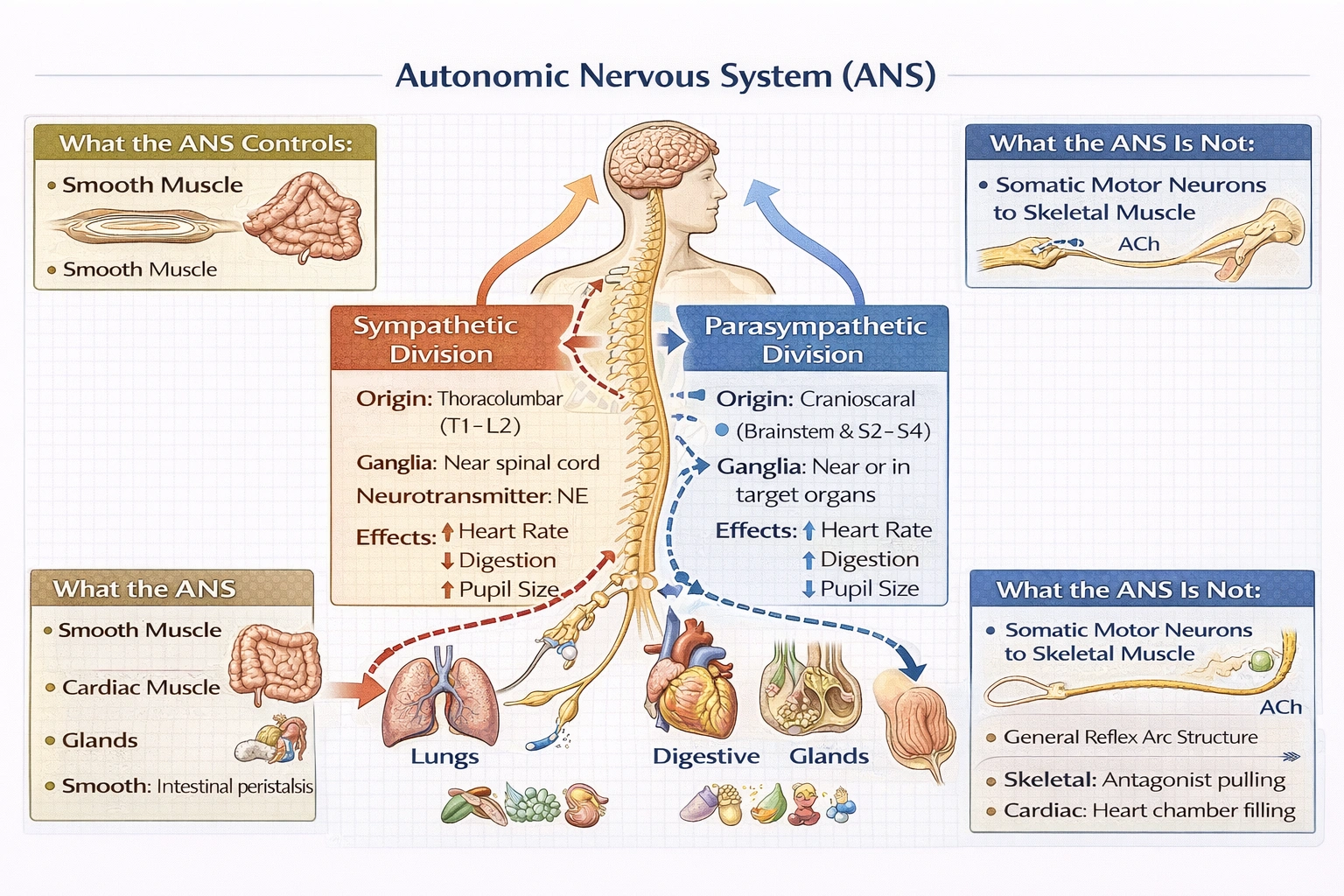
Which of the following about the autonomic nervous system (ANS) is true?
A.
It controls the reflex arc
B. It contains motor (efferent) neurons
C. It contains sensory (afferent) neurons
D. It contains both parasympathetic nerves and sympathetic nerves
Rationale
The autonomic nervous system comprises both parasympathetic and sympathetic divisions that function antagonistically to regulate visceral functions—representing its defining organizational feature as a dual-innervation control system for homeostasis.
The ANS maintains internal equilibrium through complementary parasympathetic ("rest and digest") and sympathetic ("fight or flight") outputs that modulate heart rate, digestion, respiration, and other involuntary processes—often with opposing effects on the same target organs to enable precise physiological control.
A) It controls the reflex arc
Reflex arcs involve sensory input → integration (spinal cord/brainstem) → motor output—but aren't exclusively autonomic. Somatic reflexes (e.g., patellar reflex) use somatic motor neurons to skeletal muscles. Autonomic reflexes (e.g., baroreceptor reflex) do involve ANS efferents, but the ANS doesn't "control" reflex arcs as a defining characteristic—reflexes represent neural circuit patterns that may or may not involve autonomic components. This statement overgeneralizes and misattributes reflex control exclusively to ANS.
B) It contains motor (efferent) neurons
The ANS does contain visceral efferent neurons—but so does the somatic nervous system (somatic motor neurons to skeletal muscles). While true, this isn't the defining or most comprehensive characteristic distinguishing the ANS. The ANS is specifically characterized by its two-division organization (sympathetic/parasympathetic) and involuntary control—not merely efferent function shared with somatic system. This statement is accurate but incomplete as a defining feature.
C) It contains sensory (afferent) neurons
Visceral sensory (afferent) neurons travel alongside autonomic nerves but technically belong to the general sensory division of the peripheral nervous system—not the ANS proper. The ANS is defined as the efferent (motor) division controlling visceral effectors; afferent limbs of autonomic reflexes are classified separately as visceral sensory pathways. Including afferent neurons in ANS definition confuses efferent control systems with sensory input pathways—though anatomically associated, they're functionally and classificationally distinct.
D) It contains both parasympathetic nerves and sympathetic nerves
The ANS is fundamentally organized into two anatomically and functionally distinct divisions:
• Sympathetic division: Thoracolumbar origin (T1-L2), short preganglionic/long postganglionic fibers, ganglia near spinal cord, widespread effects via norepinephrine release mobilizing energy resources during stress.
• Parasympathetic division: Craniosacral origin (brainstem CN III, VII, IX, X; S2-S4), long preganglionic/short postganglionic fibers, ganglia near or within target organs, localized effects via acetylcholine release—conserving energy during rest.
Most visceral organs receive dual innervation enabling precise control (e.g., sympathetic increases heart rate, parasympathetic decreases it). This dual-division organization constitutes the ANS's defining structural and functional characteristic—distinguishing it from the single-division somatic motor system.
Conclusion:
While the ANS contains efferent neurons (B) and associates with visceral afferents (C), its defining organizational feature is the complementary sympathetic-parasympathetic division structure enabling antagonistic control of visceral functions. Option D correctly identifies this fundamental characteristic that distinguishes ANS organization from other nervous system components. Option A misattributes reflex control exclusively to ANS. Understanding this dual-innervation principle proves essential clinically: autonomic dysfunction may affect one division preferentially (e.g., diabetic neuropathy damaging parasympathetic fibers causing gastroparesis), and pharmacological agents target specific divisions (beta-blockers inhibiting sympathetic cardiac effects). The sympathetic-parasympathetic balance represents homeostasis's neural substrate—making option D the most accurate and comprehensive statement about ANS organization.
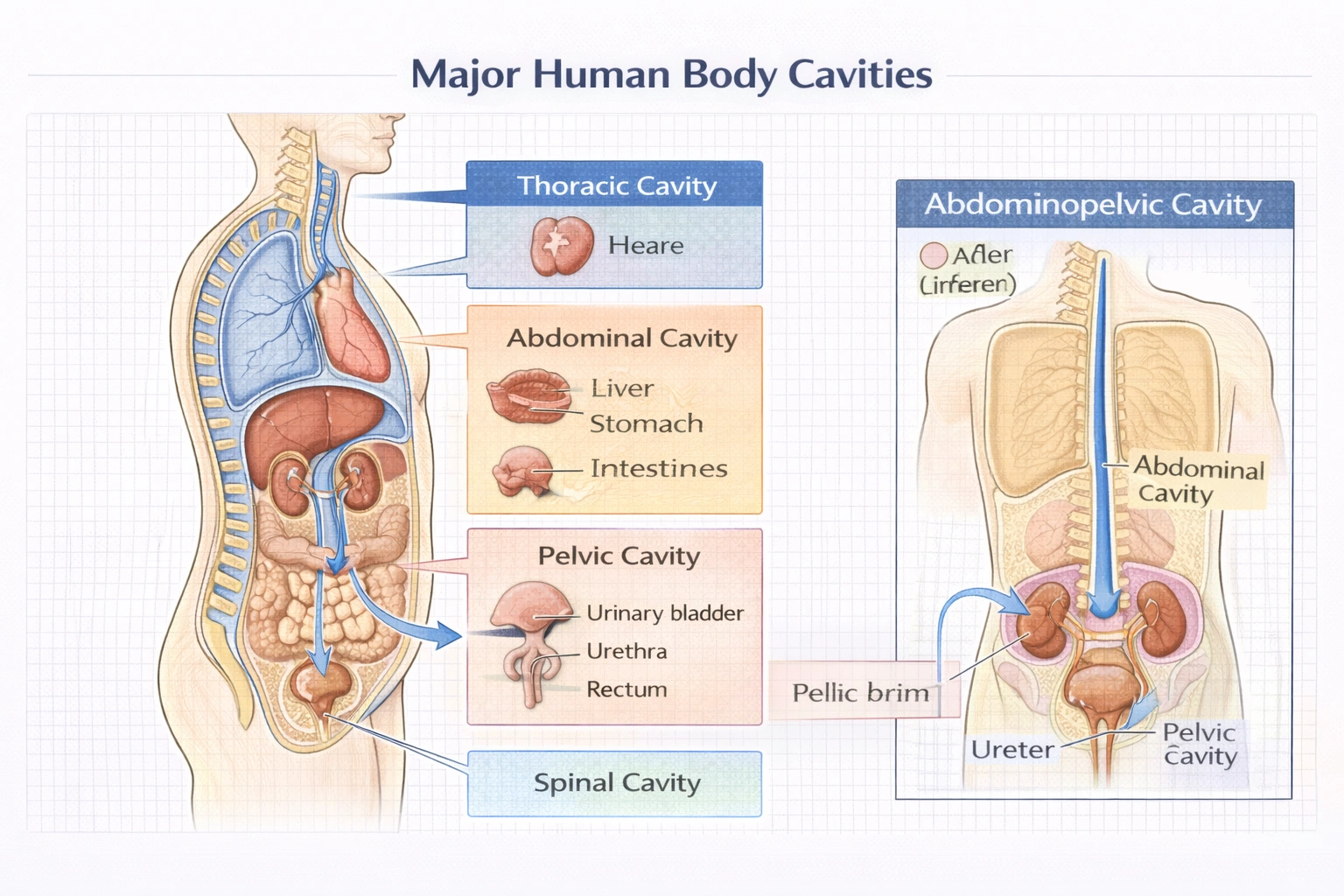
Which of the following is the body cavity that contains the urinary bladder urethra and ureters?
A.
The thoracic cavity
B. The pelvic cavity
C. The abdominal cavity
D. The spinal cavity
Rationale
The pelvic cavity houses the urinary bladder, proximal urethra, and distal portions of the ureters bounded by the pelvic brim superiorly, pelvic bones laterally, and pelvic floor muscles inferiorly, providing structural protection and spatial organization for urogenital and lower digestive organs.
The pelvic cavity represents the inferior compartment of the abdominopelvic cavity, separated from the abdominal cavity by the pelvic brim (anatomical landmark formed by sacral promontory and arcuate line of ilium)—though functionally continuous, these compartments contain distinct organ sets with different clinical implications.
A) The thoracic cavity
The thoracic cavity contains heart, lungs, esophagus, and major vessels—separated from abdominopelvic cavity by the diaphragm. Urinary structures reside inferior to the diaphragm and have no anatomical presence in the thorax. Thoracic cavity pressure dynamics (negative intrapleural pressure) differ fundamentally from pelvic pressure environments—making this option anatomically impossible.
B) The pelvic cavity
The pelvic cavity specifically contains:
• Urinary organs: Urinary bladder (when empty or moderately full), entire female urethra, proximal male urethra, distal ureters as they enter bladder wall at ureterovesical junctions
• Reproductive organs: Uterus, vagina, ovaries (female); prostate, seminal vesicles (male)
• Digestive organs: Rectum and anal canal
The bony pelvis provides rigid protection for these structures, while pelvic floor muscles (levator ani, coccygeus) support organs against gravity and control sphincter functions. During extreme bladder distension, the bladder dome may extend into the abdominal cavity—but its primary anatomical location remains pelvic.
C) The abdominal cavity
The abdominal cavity contains stomach, liver, intestines, kidneys, spleen, and pancreas—but not the bladder or urethra under normal conditions. The kidneys and proximal ureters reside retroperitoneally within the abdominal cavity, but the distal ureters, bladder, and urethra descend through the pelvic brim into the pelvic cavity. Confusing abdominal and pelvic compartments is common but anatomically significant—pelvic fractures risk bladder/urethral injury while abdominal trauma typically affects intestines/liver.
D) The spinal cavity
The spinal (vertebral) cavity contains the spinal cord and meninges within the vertebral foramen—completely separate from urinary structures. No portion of the urinary drainage system passes through or resides within the spinal canal. This option confuses body cavity classifications entirely—spinal cavity belongs to the dorsal cavity system while urinary organs reside in ventral cavity subdivisions.
Conclusion:
The pelvic cavity specifically houses the terminal urinary structures (bladder, urethra, distal ureters) along with reproductive and lower digestive organs—distinguished from the abdominal cavity containing proximal urinary structures (kidneys, upper ureters). Option B correctly identifies the pelvic cavity as the anatomical compartment for these organs, while option C represents a common misconception confusing abdominal versus pelvic organ locations. Understanding this compartmentalization proves clinically essential: pelvic examinations assess bladder fullness, pelvic fractures risk urethral disruption (especially in males), and pelvic surgeries require different approaches than abdominal procedures. The pelvic brim serves as the critical anatomical boundary separating these functionally related but spatially distinct compartments within the larger abdominopelvic cavity.
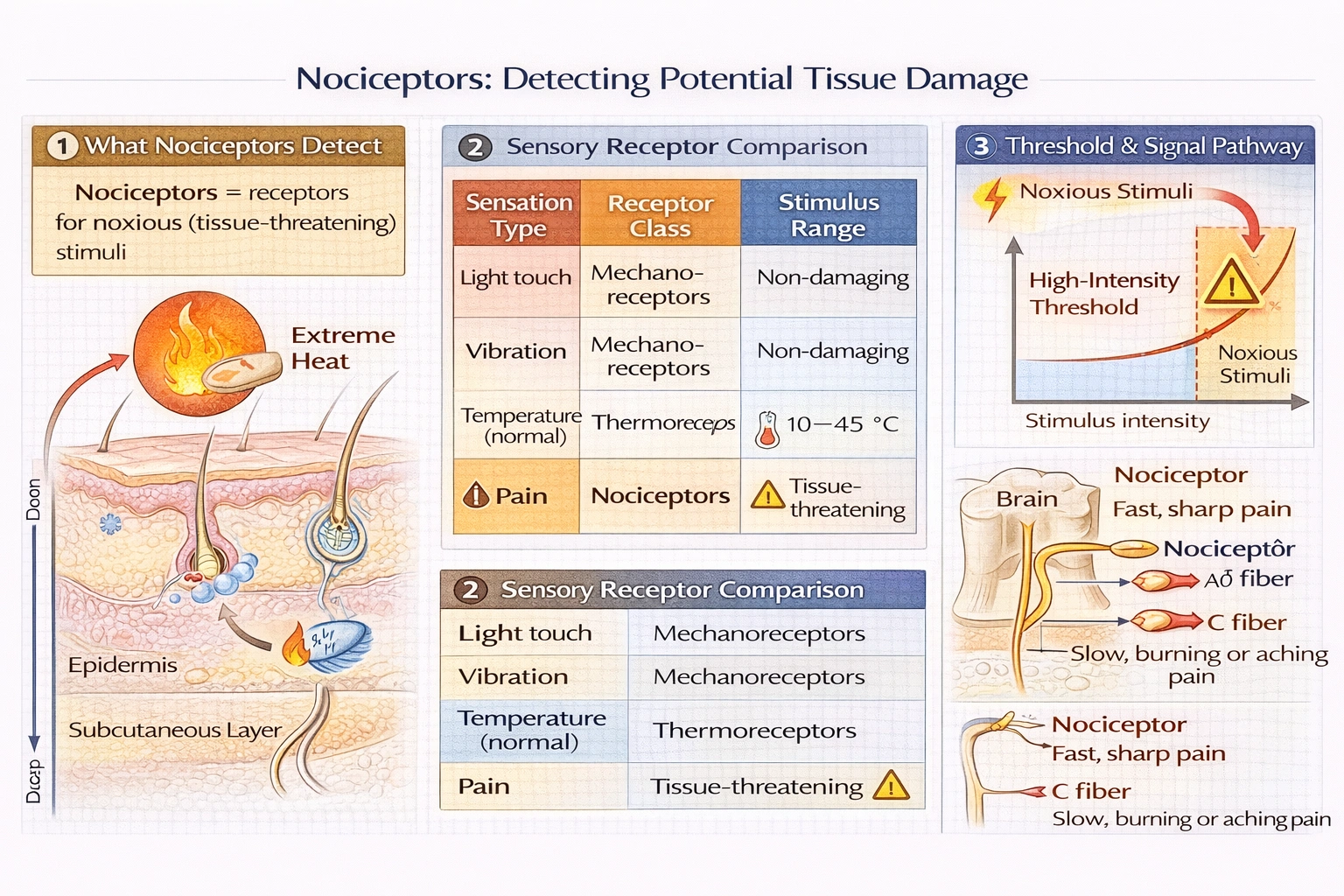
Nociceptors detect which of the following?
A.
Deep pressure
B. Vibration
C. Pain
D. Temperature
Rationale
Nociceptors are specialized sensory receptors that respond to potentially damaging (noxious) stimuli such as intense mechanical pressure, extreme heat or cold, and chemical irritants transducing them into pain signals via Aδ and C nerve fibers to alert the body of actual or threatened tissue damage.
A) Deep pressure
Deep pressure detection occurs through mechanoreceptors like Pacinian corpuscles (rapidly adapting vibration/pressure) and Ruffini endings (slowly adapting stretch)—not nociceptors. Nociceptors respond only to potentially damaging pressure intensities (e.g., pinch causing tissue deformation beyond physiological limits), not normal deep pressure sensations. Distinguishing normal pressure from painful pressure depends on stimulus intensity activating different receptor populations.
B) Vibration
Vibration detection involves rapidly adapting mechanoreceptors: Meissner's corpuscles (low-frequency) and Pacinian corpuscles (high-frequency). Nociceptors don't respond to vibration within normal ranges; only extremely intense vibrations causing tissue damage might activate nociceptors secondarily—but vibration itself isn't their stimulus modality.
C) Pain
Nociceptors specifically detect noxious stimuli that produce pain perception:
• Mechanical nociceptors: Intense pressure, pinch, cutting
• Thermal nociceptors: Temperatures <5°C or >45°C
• Polymodal nociceptors: Respond to multiple noxious modalities plus chemical irritants (bradykinin, histamine, H⺠ions from inflammation)
Nociceptor activation doesn't always produce conscious pain (e.g., during stress-induced analgesia), and pain can occur without peripheral nociceptor activation (central pain syndromes)—but nociceptors are the primary peripheral detectors of tissue-threatening stimuli that normally lead to pain perception. The term "nociceptor" literally means "pain receptor" (from Latin nocere = to hurt).
D) Temperature
Temperature detection involves thermoreceptors—distinct from nociceptors:
• Cold receptors: Activated 10–35°C (non-painful cooling)
• Warm receptors: Activated 30–45°C (non-painful warming)
• Cold nociceptors: Activated <5°C (painful cold)
• Heat nociceptors: Activated >45°C (painful heat)
Non-noxious temperature ranges activate dedicated thermoreceptors; only extreme temperatures activate thermal nociceptors signaling pain. Confusing thermoreceptors with nociceptors overlooks this critical intensity threshold distinguishing normal sensation from tissue threat detection.
Conclusion:
Nociceptors specifically detect stimuli intense enough to threaten tissue damage—transducing these noxious inputs into signals that typically produce pain perception. Option C correctly identifies pain detection as their primary function, distinguishing nociceptors from mechanoreceptors detecting non-noxious pressure/vibration (A, B) and thermoreceptors detecting non-noxious temperature ranges (D). Understanding this distinction proves clinically vital: neuropathic pain involves nociceptor dysfunction or sensitization (hyperalgesia), while loss of nociception (congenital insensitivity to pain) causes severe injury risk despite intact touch/temperature sensation. Nociceptors represent the peripheral gatekeepers of tissue integrity—activating protective withdrawal reflexes and conscious pain to prevent or minimize damage.
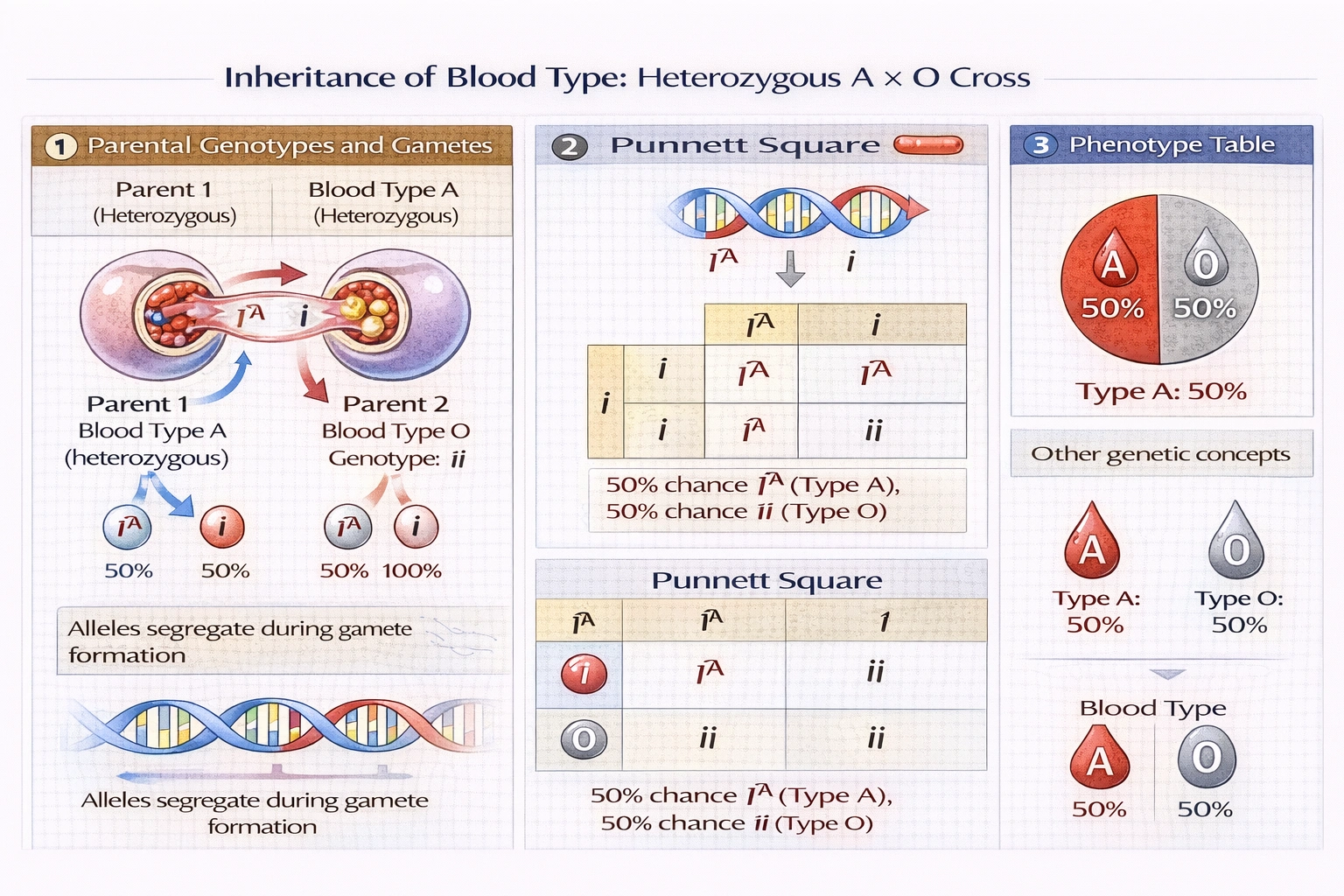
Blood type is a trait determined by multiple alleles, and two of them are co-dominant: Iᴬ codes for A blood and Iᴮ codes for B blood. i codes for O blood and is recessive to both. If an A heterozygote individual and an O individual have a child, what is the probability that the child will have A blood?
A.
25%
B. 50%
C. 75%
D. 100%
Rationale
The probability that the child will have blood type A is 50%, resulting from a cross between genotype Iá´¬i (heterozygous type A) and ii (type O) that produces offspring genotypes Iá´¬i (type A) and ii (type O) in equal 1:1 proportions.
Blood type inheritance follows codominance between Iᴬ and Iᴮ alleles with both dominant over recessive i—creating four phenotypes (A, B, AB, O) from six possible genotypes (IᴬIᴬ, Iᴬi, IᴮIᴮ, Iᴮi, IᴬIᴮ, ii) through Mendelian segregation of alleles during gamete formation.
Parental genotypes:
• Parent 1 (A heterozygote): Iᴬi → produces gametes Iᴬ or i (50% each)
• Parent 2 (O type): ii → produces gametes i only (100%)
Punnett square analysis:
i i
Iá´¬ Iá´¬i Iá´¬i
i ii ii
Offspring genotypes and phenotypes:
• Iᴬi (50% probability): Type A blood (Iᴬ dominant over i)
• ii (50% probability): Type O blood (no dominant allele present)
A) 25%
This would result from a cross between two heterozygotes (Iᴬi × Iᴬi) where 25% of offspring are IᴬIᴬ—but the O parent (ii) cannot contribute Iᴬ allele, making homozygous IᴬIᴬ offspring impossible. This option misapplies heterozygous cross ratios to a heterozygous × homozygous recessive cross.
B) 50%
Correct probability: Half the offspring inherit Iᴬ from the heterozygous parent and i from the O parent (Iᴬi = type A), while half inherit i from both parents (ii = type O). The 1:1 phenotypic ratio reflects the heterozygous parent's 50% chance of transmitting the Iᴬ allele versus i allele—standard for monohybrid test crosses.
C) 75%
This ratio occurs in heterozygous × heterozygous crosses (Iᴬi × Iᴬi) where 75% show dominant phenotype (IᴬIᴬ + Iᴬi)—but requires both parents to carry dominant allele. The O parent (ii) lacks Iᴬ allele entirely, making 75% impossible. This option incorrectly assumes O parent contributes dominant allele.
D) 100%
This would require both parents to be homozygous dominant (IᴬIᴬ × IᴬIᴬ) or one homozygous dominant and one heterozygous (IᴬIᴬ × Iᴬi)—but the O parent (ii) contributes only recessive i alleles. At least 50% of offspring must inherit i from both parents (ii = type O), making 100% type A impossible.
Conclusion:
The cross between heterozygous type A (Iᴬi) and type O (ii) follows a standard test cross pattern yielding 50% type A and 50% type O offspring—demonstrating Mendelian segregation where the heterozygous parent transmits dominant and recessive alleles with equal probability. Option B correctly calculates this probability, while other options misapply ratios from different parental genotype combinations. Understanding blood type inheritance proves clinically essential for transfusion medicine (predicting compatible donors/recipients) and paternity testing (excluding impossible parentage based on offspring blood types)—where precise allele transmission probabilities determine biological relationship likelihoods.
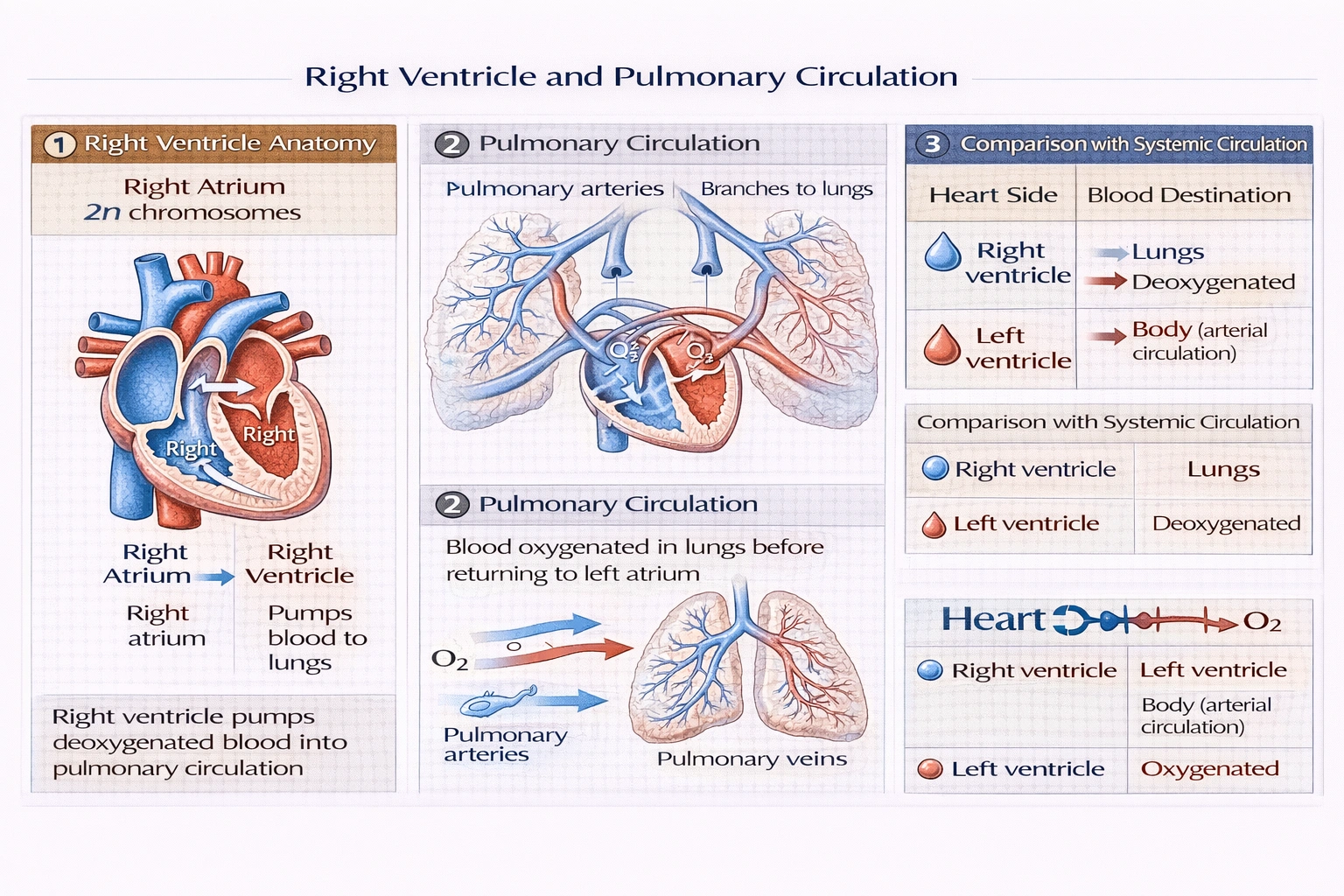
Blood leaves the right ventricle and goes to the
A.
lungs
B. kidneys
C. right atrium
D. arterial circulation to the body
Rationale
Blood leaving the right ventricle travels to the lungs. Deoxygenated blood ejected from the right ventricle enters the pulmonary artery, which branches to deliver blood to pulmonary capillaries surrounding alveoli where gas exchange occurs—oxygen diffuses into blood while carbon dioxide diffuses out—before oxygenated blood returns to the left atrium via pulmonary veins.
A) lungs
The pulmonary circulation pathway follows: right ventricle → pulmonary valve → pulmonary trunk → left/right pulmonary arteries → pulmonary capillaries (gas exchange) → pulmonary veins → left atrium. This low-pressure circuit (25/10 mmHg) contrasts with systemic circulation (120/80 mmHg), protecting delicate alveolar capillaries from high-pressure damage while enabling efficient oxygenation—essential for maintaining arterial oxygen saturation above 95%.
B) kidneys
Renal blood supply originates from the abdominal aorta via renal arteries—part of systemic circulation receiving oxygenated blood from the left ventricle. The right ventricle exclusively supplies pulmonary circulation; no direct vascular connection exists between right ventricle and kidneys. Renal perfusion requires oxygenated blood to support filtration functions—impossible with deoxygenated right ventricular output.
C) right atrium
Blood flows from right atrium to right ventricle through the tricuspid valve during ventricular diastole—not in reverse. The right ventricle's muscular contraction (systole) generates pressure exceeding atrial pressure, closing the tricuspid valve and preventing backflow. Anatomical one-way valves and pressure gradients enforce unidirectional flow: atria → ventricles → arteries—making retrograde flow from ventricle to atrium physiologically impossible under normal conditions.
D) arterial circulation to the body
Systemic arterial circulation receives oxygenated blood exclusively from the left ventricle via the aorta. The right ventricle's output enters pulmonary arteries carrying deoxygenated blood—mixing oxygenated and deoxygenated streams would catastrophically reduce oxygen delivery to tissues. Complete septation ensures right heart handles only pulmonary circulation while left heart manages systemic circulation—a fundamental design principle of the four-chambered heart.
Conclusion:
Pulmonary circulation represents the exclusive destination for right ventricular output—a specialized low-pressure circuit optimized for gas exchange without exposing delicate lung tissue to systemic pressures. Options B, C, and D confuse pulmonary and systemic circuits or violate unidirectional flow principles enforced by cardiac valves and pressure gradients. Understanding this separation proves essential clinically: right heart failure causes pulmonary edema (backed-up pulmonary circulation), while left heart failure produces systemic edema—requiring distinct therapeutic approaches based on circulatory compartmentalization.
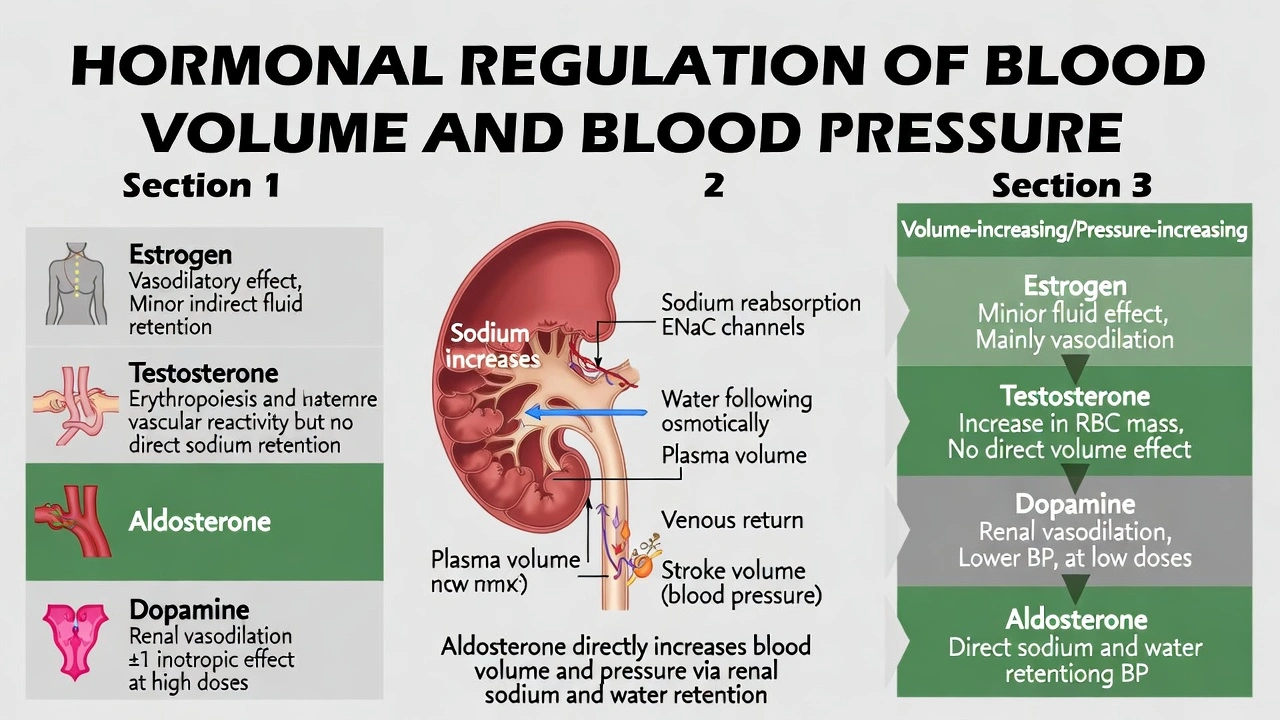
The hormone that can promote increased blood volume and increased blood pressure is
A.
estrogen
B. testosterone
C. aldosterone
D. dopamine
Rationale
Aldosterone increases blood volume and blood pressure. Aldosterone, a mineralocorticoid secreted by the adrenal cortex, enhances sodium reabsorption in renal distal tubules and collecting ducts—creating an osmotic gradient that drives water retention, thereby expanding plasma volume and elevating arterial pressure through increased cardiac preload and stroke volume.
A) estrogen
Estrogen influences vascular tone through nitric oxide modulation and affects renin-angiotensin system components, but doesn't directly regulate sodium/water balance for volume expansion. While estrogen therapy may cause mild fluid retention as a side effect, this represents a secondary pharmacological action—not a primary physiological mechanism for blood pressure regulation like aldosterone's direct renal effects. Estrogen's cardiovascular effects are predominantly vasodilatory, often lowering rather than raising blood pressure.
B) testosterone
Testosterone promotes erythropoiesis (increasing red blood cell mass) and may enhance vascular reactivity, but lacks direct renal sodium-retaining actions. Supraphysiological testosterone doses can elevate hematocrit and potentially increase viscosity-related pressure, but physiological testosterone levels don't significantly influence blood volume regulation. Unlike aldosterone, testosterone doesn't act on renal epithelial sodium channels (ENaC) to drive osmotic water retention—the primary mechanism for rapid blood volume expansion.
C) aldosterone
Aldosterone binds mineralocorticoid receptors in principal cells of renal collecting ducts, triggering synthesis and insertion of epithelial sodium channels (ENaC) and Naâº/K⺠ATPase pumps—increasing sodium reabsorption. Water follows osmotically via aquaporin-2 channels, expanding plasma volume by 5–10% within hours. This volume expansion increases venous return (preload), elevating stroke volume via Starling's law and subsequently raising arterial pressure. The renin-angiotensin-aldosterone system (RAAS) activates aldosterone release specifically in response to hypotension or volume depletion—making it the primary hormonal regulator of blood volume and pressure homeostasis.
D) dopamine
Dopamine functions as a neurotransmitter and renal vasodilator at low doses—actually decreasing blood pressure by dilating renal and mesenteric arteries to increase renal blood flow and sodium excretion. At high pharmacological doses, dopamine stimulates βâ‚-adrenergic receptors increasing heart rate and contractility, but this represents acute inotropic support—not sustained volume expansion. Dopamine lacks aldosterone's direct tubular actions for sodium/water retention and isn't part of physiological long-term blood pressure regulation.
Conclusion:
Aldosterone uniquely serves as the primary hormonal regulator of blood volume through direct renal sodium reabsorption—creating osmotic water retention that expands plasma volume and elevates pressure via hemodynamic mechanisms. Options A, B, and D either lack volume-expanding actions or produce opposite effects. Understanding aldosterone's role proves clinically essential: hyperaldosteronism (Conn's syndrome) causes hypertension and hypokalemia, while mineralocorticoid antagonists (spironolactone) treat heart failure by counteracting pathological volume retention—demonstrating this hormone's central importance in cardiovascular homeostasis.
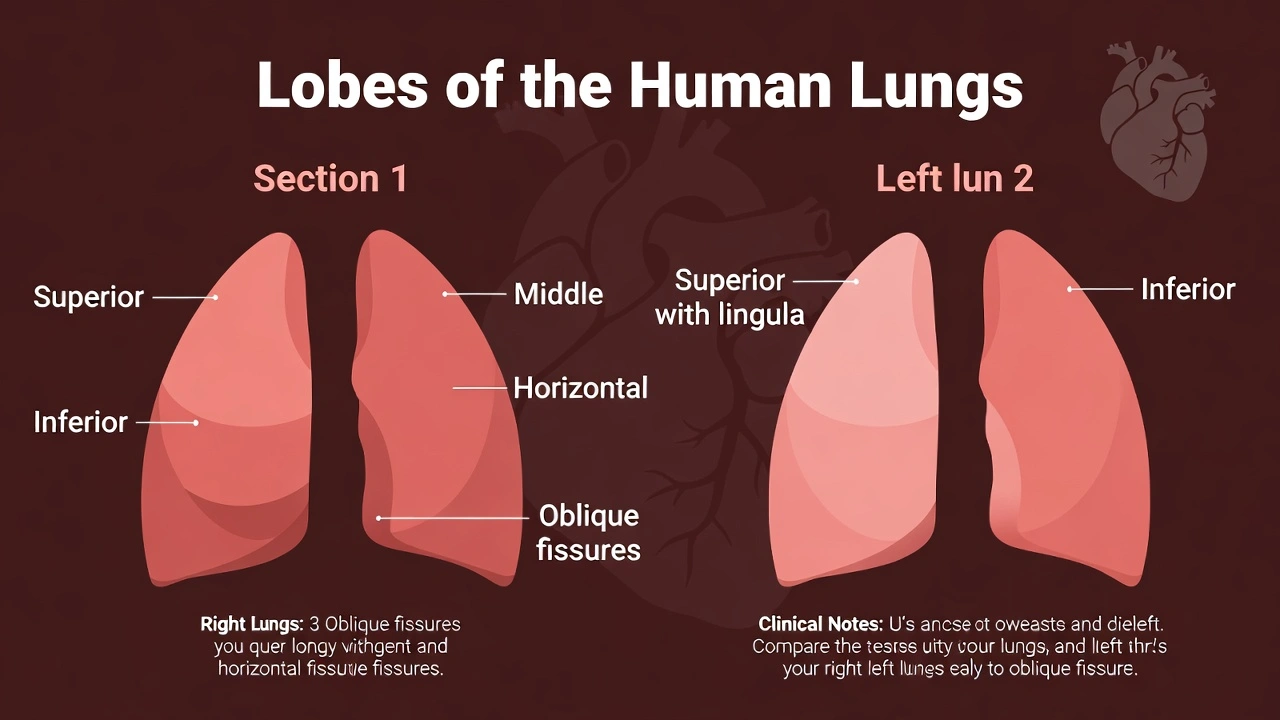
The right lung has ____ lobes and the left lung has __ lobes.
A.
2,3
B. 3,2
C. 3,3
D. 2,2
Rationale
The right lung has three lobes and the left lung has two lobes. Asymmetrical lung lobation accommodates the heart's leftward position within the thoracic cavity—the right lung's three lobes (superior, middle, inferior) maximize space utilization on the cardiac-free right side, while the left lung's two lobes (superior, inferior) with cardiac notch provide space for the heart's apex and left ventricle.
A) 2, 3
This reverses the actual anatomy—no human possesses a two-lobed right lung and three-lobed left lung. Such asymmetry would position the heart on the right side (dextrocardia), an extremely rare congenital condition occurring in approximately 1 in 12,000 individuals—typically associated with other congenital anomalies. Standard human anatomy universally features three right lobes and two left lobes regardless of cardiac position.
B) 3, 2
The right lung contains three lobes separated by oblique and horizontal fissures:
• Superior lobe: Anterior and apical segments
• Middle lobe: Medial and lateral segments (unique to right lung)
• Inferior lobe: Superior (apical) and basal segments
The left lung has two lobes separated by an oblique fissure:
• Superior lobe: Includes lingula (homologous to right middle lobe)
• Inferior lobe: Similar segmental anatomy to right inferior lobe
This asymmetry accommodates the heart's position—the left lung's cardiac notch and smaller size provide space for the heart, while the right lung's additional middle lobe utilizes space occupied by the liver's dome inferiorly.
C) 3, 3
Three lobes on both sides occurs only in some mammals (e.g., dogs) but not humans. Human left lung development suppresses the horizontal fissure due to cardiac constraint during embryogenesis—resulting in fusion of potential middle lobe tissue into the superior lobe (forming the lingula). Bilateral three-lobe anatomy would crowd the mediastinum, compressing the heart and great vessels—evolutionarily disadvantageous for upright posture and high metabolic demands.
D) 2, 2
Two lobes bilaterally characterizes fetal lung development before horizontal fissure formation on the right—but not mature human anatomy. Persistent bilateral two-lobe configuration would represent a developmental anomaly potentially causing respiratory compromise due to reduced surface area. Normal postnatal anatomy requires right middle lobe development for adequate gas exchange capacity matching metabolic needs.
Conclusion:
The 3:2 lobar asymmetry represents an evolutionary adaptation optimizing thoracic space utilization around the mediastinal structures—particularly accommodating the leftward cardiac position while maximizing respiratory surface area. Option B correctly identifies this universal human anatomical pattern, essential for clinical procedures: right middle lobe pneumonia requires different positioning for drainage than left lower lobe infections, and surgical approaches differ based on fissure anatomy. Misidentifying lobar configuration could lead to procedural errors during bronchoscopy, thoracentesis, or lobectomy—emphasizing the clinical importance of this fundamental anatomical distinction.
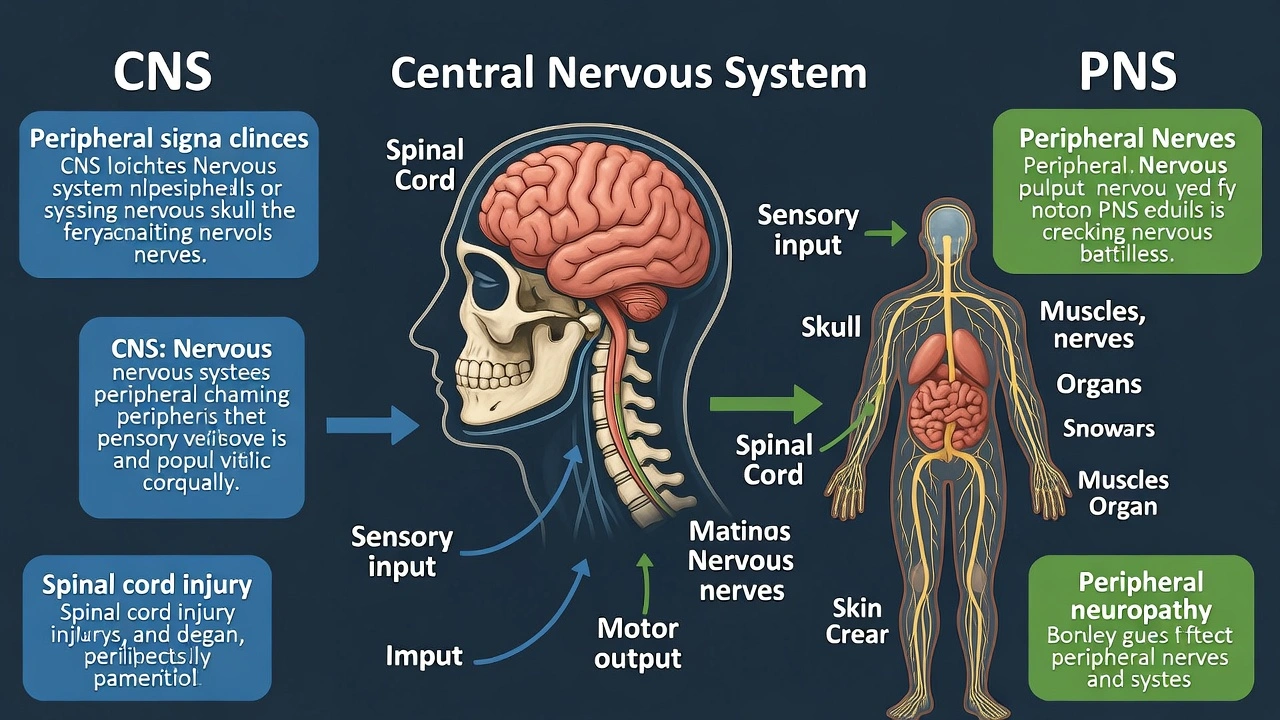
The central nervous system is composed of the ___ and the ___
A.
brain,spinal cord
B. brain, peripheral nerves
C. spinal cord, peripheral nerves
D. spinal cord, musculature system
Rationale
The central nervous system consists of the brain and spinal cord. The central nervous system (CNS) integrates and processes sensory information and coordinates bodily responses through its two structural components: the brain (encased in the cranium) and spinal cord (protected by the vertebral column). These structures receive input from and send output to the peripheral nervous system (PNS), which includes all nerves outside the CNS.
A) brain, spinal cord
This option correctly identifies the two exclusive components of the CNS. The brain processes complex functions (cognition, emotion, sensory integration), while the spinal cord transmits signals between brain and body and mediates reflex arcs. Together they form the body's central processing unit, distinct from peripheral nerves that extend to limbs and organs.
B) brain, peripheral nerves
Peripheral nerves belong to the peripheral nervous system (PNS), not the CNS. The PNS comprises cranial nerves (except optic and olfactory tracts within CNS) and spinal nerves that connect CNS to sensory receptors, muscles, and glands. Including peripheral nerves in CNS definition misrepresents fundamental neuroanatomical organization.
C) spinal cord, peripheral nerves
This option omits the brain—the primary CNS structure—and incorrectly includes peripheral nerves. Without the brain, the CNS would lack higher processing capabilities; peripheral nerves remain PNS components regardless of spinal cord association. This combination describes neither CNS nor a coherent anatomical system.
D) spinal cord, musculature system
The musculature system (skeletal, smooth, cardiac muscle) is an effector system controlled by the nervous system—not part of the CNS itself. Muscles receive motor innervation from peripheral nerves originating in CNS but constitute a separate organ system. Including musculature confuses neural control structures with their targets.
Conclusion:
The CNS exclusively comprises brain and spinal cord—protected within bony encasements and functionally distinct from the peripheral nervous system that extends throughout the body. Option A provides the anatomically precise definition essential for understanding neurological organization, clinical localization of lesions (e.g., spinal cord injury versus peripheral neuropathy), and neuroimaging interpretation. Misidentifying CNS components could lead to diagnostic errors in neurological assessment.
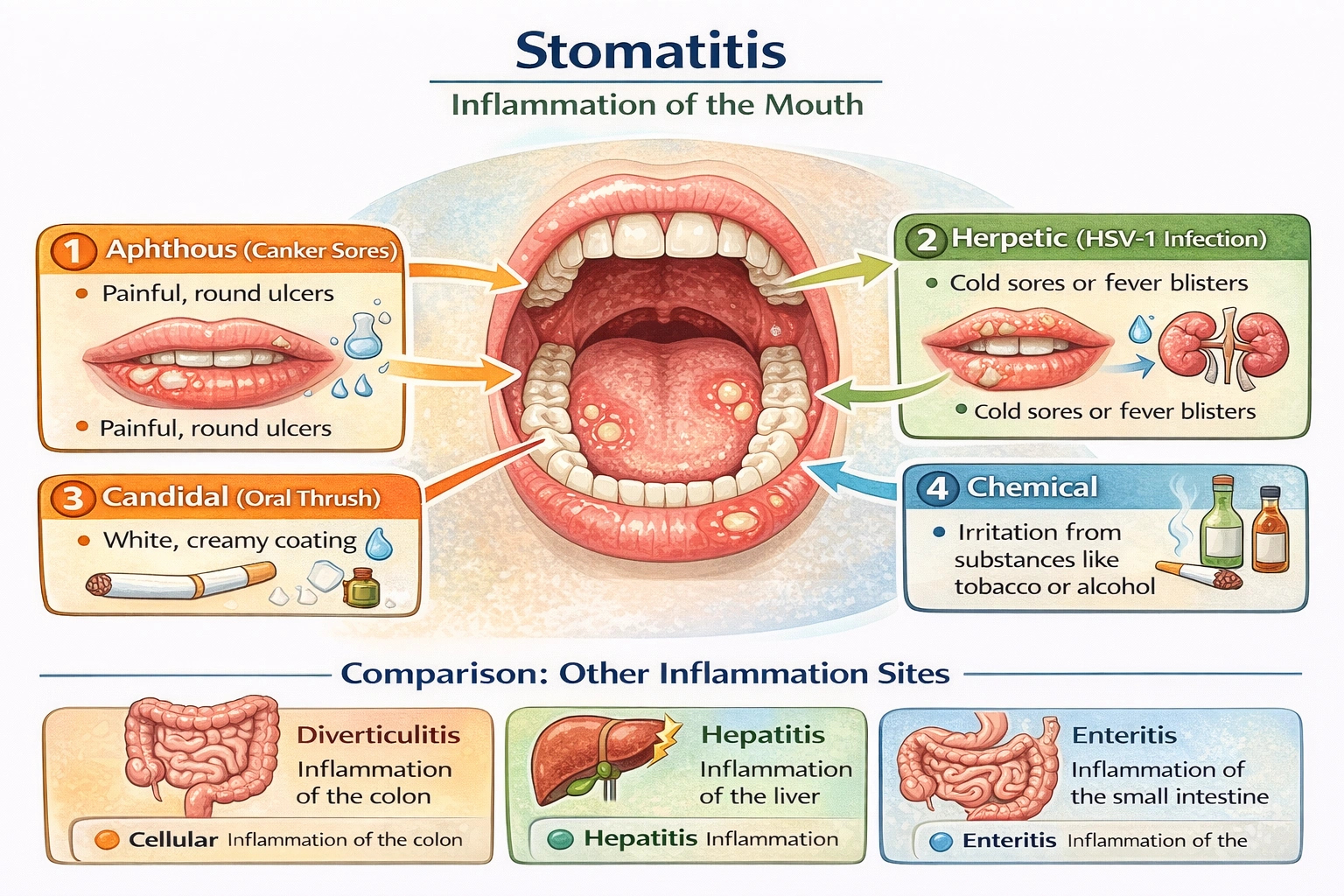
___ is a state of inflammation of the mouth.
A.
diverticulitis
B. hepatitis
C. enteritis
D. stomatitis
Rationale
Stomatitis describes inflammation of the mouth. Stomatitis encompasses inflammatory conditions affecting oral mucosa, including gingivitis (gums), glossitis (tongue), and cheilitis (lips)—caused by infection, irritation, autoimmune disorders, or nutritional deficiencies, producing symptoms like redness, swelling, pain, and ulceration.
A) diverticulitis
Diverticulitis involves inflammation of diverticula (pouches) in the colon wall—typically sigmoid colon—causing left lower quadrant pain, fever, and potential perforation. This gastrointestinal condition affects the large intestine, not the oral cavity, representing a completely different anatomical location and pathophysiology.
B) hepatitis
Hepatitis denotes liver inflammation from viral infection (hepatitis A–E), toxins (alcohol), or autoimmune processes—manifesting as jaundice, elevated liver enzymes, and hepatomegaly. The liver resides in the upper right abdomen, anatomically and functionally distinct from the oral cavity.
C) enteritis
Enteritis refers to small intestine inflammation—commonly from infection (Salmonella, norovirus) or Crohn's disease—causing diarrhea, cramping, and malabsorption. The small intestine extends from pylorus to ileocecal valve, far removed from oral structures.
D) stomatitis
Stomatitis derives from Greek stoma (mouth) + -itis (inflammation). Types include:
• Aphthous stomatitis: Recurrent canker sores of unknown etiology
• Herpetic stomatitis: HSV-1 infection causing painful vesicles/ulcers
• Candidal stomatitis: Fungal overgrowth (thrush) in immunocompromised individuals
• Chemical stomatitis: Irritation from tobacco, alcohol, or caustic substances
Diagnosis involves visual examination and sometimes culture/biopsy; treatment targets underlying cause with topical analgesics, antivirals, or antifungals as appropriate.
Conclusion:
Stomatitis specifically denotes oral cavity inflammation—distinguished from inflammatory conditions affecting other gastrointestinal segments (diverticulitis, enteritis) or distant organs (hepatitis). Option D provides the precise medical term essential for accurate diagnosis and treatment planning. Misidentification could lead to inappropriate therapy—prescribing antivirals for herpetic stomatitis versus antibiotics for bacterial enteritis requires correct anatomical localization of inflammation.
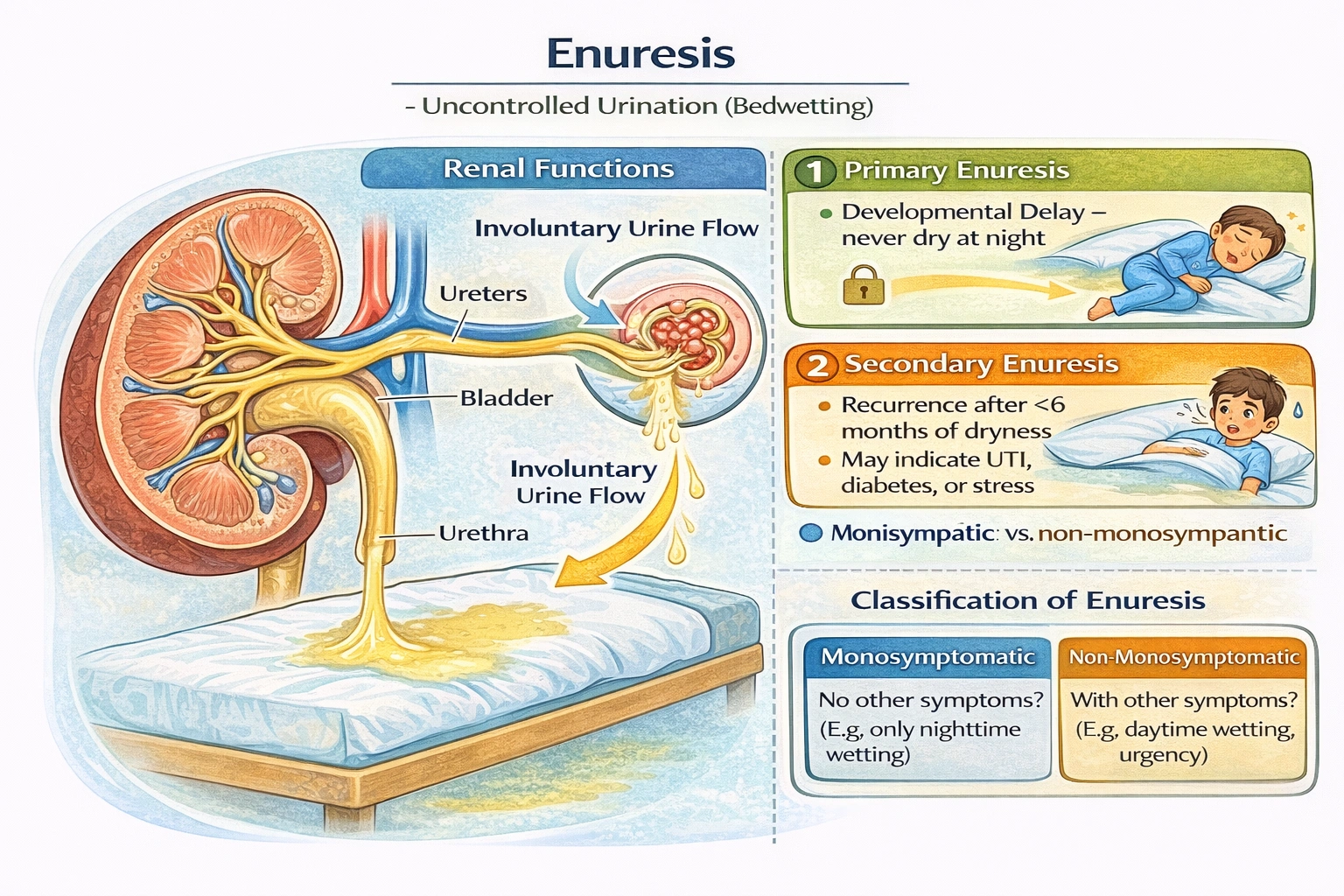
Which of the following terms matches the definition: uncontrolled urination.
A.
enuresis
B. diuretic
C. pyuria
D. ureteritis
Rationale
Enuresis describes uncontrolled urination. Enuresis refers to involuntary voiding of urine—typically nocturnal enuresis (bedwetting) in children beyond age 5 or secondary enuresis in adults—resulting from delayed bladder maturation, hormonal imbalances (insufficient ADH at night), small functional bladder capacity, or neurological conditions.
A) enuresis
Enuresis derives from Greek enourein (to urinate). Classification includes:
• Primary enuresis: Never achieved consistent nighttime dryness (developmental delay)
• Secondary enuresis: Recurrence after ≥6 months of dryness (may indicate UTI, diabetes, psychological stress)
• Monosymptomatic: Only nighttime wetting without daytime symptoms
• Non-monosymptomatic: Accompanied by daytime urgency, frequency, or incontinence
Treatment approaches include behavioral modification (bedwetting alarms), desmopressin (synthetic ADH), and addressing underlying causes—distinguishing it from normal infantile incontinence or stress incontinence in adults.
B) diuretic
Diuretics are substances (medications like furosemide, or natural compounds like caffeine) that increase urine production by inhibiting renal reabsorption—but they don't cause uncontrolled urination. Diuretic users maintain voluntary bladder control despite increased urine volume; the term describes a pharmacological agent or effect, not a loss of continence.
C) pyuria
Pyuria denotes pus (white blood cells) in urine—indicating urinary tract infection, inflammation, or sterile inflammation from stones/tumors. While UTIs may cause urgency contributing to incontinence, pyuria itself describes a laboratory finding (cloudy urine, positive leukocyte esterase) rather than loss of bladder control.
D) ureteritis
Ureteritis refers to inflammation of the ureters—rarely diagnosed independently as ureters typically become inflamed secondarily to kidney infection (pyelonephritis) or bladder infection extending upward. Ureteritis causes flank pain and may obstruct urine flow, but doesn't directly produce uncontrolled urination—bladder dysfunction causes incontinence, not ureter pathology.
Conclusion:
Enuresis specifically denotes involuntary urine loss—distinct from increased urine production (diuretics), urinary infection signs (pyuria), or upper urinary tract inflammation (ureteritis). Option A provides the precise clinical term essential for accurate diagnosis and management: primary enuresis often resolves with maturation, while secondary enuresis requires investigation for underlying pathology. Mislabeling could delay appropriate intervention for conditions like diabetes insipidus or neurological disorders presenting with incontinence.
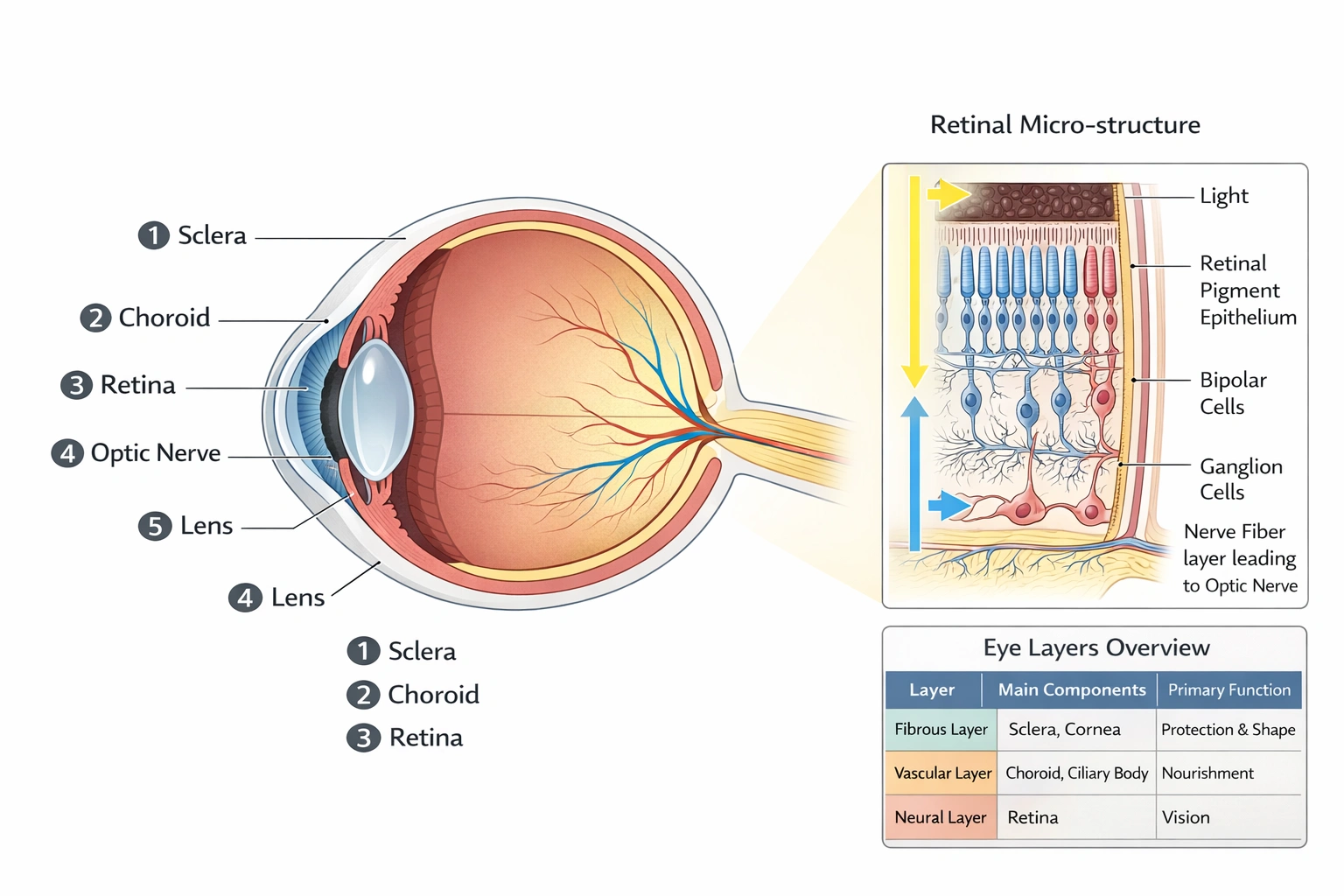
The inner layer of the eye is the ___
A.
cornea
B. sclera
C. retina
D. rods
Rationale
The retina forms the inner layer of the eye. The retina comprises the photosensitive neural tissue lining the posterior two-thirds of the eyeball—containing photoreceptors that convert light into electrical signals processed through multiple neuronal layers before transmission via the optic nerve to visual centers in the brain.
A) cornea
The cornea constitutes the transparent anterior portion of the outer fibrous tunic—not an inner layer. Positioned at the eye's front surface, it serves as a protective window and refractive element but lies external to all other ocular structures including aqueous humor, lens, and vitreous body—making it anatomically the most superficial layer rather than innermost.
B) sclera
The sclera forms the opaque posterior portion of the outer fibrous tunic—providing structural support and muscle attachment points. As the eye's external coat, it lies superficial to the choroid (middle vascular layer) and retina (inner neural layer)—positioned farthest from the optical axis rather than innermost.
C) retina
The retina's layered structure includes:
• Pigmented epithelium: Outermost retinal layer adjacent to choroid; absorbs stray light and supports photoreceptor metabolism
• Photoreceptor layer: Rods (scotopic vision) and cones (photopic/color vision) with outer/inner segments
• Outer nuclear layer: Photoreceptor cell bodies
• Outer plexiform layer: Synapses between photoreceptors and bipolar/horizontal cells
• Inner nuclear layer: Bipolar, horizontal, and amacrine cell bodies
• Inner plexiform layer: Synapses between bipolar and ganglion/amacrine cells
• Ganglion cell layer: Output neurons whose axons form optic nerve
• Nerve fiber layer: Ganglion cell axons converging toward optic disc
This neural architecture transforms light patterns into encoded neural signals—making the retina functionally an extension of the central nervous system (diencephalic origin).
D) rods
Rods represent one photoreceptor cell type within the retina—not the entire inner layer. While essential for dim-light vision, rods constitute only a component of retinal structure alongside cones, bipolar cells, ganglion cells, and supporting elements. Identifying rods as the inner layer confuses cellular constituents with tissue-level organization.
Conclusion:
The retina serves as the eye's innermost neural layer responsible for phototransduction and initial visual processing—distinct from outer protective layers (sclera/cornea) or cellular components within the retina itself. Option C correctly identifies this critical sensory structure, essential for understanding visual pathology: retinal detachment separates this inner layer from its blood supply causing vision loss, while macular degeneration affects central retinal photoreceptors—conditions requiring precise anatomical knowledge for diagnosis and treatment planning.
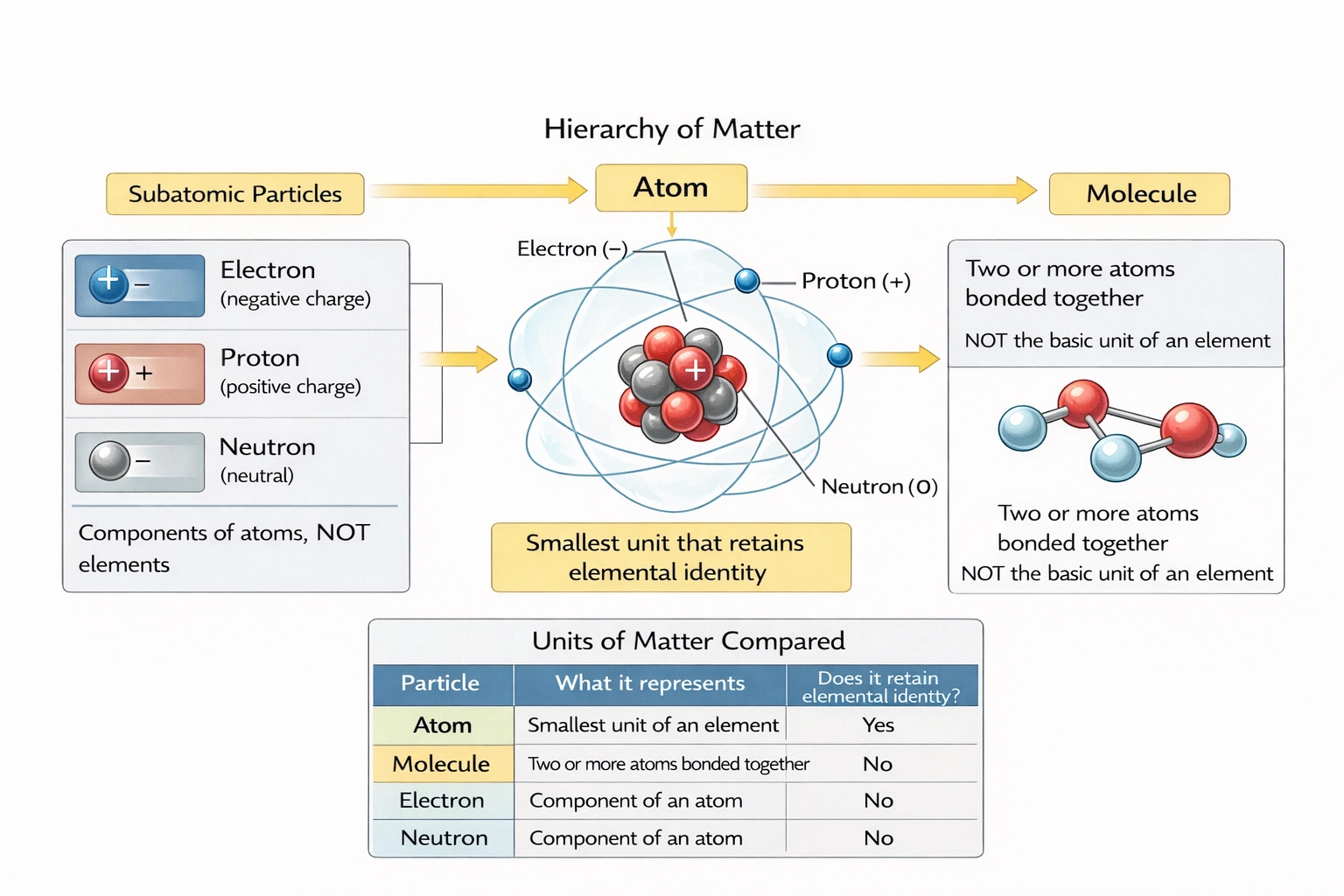
A/an ___ is the simplest unit of an element.
A.
atom
B. molecule
C. electron
D. neutron
Rationale
An atom is the simplest unit of an element. Atoms represent the smallest particles retaining an element's chemical identity—comprising a nucleus (protons and neutrons) surrounded by electrons—with the number of protons (atomic number) defining the element's identity and chemical behavior.
A) atom
Atoms cannot be subdivided by chemical means while retaining elemental properties—nuclear reactions can split atoms but transform them into different elements. Each element has unique atoms characterized by:
• Atomic number (Z): Proton count defining elemental identity (e.g., all carbon atoms have Z=6)
• Mass number (A): Sum of protons and neutrons (isotopes vary in neutron count)
• Electron configuration: Determines chemical bonding behavior and reactivity
Chemical reactions rearrange atoms but don't alter their elemental identity—demonstrating atoms as the fundamental units participating in chemical change while preserving elemental characteristics.
B) molecule
Molecules consist of two or more atoms chemically bonded together—representing the simplest unit of a compound (e.g., H₂O) or elemental substance existing as diatomic molecules (O₂, N₂). Molecules can be subdivided into constituent atoms through chemical reactions while retaining elemental identities—making them composite structures rather than simplest elemental units.
C) electron
Electrons are subatomic particles carrying negative charge—constituents of atoms but not representative of elemental identity. All elements contain electrons, but electron count alone doesn't define an element (ions of different elements can have identical electron counts). Removing or adding electrons creates ions but doesn't change elemental identity—proton count remains definitive.
D) neutron
Neutrons are neutral subatomic particles within atomic nuclei—varying in number to create isotopes of the same element. Neutrons alone don't define elemental identity; different elements can have identical neutron counts (e.g., â¶Li and â·Be both have 3 neutrons). Free neutrons decay with 10.3-minute half-life and don't constitute stable matter—making them unsuitable as elemental building blocks.
Conclusion:
Atoms uniquely serve as the smallest particles preserving an element's chemical identity—defined by proton count and participating as indivisible units in chemical reactions. Option A correctly identifies atoms as elemental building blocks, distinguishing them from composite molecules (B) or subatomic constituents (C, D) that lack elemental specificity. This concept underpins all chemistry: the law of definite proportions arises because compounds form from whole-atom combinations, and the periodic table organizes elements by atomic structure—making atomic theory foundational to chemical science.
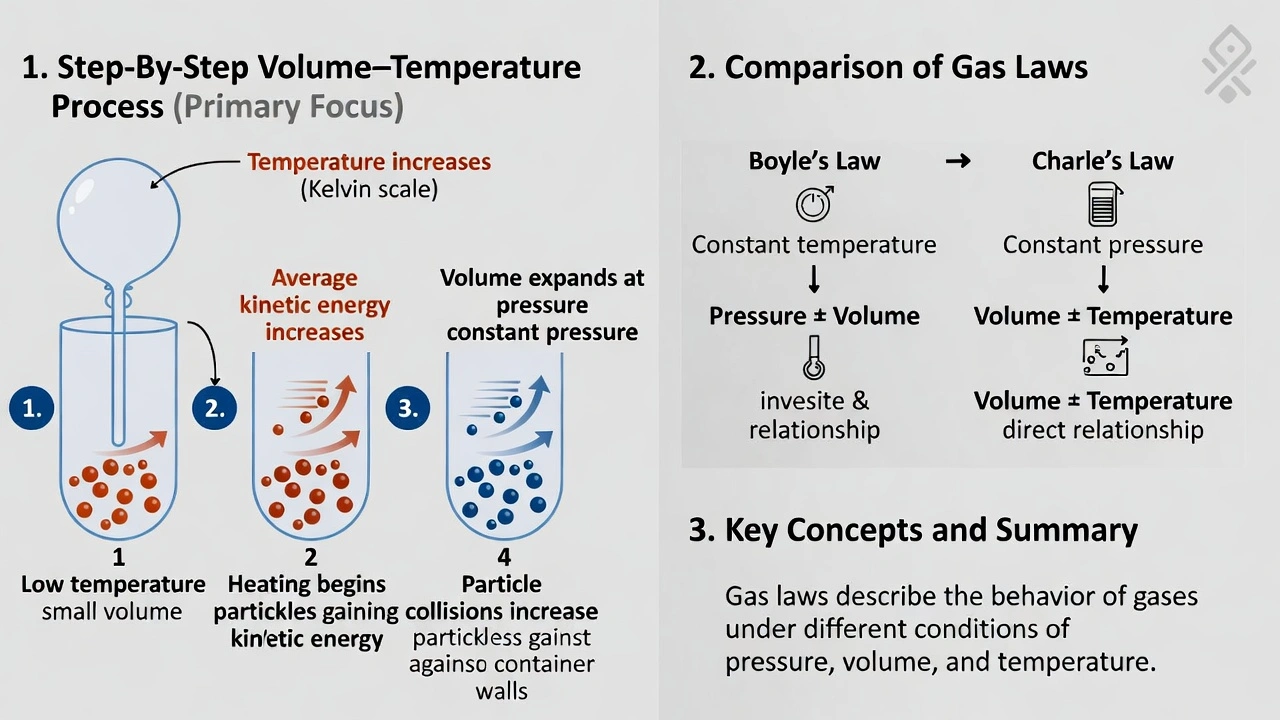
Which of the following terms matches the definition: the volume of a gas varies indirectly with temperature with pressure constant.
A.
Boyle's law
B. Charles' law
C. Johnson's law
D. Avogadro's law
Rationale
Charles' law describes gas volume-temperature relationships at constant pressure.
Note: The question contains an error—"indirectly" should read "directly." Charles' law states volume varies directly (not indirectly) with absolute temperature at constant pressure (V ∠T). No gas law describes inverse volume-temperature relationship at constant pressure as this would violate thermodynamic principles.
Charles' law establishes that gas volume increases proportionally with absolute temperature when pressure remains constant—demonstrated by Vâ‚/Tâ‚ = Vâ‚‚/Tâ‚‚ where temperature must be expressed in Kelvin to maintain proportionality through absolute zero.
A) Boyle's law
Boyle's law describes inverse pressure-volume relationship at constant temperature (P ∠1/V or PV = constant)—not volume-temperature relationships. Compressing a gas syringe decreases volume while increasing pressure at fixed temperature—demonstrating Boyle's principle. This law addresses different variables (P and V) under different constraints (constant T) than the question's focus.
B) Charles' law
Charles' law (Jacques Charles, 1787) states V ∠T at constant pressure and moles:
• Heating gas in flexible container (balloon) increases volume proportionally with Kelvin temperature
• Cooling reduces volume linearly—extrapolating to zero volume at absolute zero (-273.15°C)
• Mathematical form: Vâ‚/Tâ‚ = Vâ‚‚/Tâ‚‚ (T in Kelvin)
Critical correction: The question incorrectly states "varies indirectly" (inverse relationship). Charles' law describes direct proportionality—volume increases as temperature increases. An inverse volume-temperature relationship at constant pressure would violate the kinetic molecular theory and isn't observed in nature. Despite the wording error, Charles' law is the only option addressing volume-temperature relationships.
C) Johnson's law
No recognized gas law bears this name in physics or chemistry—likely a distractor option. Standard gas laws include Boyle's, Charles', Gay-Lussac's, Avogadro's, and the combined ideal gas law—none attributed to "Johnson" in scientific literature.
D) Avogadro's law
Avogadro's law states volume varies directly with moles of gas at constant temperature and pressure (V ∠n)—not with temperature. Equal volumes of gases at identical T and P contain equal molecule counts—fundamental to stoichiometry and molar volume concepts (22.4 L/mol at STP). This law addresses quantity (n) rather than thermal effects (T).
Conclusion:
Charles' law uniquely describes volume-temperature relationships for gases at constant pressure—though the question erroneously states "indirectly" when the relationship is direct proportionality. Option B correctly identifies the relevant law despite the wording flaw, essential for understanding thermal expansion of gases in engineering applications (hot air balloons, thermometer design) and correcting the misconception that volume might decrease with rising temperature under constant pressure—a physically impossible scenario violating energy conservation principles.
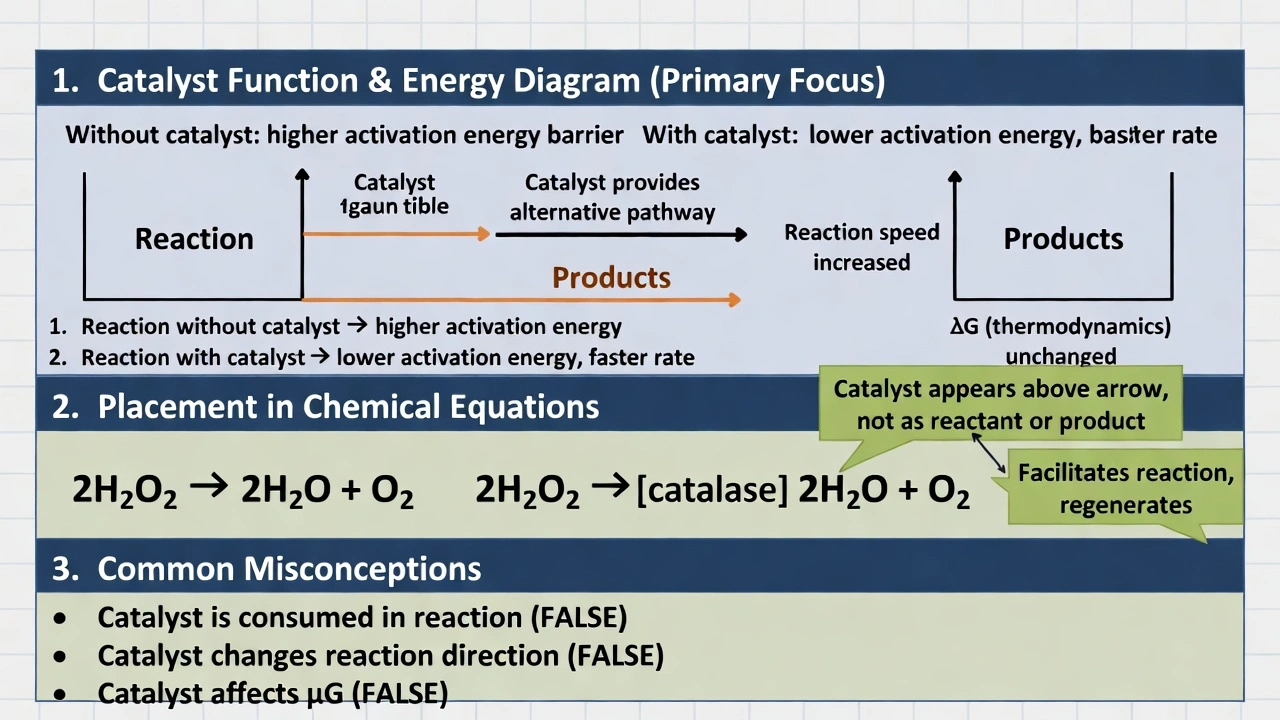
Which of the following is not true of a reaction catalyst's potential?
A.
it can speed up a reaction
B. it can cause a reaction
C. it is never destroyed
D. it is always found on the right side of an equation
Rationale
Catalysts are not always found on the right side of chemical equations. Catalysts appear above the reaction arrow or noted separately in equations—not as reactants (left side) or products (right side)—because they participate in reaction mechanisms without being consumed or appearing in the net reaction stoichiometry.
A) it can speed up a reaction
Catalysts accelerate reactions by providing alternative pathways with lower activation energy—enabling more molecular collisions to succeed at given temperatures. Enzymes increase biochemical reaction rates by factors of 10â¶â€“10¹²; catalytic converters accelerate automotive exhaust conversion. This rate enhancement without thermodynamic alteration (ΔG unchanged) represents catalysts' primary function—making this statement true.
B) it can cause a reaction
Catalysts cannot cause thermodynamically impossible reactions (ΔG > 0)—they only accelerate spontaneous reactions (ΔG < 0) that would occur eventually without catalyst. Catalysts affect kinetics (reaction speed), not thermodynamics (reaction feasibility). A catalyst added to non-spontaneous reactants produces no reaction—demonstrating catalysts don't "cause" reactions but accelerate inherently favorable ones. This statement is misleading but technically false in absolute terms.
C) it is never destroyed
Catalysts regenerate in each reaction cycle—participating in intermediate steps but emerging unchanged in the net reaction. Enzymes release after substrate conversion; platinum in catalytic converters remains after exhaust treatment. While catalysts may degrade over time (enzyme denaturation, catalyst poisoning), they aren't stoichiometrically consumed in the reaction they catalyze—making this generally true for ideal catalysts.
D) it is always found on the right side of an equation
Catalysts never appear on either side of balanced chemical equations—they're written above the arrow:
2H₂O₂ →[catalase] 2H₂O + O₂
or noted separately: "Reaction catalyzed by Pt."
Including catalysts as products (right side) would imply consumption and regeneration within the equation—violating mass balance since catalysts don't undergo net change. Catalysts facilitate reactions without appearing in stoichiometric accounting—making this statement definitively false.
Conclusion:
Catalysts function without appearing in reaction stoichiometry—positioned above arrows rather than as reactants or products. Option D correctly identifies the false statement, essential for proper chemical equation representation: misplacing catalysts as products would misrepresent reaction mechanisms and violate conservation of mass principles. Understanding catalyst notation prevents errors in reaction balancing and mechanism elucidation—critical for chemical engineering design and biochemical pathway analysis where catalyst identity and placement clarify rate-determining steps.
What are the two types of measurement important in science?
A.
quantitative and numerical
B. qualitative and descriptive
C. numerical and scientific
D. quantitative and qualitative
Rationale
Quantitative and qualitative are the two fundamental measurement types in science. Quantitative measurements provide numerical data that can be measured and statistically analyzed while qualitative measurements describe qualities and characteristics without numerical values forming the complementary foundation of scientific observation and analysis.
A) quantitative and numerical
This option is incorrect because numerical measurements are essentially the same as quantitative measurements. Both involve numerical data with quantitative being the precise scientific term for numerical measurements. Quantitative measurements include specific values like mass volume or temperature that can be measured and expressed with units.
B) qualitative and descriptive
This option is incorrect because descriptive measurements are essentially the same as qualitative measurements. Both describe qualities characteristics or observations without numerical values. The scientific term for this type of measurement is qualitative not descriptive.
C) numerical and scientific
This option is incorrect because scientific is not a recognized category of measurement in science. Numerical measurements are a subset of quantitative measurements but scientific describes the field not a type of measurement. Quantitative measurements are the standard scientific term for numerical data.
D) quantitative and qualitative
These represent the two distinct and complementary measurement types in science:
• Quantitative measurements provide numerical data that can be measured and analyzed statistically (e.g., 25 grams 37°C 10 meters)
• Qualitative measurements describe non-numerical characteristics and observations (e.g., color texture odor)
All scientific investigations require both types for comprehensive understanding with quantitative data providing precision and qualitative data providing context and descriptive richness.
Conclusion:
Quantitative and qualitative measurements form the essential duality of scientific observation with quantitative measurements providing numerical precision and qualitative measurements capturing descriptive characteristics. Both types are necessary for complete scientific analysis and understanding. Option D correctly identifies these two fundamental measurement categories essential for all scientific disciplines.

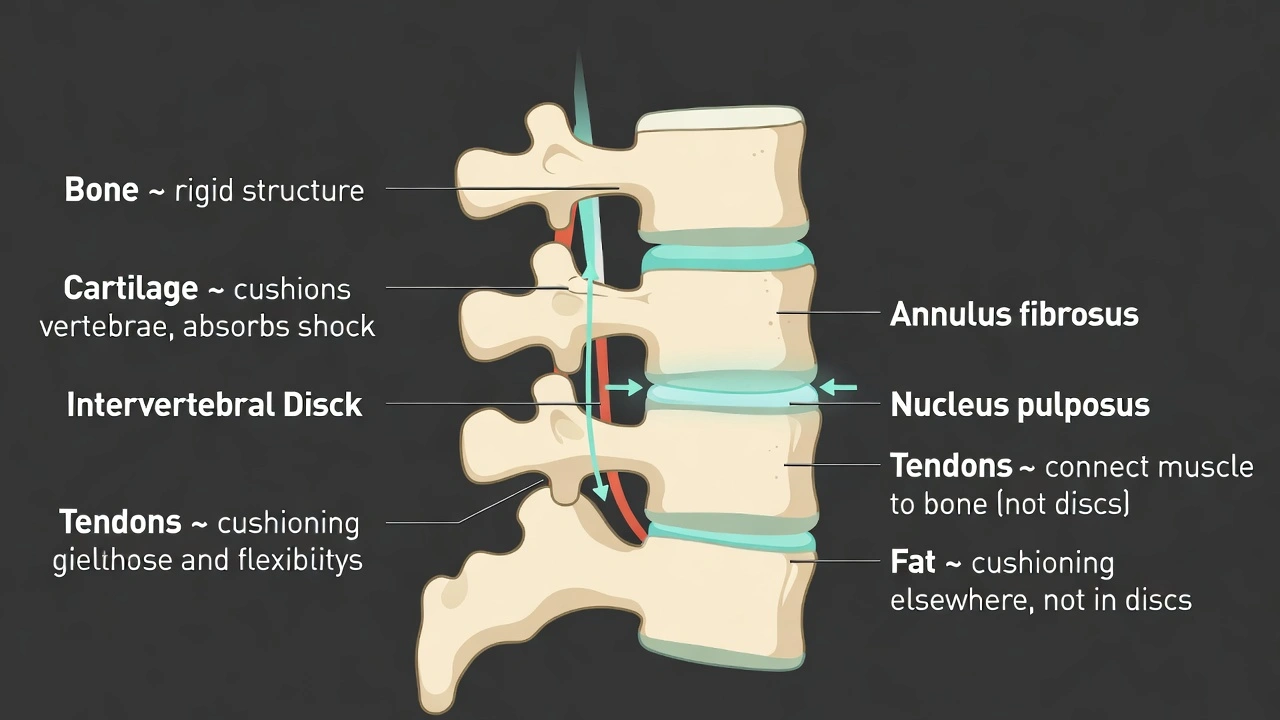
Which substance makes up the pads that provide support between the vertebrae?
A.
bone
B. cartilage
C. tendon
D. fat
Rationale
Cartilage forms the intervertebral discs between vertebrae. Intervertebral discs consist of fibrocartilage with an outer annulus fibrosus of concentric collagen layers surrounding an inner nucleus pulposus of gelatinous material providing shock absorption flexibility and weight distribution along the spinal column while preventing direct bone-on-bone contact between adjacent vertebrae.
A) bone
Bone forms the vertebral bodies themselves but not the intervening pads. Direct bone contact between vertebrae would create rigid immobile segments incapable of absorbing impact or permitting spinal flexion extension and rotation. Bone lacks the compressibility and resilience required for shock absorption making it unsuitable for intervertebral cushioning.
B) cartilage
Intervertebral discs are composed of specialized fibrocartilage containing both type I and type II collagen fibers. The annulus fibrosus provides tensile strength to contain the nucleus pulposus while the nucleus pulposus acts as a hydraulic shock absorber distributing compressive forces radially. The high water content (approximately 80% in youth) enables elastic deformation under load followed by recovery when pressure releases. This cartilaginous structure permits spinal movement while protecting vertebrae from fracture during impact.
C) tendon
Tendons connect muscle to bone transmitting contractile forces but do not form intervertebral structures. No tendons exist between vertebrae as tendons require muscular attachment points and function in force transmission rather than cushioning. Tendons lack the layered structure and water content necessary for shock absorption making them mechanically unsuited for intervertebral support.
D) fat
Fat tissue provides insulation energy storage and some cushioning in subcutaneous layers but does not form intervertebral discs. Adipose tissue lacks structural integrity under compressive loads and would deform permanently without elastic recovery. Fat cannot withstand the repetitive loading cycles experienced by the spine nor provide the controlled resistance necessary for maintaining disc height and spinal alignment.
Conclusion:
Fibrocartilage uniquely combines tensile strength compressibility and elastic recovery making it the ideal biological material for intervertebral discs. Option B correctly identifies cartilage as the substance forming these essential spinal cushions distinguishing it from rigid bone load-transmitting tendons or non-structural fat tissue. Understanding disc composition proves clinically significant as disc degeneration involves cartilage breakdown leading to herniation loss of disc height and nerve compression requiring different treatments than bone or soft tissue injuries.
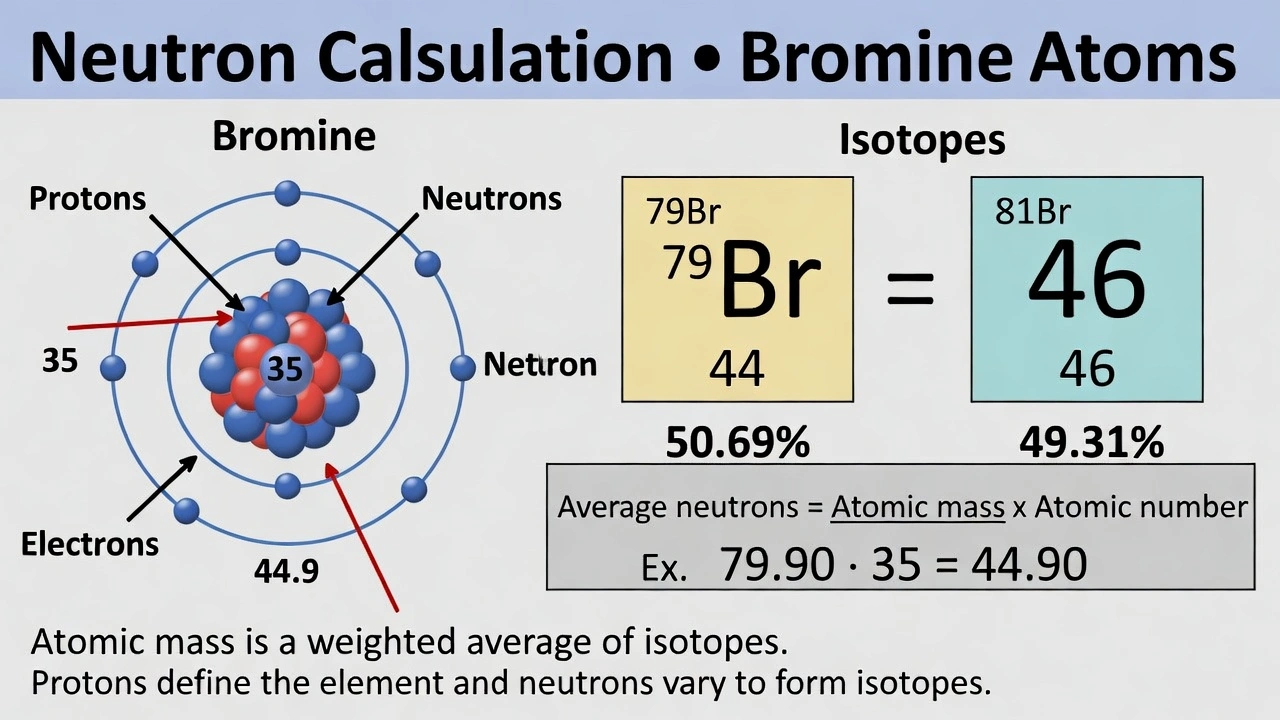
On average, how many neutrons does one atom of bromine (Br) have?
A.
35
B. 44.9
C. 45
D. 79.9
Rationale
The average number of neutrons in a bromine atom is 44.90. To determine the average number of neutrons in an atom, subtract the atomic number (proton count) from the atomic mass. The atomic mass represents the weighted average of all isotopes while the atomic number indicates the fixed proton count for that element.
A) 35
This option represents bromine's atomic number which equals the number of protons not neutrons. Bromine has 35 protons in all its isotopes but varies in neutron count across isotopes. â·â¹Br has 44 neutrons while â¸Â¹Br has 46 neutrons. The average neutron count must account for both isotopes' natural abundance.
B) 44.90
Bromine has an atomic number of 35 and an atomic mass of 79.90. The average number of neutrons is calculated as atomic mass minus atomic number: 79.90 - 35 = 44.90. This value reflects the weighted average based on natural isotope abundance: â·â¹Br (50.69%) and â¸Â¹Br (49.31%). The fractional neutron count arises because it represents an average across the natural mixture of isotopes not a count in any single atom.
C) 45
This option represents a rounded value of the average neutron count but the precise calculation yields 44.90. The question asks for the average number of neutrons which is a precise value derived from atomic mass and atomic number not a rounded integer. While 45 is close the accurate value is 44.90 as shown in standard reference tables.
D) 79.90
This option represents bromine's atomic mass not the neutron count. The atomic mass includes the combined mass of protons and neutrons with electrons contributing negligibly. The atomic mass value itself cannot represent the neutron count as it encompasses both protons and neutrons.
Conclusion:
The average number of neutrons in a bromine atom equals its atomic mass minus its atomic number: 79.90 - 35 = 44.90. This value represents the weighted average across bromine's natural isotope distribution. Option B correctly identifies this precise average neutron count essential for understanding atomic structure and isotope calculations.
Which of the following cannot exist in RNA?
A.
Uracil
B. Thymine
C. Cytosine
D. Guanine
Rationale
Thymine cannot exist in RNA. RNA nucleotides contain adenine guanine cytosine and uracil while DNA contains adenine guanine cytosine and thymine with uracil replacing thymine as the complementary base to adenine in RNA molecules.
A) Uracil
Uracil is a pyrimidine base unique to RNA that pairs with adenine during transcription and translation. Uracil differs from thymine by lacking a methyl group at the 5' carbon position. This structural difference allows cellular machinery to distinguish RNA from DNA and facilitates RNA's typically shorter lifespan compared to DNA.
B) Thymine
Thymine is a methylated pyrimidine base found exclusively in DNA where it pairs with adenine. The methyl group at thymine's 5' carbon position provides stability to the DNA double helix and helps protect genetic information from degradation. RNA polymerases cannot incorporate thymine nucleotides during transcription using uracil instead. No naturally occurring RNA molecules contain thymine though rare modified bases may appear in specialized RNA types.
C) Cytosine
Cytosine is a pyrimidine base present in both DNA and RNA where it pairs with guanine through three hydrogen bonds. This base conservation across nucleic acids reflects cytosine's fundamental role in genetic coding and its chemical stability in both double-stranded DNA and various RNA conformations.
D) Guanine
Guanine is a purine base found in both DNA and RNA pairing with cytosine through three hydrogen bonds. Its presence in all nucleic acids underscores its essential role in maintaining consistent base-pairing rules across genetic information storage (DNA) and transfer (RNA) systems.
Conclusion:
The fundamental chemical distinction between DNA and RNA lies in their pyrimidine bases with thymine exclusive to DNA and uracil exclusive to RNA. Option B correctly identifies thymine as the base that cannot exist in RNA. This distinction enables cellular enzymes to recognize and process each nucleic acid type appropriately and explains why reverse transcriptase must convert uracil to thymine when synthesizing DNA from RNA templates.
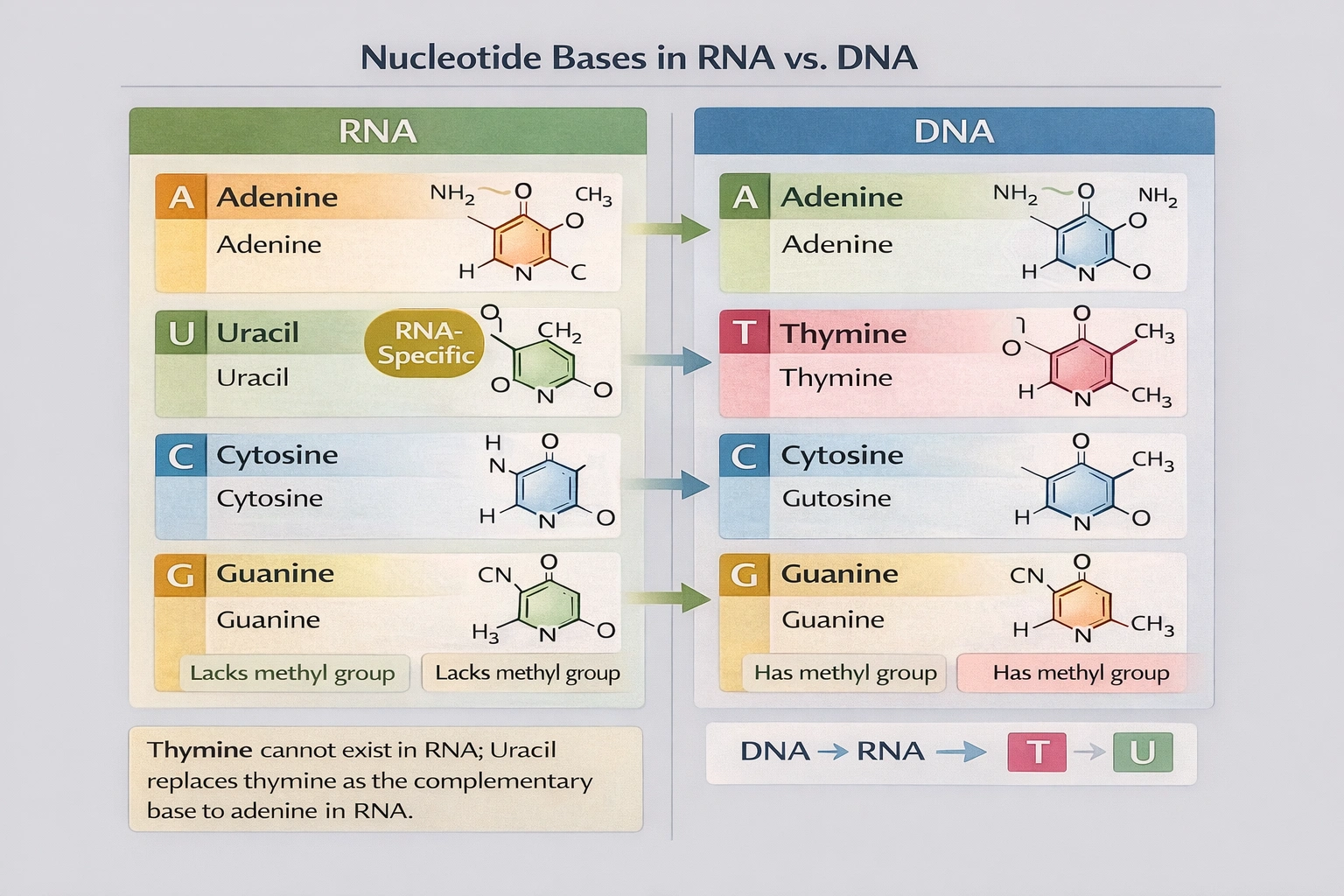
Which of the following describes the transport network that is responsible for the transference of proteins throughout a cell?
A.
Golgi apparatus
B. Endoplasmic reticulum
C. Mitochondria
D. Nucleolus
Rationale
The endoplasmic reticulum serves as the primary intracellular transport network for proteins. The endoplasmic reticulum forms an extensive interconnected membrane system extending from the nuclear envelope throughout the cytoplasm with rough ER synthesizing and transporting proteins and smooth ER handling lipid synthesis and detoxification creating a continuous pathway for molecular trafficking.
A) Golgi apparatus
The Golgi apparatus functions as a processing and sorting center that modifies packages and distributes proteins received from the ER. It consists of stacked flattened cisternae that add carbohydrate groups sort proteins into vesicles and direct them to final destinations. However it does not constitute the primary transport network itself but rather a processing station along the transport pathway.
B) Endoplasmic reticulum
The endoplasmic reticulum forms a continuous membrane network with two specialized regions:
• Rough ER (studded with ribosomes) synthesizes transmembrane and secretory proteins that enter the ER lumen for folding and initial modification
• Smooth ER (lacks ribosomes) handles lipid synthesis calcium storage and detoxification
Proteins move through the ER network via vesicular transport or direct membrane continuity then exit in transport vesicles destined for the Golgi apparatus. This extensive membranous system functions as the cell's primary intracellular highway for protein transference.
C) Mitochondria
Mitochondria serve as cellular power plants generating ATP through oxidative phosphorylation. They contain their own DNA and replicate independently but do not participate in protein transport networks. Mitochondria import specific proteins synthesized in the cytoplasm but lack the extensive membranous structure required for general protein transference throughout the cell.
D) Nucleolus
The nucleolus is a dense region within the nucleus responsible for ribosomal RNA synthesis and ribosome subunit assembly. It does not participate in protein transport after synthesis. Ribosomes assembled in the nucleolus exit the nucleus to function in protein synthesis but the nucleolus itself lacks transport capabilities for proteins moving through the cytoplasm.
Conclusion:
The endoplasmic reticulum's continuous membranous structure forms the foundational transport network for newly synthesized proteins moving from ribosomes toward their cellular destinations. Option B correctly identifies this essential organelle system distinguishing it from processing centers (Golgi apparatus) energy producers (mitochondria) or ribosome assembly sites (nucleolus). Understanding ER function proves critical for comprehending protein trafficking diseases like cystic fibrosis where misfolded proteins fail to exit the ER properly.
A substance is considered acidic if it has a pH of less than which of the following?
A.
12
B. 9
C. 7
D. 4
Rationale
Acidic substances have a pH less than 7. The pH scale ranges from 0 to 14 with 7 representing neutrality (pure water at 25°C). Values below 7 indicate acidity (increasing hydrogen ion concentration) while values above 7 indicate alkalinity (increasing hydroxide ion concentration).
A) 12
pH 12 represents a strongly basic solution with low hydrogen ion concentration (approximately 10â»Â¹Â² M). Substances with pH 12 are highly alkaline not acidic. Examples include household ammonia and lime water. Acidic substances must have pH values below 7 not 12.
B) 9
pH 9 represents a weakly basic solution with hydrogen ion concentration of approximately 10â»â¹ M. Substances like baking soda solution or seawater have pH around 9. These are alkaline not acidic though less strongly basic than pH 12 solutions.
C) 7
pH 7 represents neutrality where hydrogen ion concentration equals hydroxide ion concentration (10â»â· M each at 25°C). Pure water distilled water and some biological fluids maintain pH near 7. The dividing line between acidic and basic solutions is precisely pH 7 making this the correct threshold.
D) 4
pH 4 represents an acidic solution with hydrogen ion concentration of approximately 10â»â´ M. Examples include tomato juice and beer. While substances with pH 4 are indeed acidic the threshold for acidity is pH less than 7 not less than 4. Substances with pH 5 or 6 are also acidic though weaker than pH 4 solutions.
Conclusion:
The pH scale's neutral point at 7 serves as the definitive boundary between acidic and basic solutions with all values below 7 classified as acidic regardless of strength. Option C correctly identifies this fundamental threshold essential for understanding acid-base chemistry chemical reactions biological processes and environmental monitoring where pH classification determines appropriate handling treatment and interpretation of chemical behavior.
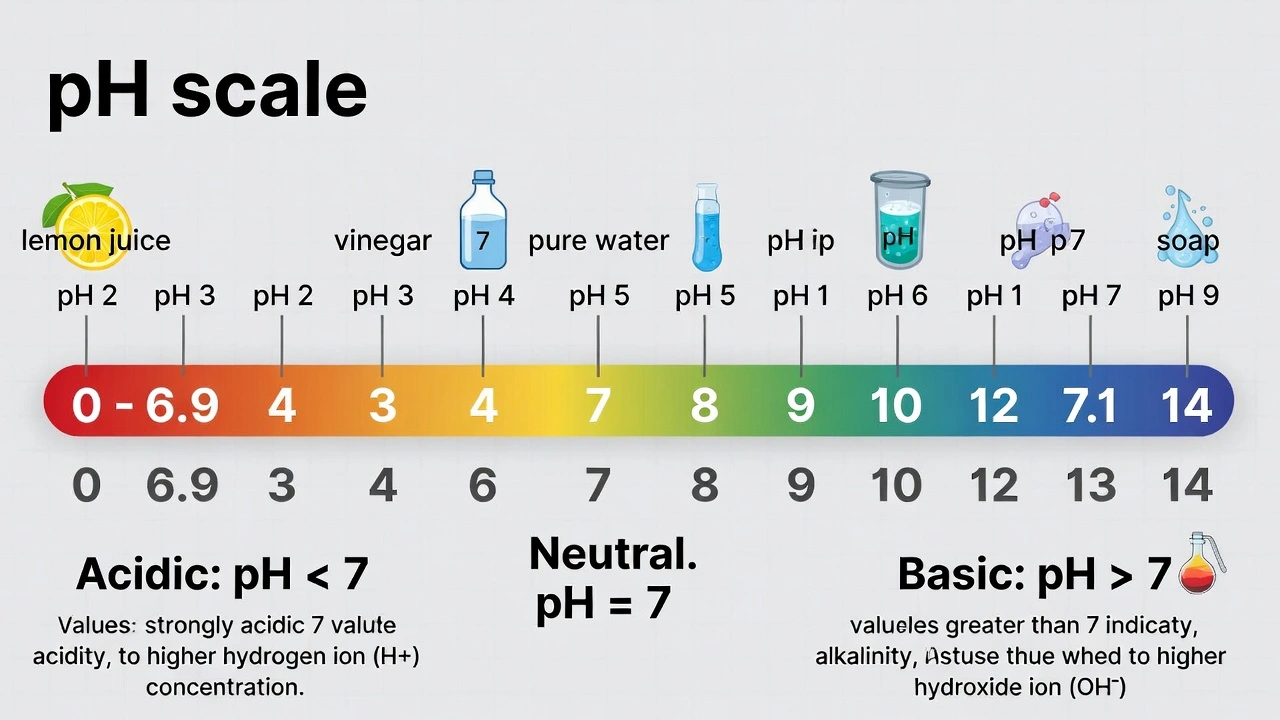
Which of the following best describes one of the roles of RNA?
A.
Manufacturing the proteins needed for DNA
B. Creating the bonds between the elements that compose DNA
C. Sending messages about the correct sequence of proteins in DNA
D. Forming the identifiable double helix shape of DNA
Rationale
RNA sends messages about protein sequences from DNA to ribosomes. Messenger RNA (mRNA) carries genetic information transcribed from DNA to ribosomes where it serves as the template for protein synthesis with its nucleotide sequence determining the amino acid sequence of the resulting polypeptide.
A) Manufacturing the proteins needed for DNA
Proteins are manufactured by ribosomes not by RNA. RNA molecules (particularly rRNA) form structural and catalytic components of ribosomes but do not themselves manufacture proteins. Ribosomes (composed of rRNA and proteins) catalyze peptide bond formation during translation. DNA does not require proteins to exist though proteins like histones help package DNA.
B) Creating the bonds between the elements that compose DNA
DNA bonds form through chemical interactions not RNA mediation. Phosphodiester bonds between nucleotides form during DNA replication catalyzed by DNA polymerase enzymes. Hydrogen bonds between complementary bases form spontaneously through molecular attraction. RNA does not create or mediate these bonds though RNA primers initiate DNA synthesis during replication.
C) Sending messages about the correct sequence of proteins
Messenger RNA performs this essential function. During transcription RNA polymerase synthesizes mRNA complementary to a DNA template strand. This mRNA carries the genetic code from the nucleus to cytoplasmic ribosomes where transfer RNA (tRNA) molecules match codons on mRNA to specific amino acids assembling polypeptides with sequences dictated by the original DNA. The phrase "sequence of proteins in DNA" contains imprecise wording since DNA contains sequences that code for proteins (not proteins themselves) but the intended meaning refers to RNA's messenger role.
D) Forming the identifiable "double helix" shape of DNA
DNA itself forms the double helix structure through complementary base pairing and sugar-phosphate backbone interactions. RNA typically exists as a single-stranded molecule that may fold into complex shapes but does not form DNA's double helix. RNA's structural role differs fundamentally from DNA's helical configuration.
Conclusion:
RNA's primary role in genetic information flow involves transmitting coded instructions from DNA to protein synthesis machinery. Option C correctly identifies this messenger function (despite slightly imprecise wording about "sequence of proteins in DNA"). The central dogma (DNA → RNA → protein) depends on RNA's ability to carry genetic messages enabling accurate translation of genetic information into functional proteins essential for all cellular processes.
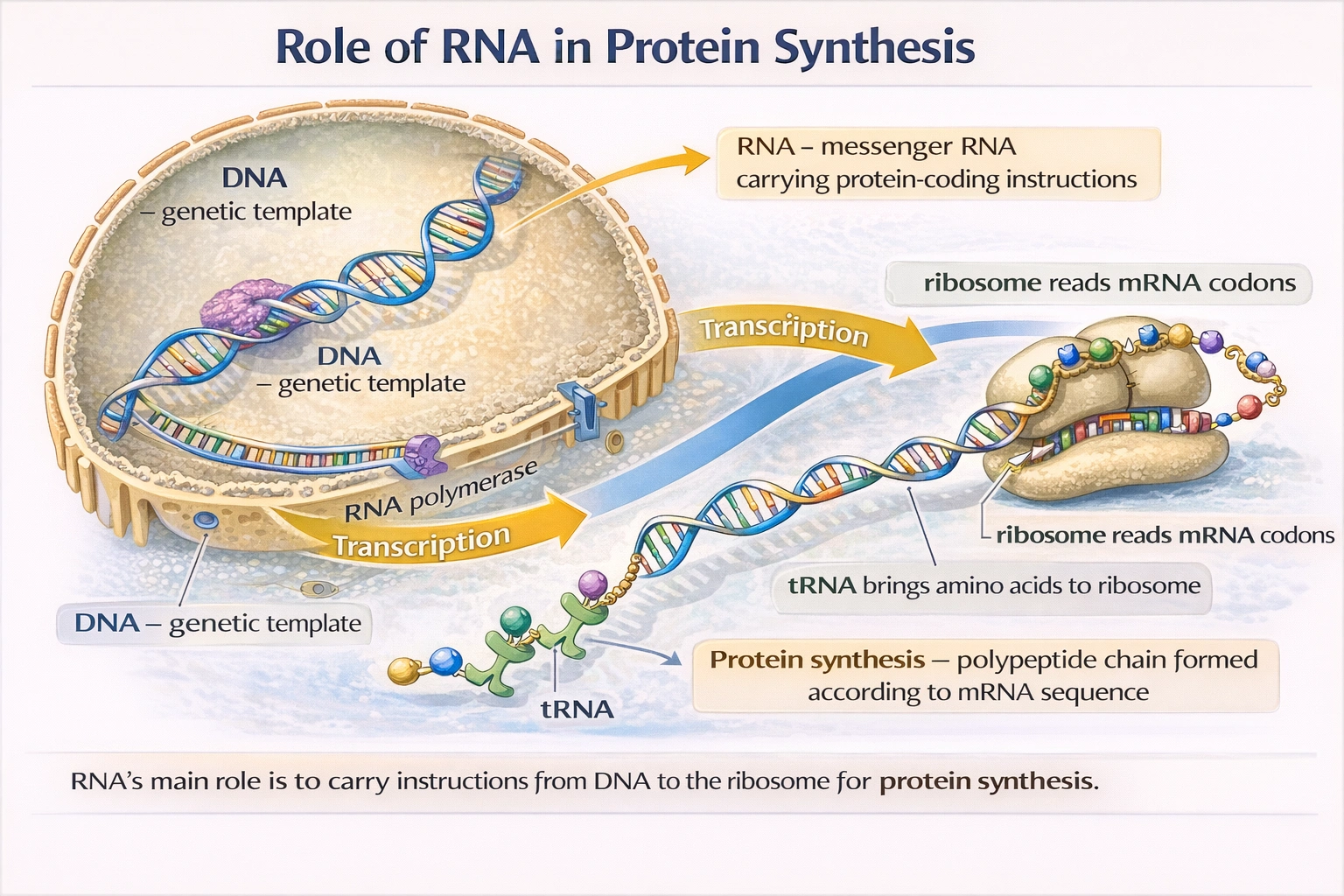
Which part of the cell is often called the cell "power house" because it provides energy for cellular functions?
A.
Nucleus
B. Cell membrane
C. Mitochondria
D. Cytoplasm
Rationale
Mitochondria are called the cell's powerhouse. Mitochondria generate adenosine triphosphate (ATP) through aerobic respiration specifically via the Krebs cycle occurring in the mitochondrial matrix and oxidative phosphorylation across the inner mitochondrial membrane with its folded cristae providing extensive surface area for electron transport chain complexes that create proton gradients driving ATP synthase enzymes.
A) Nucleus
The nucleus stores genetic material and controls cellular activities through gene expression regulation but does not produce energy molecules. ATP generation occurs outside the nucleus in the cytoplasm and mitochondria. The nucleus consumes ATP for processes like DNA replication and transcription rather than producing it.
B) Cell membrane
The cell membrane regulates substance transport maintains cellular integrity and facilitates communication but lacks the enzymatic machinery for ATP production. While the membrane uses ATP for active transport processes it does not synthesize energy currency molecules.
C) Mitochondria
Mitochondria contain their own circular DNA and ribosomes reflecting their evolutionary origin as endosymbiotic bacteria. They execute cellular respiration converting glucose-derived pyruvate and fatty acids into approximately 30-32 ATP molecules per glucose through coordinated processes in specialized compartments. The inner membrane's electron transport chain pumps protons into the intermembrane space creating electrochemical gradients that power ATP synthase rotation producing ATP from ADP and inorganic phosphate.
D) Cytoplasm
The cytoplasm hosts glycolysis which yields a net gain of 2 ATP molecules per glucose but this anaerobic process is far less efficient than mitochondrial aerobic respiration. The cytoplasm provides the aqueous medium for metabolic reactions but lacks the organized membrane systems required for high-yield ATP production.
Conclusion:
Mitochondria uniquely serve as the primary ATP production centers in eukaryotic cells through efficient aerobic respiration generating orders of magnitude more energy than cytoplasmic glycolysis alone. Option C correctly identifies mitochondria as the powerhouse organelle essential for meeting the high energy demands of complex cellular functions from muscle contraction to neural signalling.
How is meiosis similar to mitosis?
A.
Both produce daughter cells that are genetically identical
B. Both produce daughter cells that are genetically different
C. Both occur in humans, other animals, and plants
D. Both occur asexually
Rationale
Both meiosis and mitosis occur in humans, other animals, and plants. Meiosis and mitosis represent universal eukaryotic cell division processes found across multicellular kingdoms with mitosis enabling growth and repair in somatic cells while meiosis produces gametes for sexual reproduction in animals plants and many protists.
A) Both produce daughter cells that are genetically identical
Mitosis produces two genetically identical diploid daughter cells maintaining the parent cell's chromosome number and genetic composition. Meiosis produces four genetically unique haploid gametes through crossing over in prophase I and independent assortment in metaphase I followed by two sequential divisions reducing chromosome number by half. These processes differ fundamentally in genetic outcomes.
B) Both produce daughter cells that are genetically different
Meiosis generates genetic diversity through recombination and independent assortment but mitosis preserves genetic identity (except for rare replication errors). Somatic cells produced by mitosis remain genetically uniform within an organism enabling tissue coherence while gametes produced by meiosis vary genetically to promote offspring diversity.
C) Both occur in humans, other animals, and plants
Eukaryotic organisms universally employ both division types:
• Animals undergo mitosis for growth/repair and meiosis for gamete production
• Flowering plants experience mitosis in meristems and meiosis in anthers/ovaries producing spores that develop into gametophytes
• Fungi alternate between haploid and diploid stages using both processes
This universality reflects the ancient evolutionary origin of these mechanisms predating the divergence of major eukaryotic lineages.
D) Both occur asexually
Mitosis enables asexual reproduction in some organisms (binary fission budding) but also supports growth in sexually reproducing organisms. Meiosis exclusively facilitates sexual reproduction by producing haploid gametes that restore diploidy through fertilization. Meiosis cannot occur asexually as it requires subsequent syngamy to maintain chromosome number stability across generations.
Conclusion:
While meiosis and mitosis differ dramatically in purpose chromosome behavior and genetic outcomes they share the fundamental characteristic of occurring across diverse eukaryotic kingdoms. Option C correctly identifies this universal distribution distinguishing it from inaccurate claims about genetic identity outcomes or reproductive modes. Understanding this shared presence highlights the deep evolutionary conservation of cell division mechanisms despite their functional specialization.
Two cars driving in opposite directions collide. If you ignore friction and any other outside interactions, which of the following statements is always true?
A.
The total momentum is conserved
B. The sum of the potential and kinetic energy are conserved
C. The total velocity of the cars is conserved
D. The total impulse is conserved
Rationale
Total momentum is conserved in the collision. The law of conservation of momentum states that in an isolated system with no external forces the total vector momentum before collision equals the total vector momentum after collision regardless of collision type (elastic or inelastic).
A) The total momentum is conserved
Momentum conservation derives from Newton's third law during collisions. The force car A exerts on car B equals the force car B exerts on car A but in opposite directions acting for identical time intervals producing equal magnitude but opposite direction impulses that cancel when summed for the system. Mathematically mâ‚vâ‚áµ¢ + mâ‚‚vâ‚‚áµ¢ = mâ‚vâ‚f + mâ‚‚vâ‚‚f where subscripts i and f denote initial and final states. This principle holds for all isolated system collisions regardless of energy transformations.
B) The sum of the potential and kinetic energy are conserved
Total mechanical energy (kinetic plus potential) is conserved only in elastic collisions where no energy converts to heat sound or deformation. In real car collisions significant kinetic energy transforms into thermal energy sound and structural deformation making collisions inelastic. Mechanical energy conservation does not universally apply to collisions unlike momentum conservation.
C) The total velocity of the cars is conserved
Velocity is not conserved in collisions because it depends on mass. Individual car velocities change dramatically during impact and the vector sum of velocities has no conservation principle. Only momentum (mass times velocity) conserves not velocity alone. Two cars with equal mass and opposite velocities have zero total momentum before collision and zero after if they stick together but their individual velocities change completely.
D) The total impulse is conserved
Impulse (defined as force multiplied by time interval) represents the change in momentum for a single object not a conserved quantity for a system. Total impulse on the system equals zero because internal forces cancel in action-reaction pairs but impulse itself is not a conserved property like momentum or energy.
Conclusion:
Momentum conservation represents a fundamental law of physics applying universally to isolated systems during collisions regardless of collision type or energy transformations. Option A correctly identifies this invariant principle essential for analyzing collision dynamics traffic accident reconstruction and understanding why rockets propel forward by expelling mass backward.
Which of the answer choices provided best defines the following statement? For a given mass and constant temperature, an inverse relationship exists between the volume and pressure of a gas.
A.
Ideal Gas Law
B. Boyle's Law
C. Charles' Law
D. Stefan-Boltzmann Law
Rationale
Boyle's Law describes the inverse pressure-volume relationship at constant temperature. Boyle's Law states that for a fixed amount of gas at constant temperature the product of pressure and volume remains constant (Pâ‚Vâ‚ = Pâ‚‚Vâ‚‚) meaning pressure increases as volume decreases and vice versa due to more frequent molecular collisions with container walls in reduced space.
A) Ideal Gas Law
The Ideal Gas Law (PV = nRT) combines multiple gas relationships into a single equation incorporating pressure volume moles temperature and the gas constant. While it contains Boyle's Law as a special case when n and T are constant it is not the specific law describing only the inverse P-V relationship at constant temperature.
B) Boyle's Law
Robert Boyle experimentally demonstrated in 1662 that compressing a gas syringe reduced volume while increasing pressure proportionally. When volume halved pressure doubled maintaining constant PV product. This inverse relationship arises because gas molecules in smaller volumes strike container walls more frequently per unit area increasing measured pressure. Temperature constancy ensures molecular kinetic energy remains unchanged so pressure changes stem solely from altered collision frequency not molecular speed.
C) Charles' Law
Charles' Law describes direct volume-temperature proportionality at constant pressure (V ∠T) not pressure-volume relationships. Heating gas in a flexible container increases volume as molecules move faster and push walls outward. This law addresses thermal expansion not compression effects.
D) Stefan-Boltzmann Law
The Stefan-Boltzmann Law relates to blackbody radiation stating that radiant energy emitted by a blackbody is proportional to the fourth power of its absolute temperature (E = σTâ´). This thermodynamic radiation principle has no connection to gas pressure-volume relationships.
Conclusion:
Boyle's Law specifically and exclusively defines the inverse pressure-volume relationship for gases under constant temperature and mass conditions. Option B correctly identifies this foundational gas law essential for understanding respiratory mechanics scuba diving physics and engineering applications involving gas compression where temperature effects must be controlled to isolate pressure-volume dynamics.
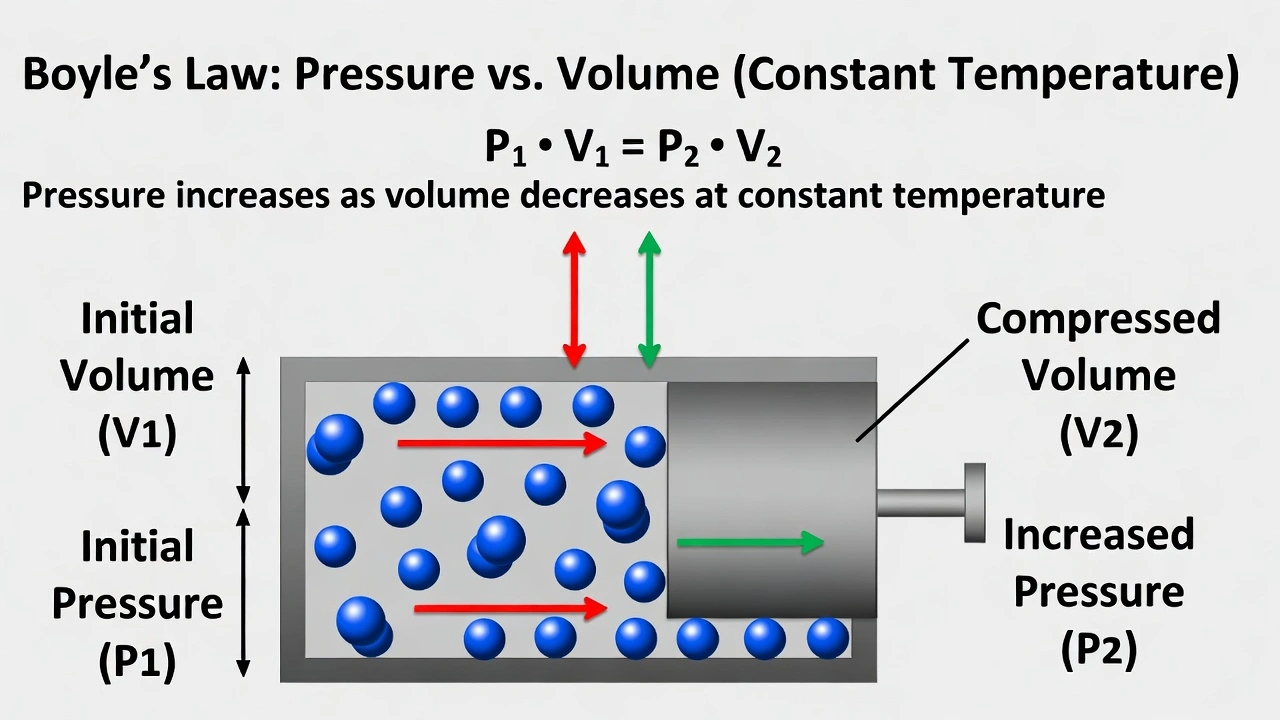
Which law of classical thermodynamics states that energy can neither be created nor destroyed?
A.
Zeroth
B. First
C. Second
D. Third
Rationale
The First Law of Thermodynamics states that energy cannot be created or destroyed. The First Law formalizes conservation of energy asserting that the total energy of an isolated system remains constant though it may transform between forms such as kinetic potential thermal chemical or electromagnetic with energy transfers occurring as work or heat.
A) Zeroth
The Zeroth Law establishes thermal equilibrium concepts stating that if two systems are each in thermal equilibrium with a third system they are in equilibrium with each other. This law provides the foundation for temperature measurement but does not address energy conservation.
B) First
The First Law expresses energy conservation mathematically as ΔU = Q - W where ΔU represents change in internal energy Q represents heat added to the system and W represents work done by the system. This principle prohibits perpetual motion machines of the first kind that would create energy from nothing. All energy transformations merely convert existing energy from one form to another with the universe's total energy remaining constant.
C) Second
The Second Law addresses entropy stating that isolated systems evolve toward maximum disorder (ΔS_universe ≥ 0) and that heat flows spontaneously only from hot to cold objects. It explains why energy transformations are never 100% efficient with some energy always dissipating as unusable heat but does not state that energy is conserved.
D) Third
The Third Law states that entropy of a perfect crystal approaches zero as temperature approaches absolute zero providing an absolute reference point for entropy calculations. It has no relation to energy conservation principles.
Conclusion:
Energy conservation represents the foundational principle of the First Law of Thermodynamics governing all physical chemical and biological processes. Option B correctly identifies this law essential for understanding why perpetual motion machines are impossible energy accounting in metabolic pathways and the fundamental constraint that all work requires energy input from existing sources.
Which type of cholesterol is considered to be the best for health?
A.
LDL
B. HDL
C. VLDL
D. VHDL
Rationale
HDL cholesterol is considered best for health. High-density lipoprotein (HDL) transports cholesterol from peripheral tissues back to the liver for excretion or recycling in a process called reverse cholesterol transport earning it the designation "good cholesterol" with higher levels associated with reduced cardiovascular disease risk.
A) LDL
Low-density lipoprotein (LDL) carries cholesterol from the liver to peripheral tissues where it can deposit in arterial walls contributing to atherosclerotic plaque formation. LDL is termed "bad cholesterol" with elevated levels strongly correlated with increased heart attack and stroke risk. Statin medications primarily target LDL reduction.
B) HDL
HDL particles scavenge excess cholesterol from arterial walls and cell membranes transporting it to the liver via apolipoprotein A-I mediated pathways. The liver then excretes cholesterol as bile acids or free cholesterol. HDL also possesses antioxidant and anti-inflammatory properties protecting endothelial function. Epidemiological studies show each 1 mg/dL increase in HDL correlates with 2-3% reduction in coronary heart disease risk.
C) VLDL
Very low-density lipoprotein (VLDL) transports endogenous triglycerides from the liver to adipose and muscle tissues. As VLDL loses triglycerides it becomes intermediate-density lipoprotein then LDL. Elevated VLDL contributes to atherogenic dyslipidemia and is associated with metabolic syndrome and insulin resistance.
D) VHDL
VHDL (very high-density lipoprotein) is not a recognized lipoprotein classification in human physiology. Standard lipoprotein categories include chylomicrons VLDL IDL LDL and HDL with no VHDL category existing in medical literature.
Conclusion:
HDL's protective role in reverse cholesterol transport and cardiovascular health distinguishes it as the beneficial cholesterol carrier. Option B correctly identifies HDL as the healthiest cholesterol type essential for understanding lipid management guidelines where raising HDL complements LDL lowering in comprehensive cardiovascular risk reduction strategies.
NEX Exams
NEX Quizzes
3 Practice Tests
NEX Quizzes
3 Practice Tests
NEX Quizzes
3 Practice Tests
NEX Quizzes
3 Practice Tests