NLN SCIENCE PRACTICE TEST
Build confidence in science-related problem solving with structured practice aligned to nursing entrance examination requirements.
Topics Covered
Anatomy terminology
Physiology processes
Biological systems
Chemical measurements
Scientific vocabulary
Applied health science concepts
00:00
What is the LAST phase of mitosis?
A.
Prophase
B. Telophase
C. Anaphase
D. Metaphase
Rationale
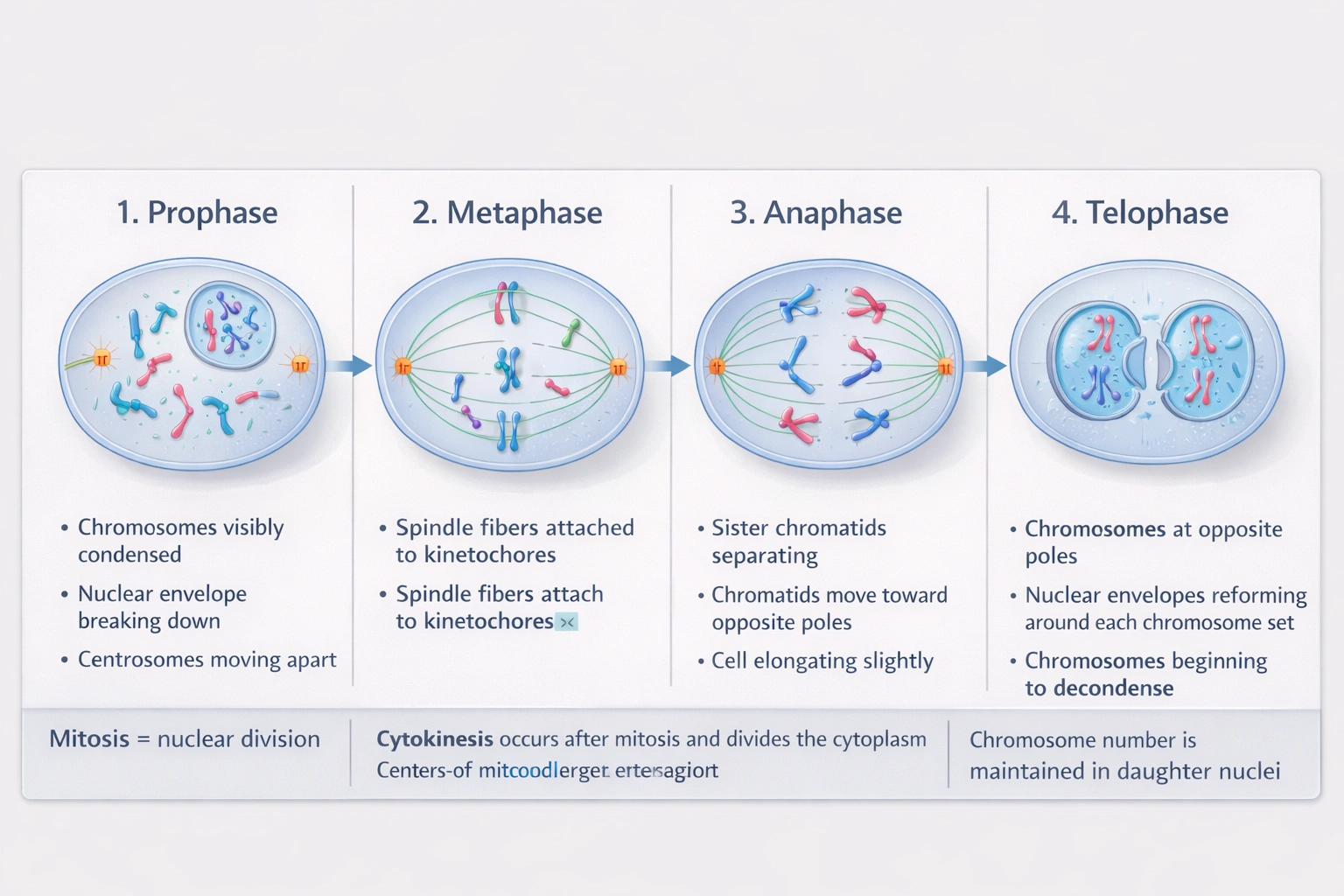
Telophase represents the final stage of mitosis where separated chromatids reach opposite poles, nuclear envelopes reassemble around each chromosome set, and chromosomes begin decondensing—preceding cytokinesis that physically divides the cytoplasm.
Mitosis progresses through four sequential phases: prophase (chromosome condensation, spindle formation), metaphase (chromosome alignment at equator), anaphase (sister chromatid separation), and telophase (nuclear reformation)—with telophase completing nuclear division before cytoplasmic division.
A) Prophase
Prophase initiates mitosis with chromatin condensation into visible chromosomes, centrosome migration to poles forming mitotic spindles, and nuclear envelope breakdown. This first phase establishes the mechanical apparatus for chromosome movement but precedes alignment, separation, and nuclear reformation—making it the initial rather than terminal stage.
B) Telophase
Telophase concludes nuclear division as: (1) separated chromatids (now individual chromosomes) arrive at opposite spindle poles; (2) nuclear envelope vesicles reassemble around each chromosome set, reforming distinct nuclei; (3) chromosomes decondense back into diffuse chromatin; and (4) nucleoli reappear within each new nucleus. Cytokinesis typically overlaps with late telophase but represents a separate process dividing cytoplasm—mitosis strictly refers to nuclear division completed at telophase termination.
C) Anaphase
Anaphase follows metaphase as sister chromatids separate synchronously and move toward opposite poles via kinetochore microtubule shortening and polar microtubule elongation. While critical for equal chromosome distribution, anaphase precedes nuclear reformation—chromosomes remain within a single cellular compartment without distinct nuclei until telophase completes nuclear envelope reassembly.
D) Metaphase
Metaphase positions chromosomes along the metaphase plate (equatorial plane) through balanced tension from opposing spindle fibers—a checkpoint ensuring proper bipolar attachment before anaphase initiation. This intermediate alignment phase occurs after prophase but before chromatid separation, placing it third in the four-phase sequence.
Conclusion:
Mitotic phase sequence follows strict temporal order essential for accurate chromosome segregation: prophase → metaphase → anaphase → telophase. Telophase uniquely completes nuclear division through nuclear envelope reformation and chromosome decondensation—distinguishing it as mitosis' terminal phase. Confusion sometimes arises from cytokinesis (cytoplasmic division) occurring concurrently with late telophase, but cytokinesis is mechanistically distinct from mitosis proper. Option B correctly identifies telophase as mitosis' final nuclear division stage—a critical distinction for understanding cell cycle regulation where mitotic exit requires telophase completion before G1 phase reentry.
What is the sensory threshold?
A.
The smallest amount of stimulus required for an individual to feel a sensation
B. The amount of stimulus required for an individual to feel pain
C. The amount of stimulus required to cause an individual to move away from the stimulus
D. The place where the stimulus is coming from
Rationale
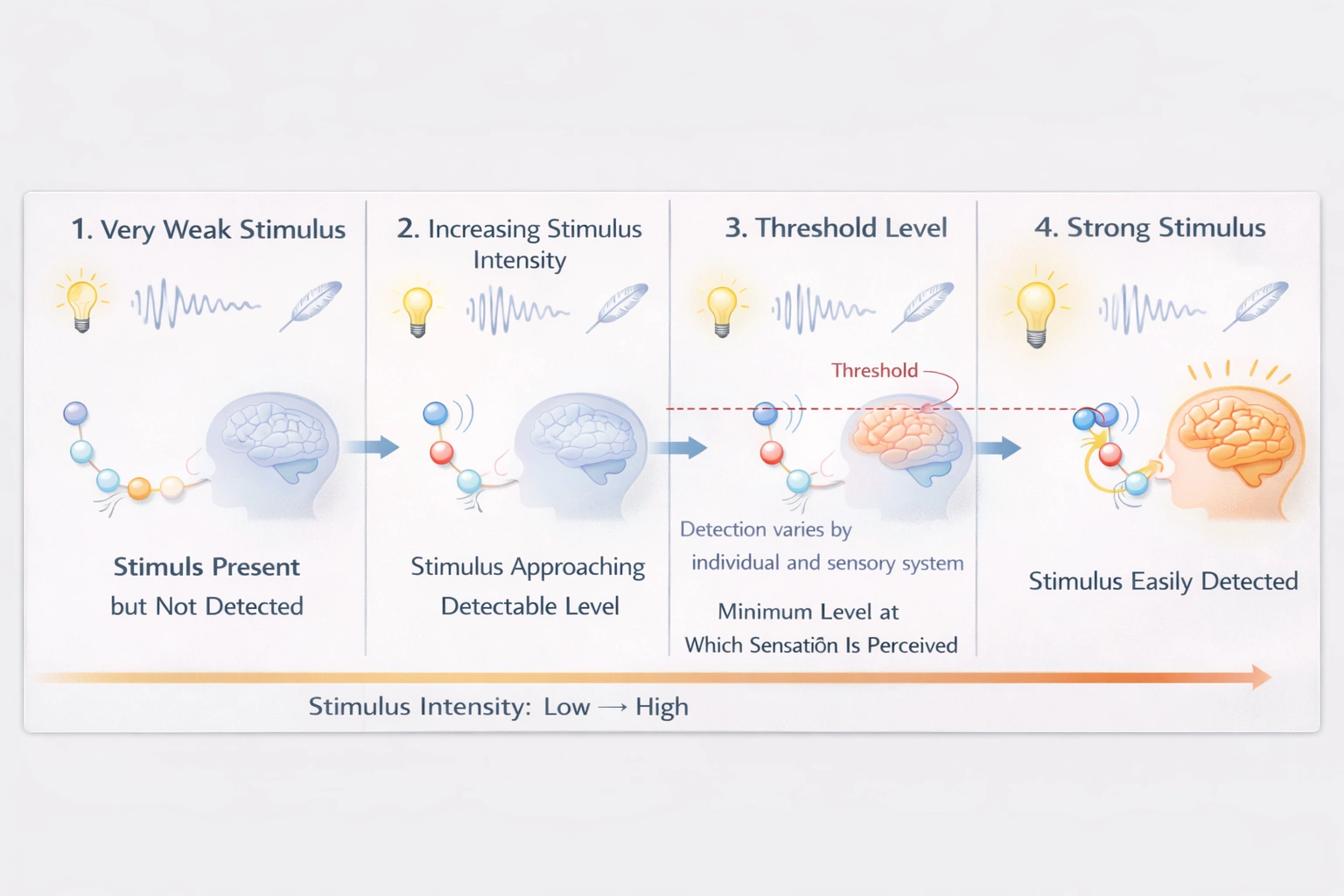
The sensory threshold represents the minimum stimulus intensity required to activate sensory receptors and generate a neural signal perceived consciously as a sensation—varying by modality and individual sensitivity.
Sensory thresholds establish the boundary between subthreshold stimuli (undetectable) and suprathreshold stimuli (detectable), governed by receptor sensitivity, neural adaptation, and central nervous system processing—fundamental to psychophysics and sensory physiology.
A) The smallest amount of stimulus required for an individual to feel a sensation
This definition aligns with the absolute threshold concept in psychophysics—the minimum detectable stimulus intensity producing sensation 50% of the time under controlled conditions. Examples: detecting a candle flame 30 miles away on dark nights (vision), hearing a ticking watch 20 feet away in quiet rooms (audition), or sensing a bee wing falling on cheek from 1 cm height (touch). Thresholds vary by sensory modality, receptor density, attention, and adaptation state—reflecting biological limits of detection rather than fixed physical constants.
B) The amount of stimulus required for an individual to feel pain
Pain threshold represents a specific subtype (nociceptive threshold) rather than the general sensory threshold concept. Many sensations occur below pain thresholds (light touch, warm temperatures, faint sounds). Pain involves specialized nociceptors activated only by potentially damaging stimuli—making this option too narrow to define the universal sensory threshold concept applicable across all modalities.
C) The amount of stimulus required to cause an individual to move away from the stimulus
This describes a behavioral response threshold influenced by motivation, context, and cognitive evaluation—not the physiological detection threshold. Individuals may detect stimuli without withdrawing (e.g., tolerating mild discomfort during medical procedures) or withdraw from subthreshold stimuli due to anticipation/anxiety. Response thresholds involve higher-order processing beyond basic sensation detection.
D) The place where the stimulus is coming from
Stimulus localization involves spatial discrimination capabilities of sensory systems (e.g., two-point discrimination in touch, sound localization in audition)—distinct from detection threshold. Localization requires multiple receptors and neural comparison mechanisms, whereas threshold concerns minimum intensity for any detection regardless of location identification.
Conclusion:
Sensory thresholds define the fundamental limits of perceptual detection across all sensory modalities—establishing the minimum stimulus energy required to activate receptor potentials that propagate to conscious awareness. Option A correctly identifies this universal psychophysical concept, distinguishing it from modality-specific thresholds (B), behavioral responses (C), or spatial discrimination (D). Understanding sensory thresholds proves essential for designing sensory aids, establishing safety standards (e.g., warning signal intensities), and diagnosing neurological conditions where threshold abnormalities indicate receptor or neural pathway dysfunction. The 50% detection criterion accounts for probabilistic neural firing and attentional fluctuations inherent in biological sensory systems.
What type of vessel carries oxygen-rich blood from the heart to other tissues of the body?
A.
Veins
B. Intestines
C. Bronchioles
D. Arteries
Rationale
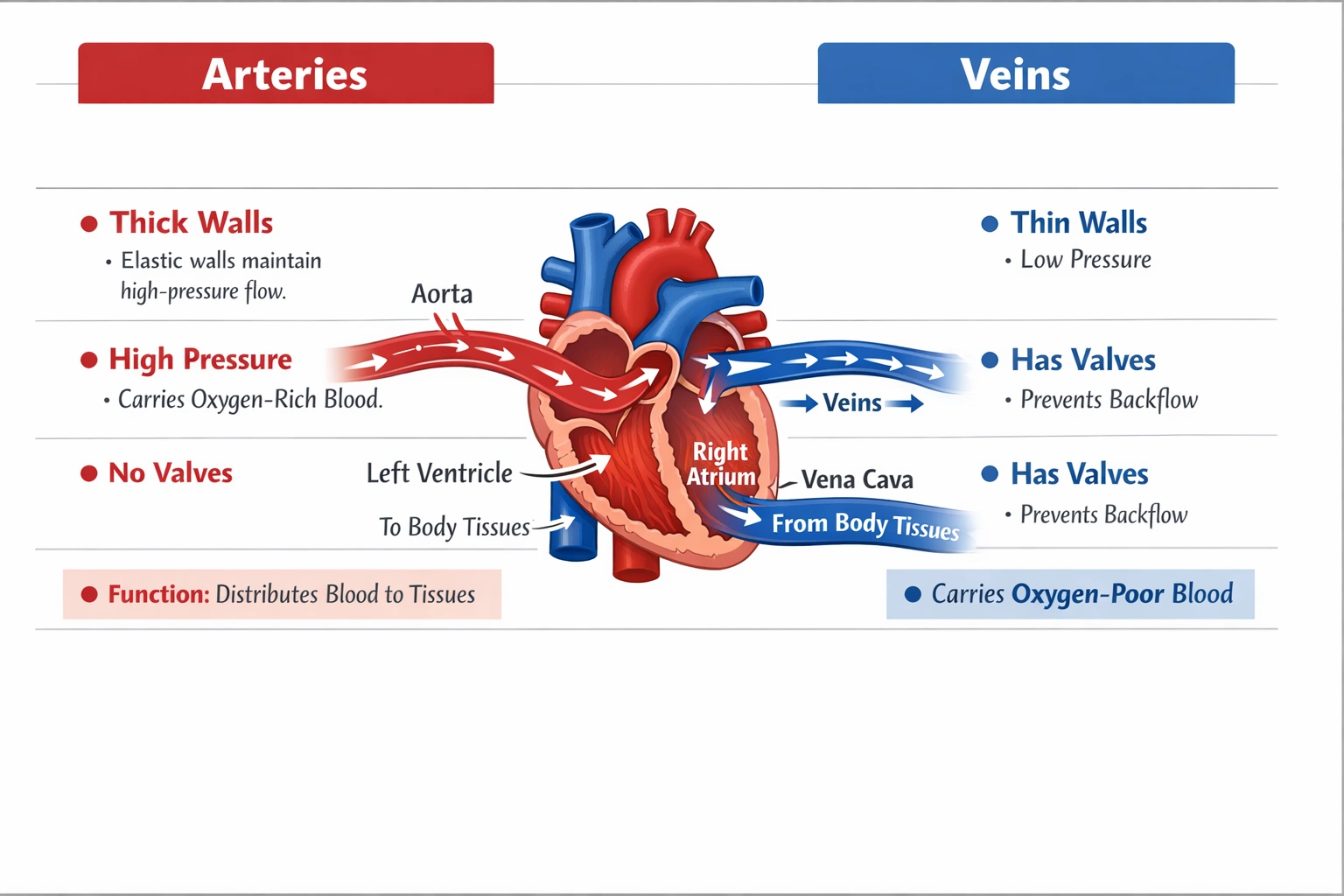
Systemic arteries transport oxygenated blood from the left ventricle through the aorta and its branches to peripheral tissues—characterized by thick, elastic walls with smooth muscle enabling high-pressure distribution throughout the body.
Arterial structure and function reflect adaptation to high-pressure ejection from the heart, with elastic recoil maintaining continuous flow during diastole and muscular regulation of regional blood distribution through vasoconstriction/vasodilation.
A) Veins
Veins return deoxygenated blood from tissues to the heart (except pulmonary veins carrying oxygenated blood from lungs to left atrium). Characterized by thin walls, large lumens, and valves preventing backflow under low pressure, veins function as capacitance vessels storing ~70% of blood volume—not high-pressure oxygen delivery conduits. Systemic veins carry oxygen-poor blood; confusing them with arteries represents a fundamental circulatory misconception.
B) Intestines
Intestines constitute digestive organs—not blood vessels. While intestinal walls contain extensive capillary networks for nutrient absorption, the intestines themselves don't transport blood between heart and tissues. This option confuses organ systems with vascular structures, representing a categorical error.
C) Bronchioles
Bronchioles are respiratory airways conducting air within lungs—not blood vessels. They branch from bronchi to terminal bronchioles ending at alveolar ducts where gas exchange occurs. Bronchioles participate in ventilation (air movement), not circulation (blood transport)—confusing respiratory and circulatory systems.
D) Arteries
Systemic arteries carry oxygenated blood under high pressure (systolic ~120 mmHg) from the left ventricle through elastic arteries (aorta, major branches) that dampen pulsatile flow via elastic recoil, then through muscular arteries that distribute blood to specific regions with smooth muscle-mediated diameter regulation. Arterioles provide final resistance regulation before capillary exchange. Pulmonary arteries uniquely carry deoxygenated blood to lungs—but the question specifies vessels carrying oxygen-rich blood to other tissues, referring exclusively to systemic circulation where arteries universally transport oxygenated blood.
Conclusion:
Arteries serve as the high-pressure distribution network delivering oxygenated blood from the heart's left ventricle to all body tissues except the lungs. Their structural adaptations—thick elastic/muscular walls, absence of valves (except semilunar valves at heart exits), and pressure-resistant design—distinguish them from low-pressure venous return vessels. Option D correctly identifies arteries as the oxygen-rich blood conduits to peripheral tissues, while other options confuse vessel types (A), organ systems (B, C), or pulmonary/systemic circulation distinctions. Understanding this fundamental circulatory principle proves essential for clinical applications—arterial blood gas sampling accesses oxygenated blood, arterial blockages cause tissue ischemia, and arterial pressure measurement reflects cardiac output and vascular resistance critical for cardiovascular assessment.
Which blood component is chiefly responsible for clotting?
A.
Platelets
B. Red blood cells
C. Antigens
D. Plasma cells
Rationale
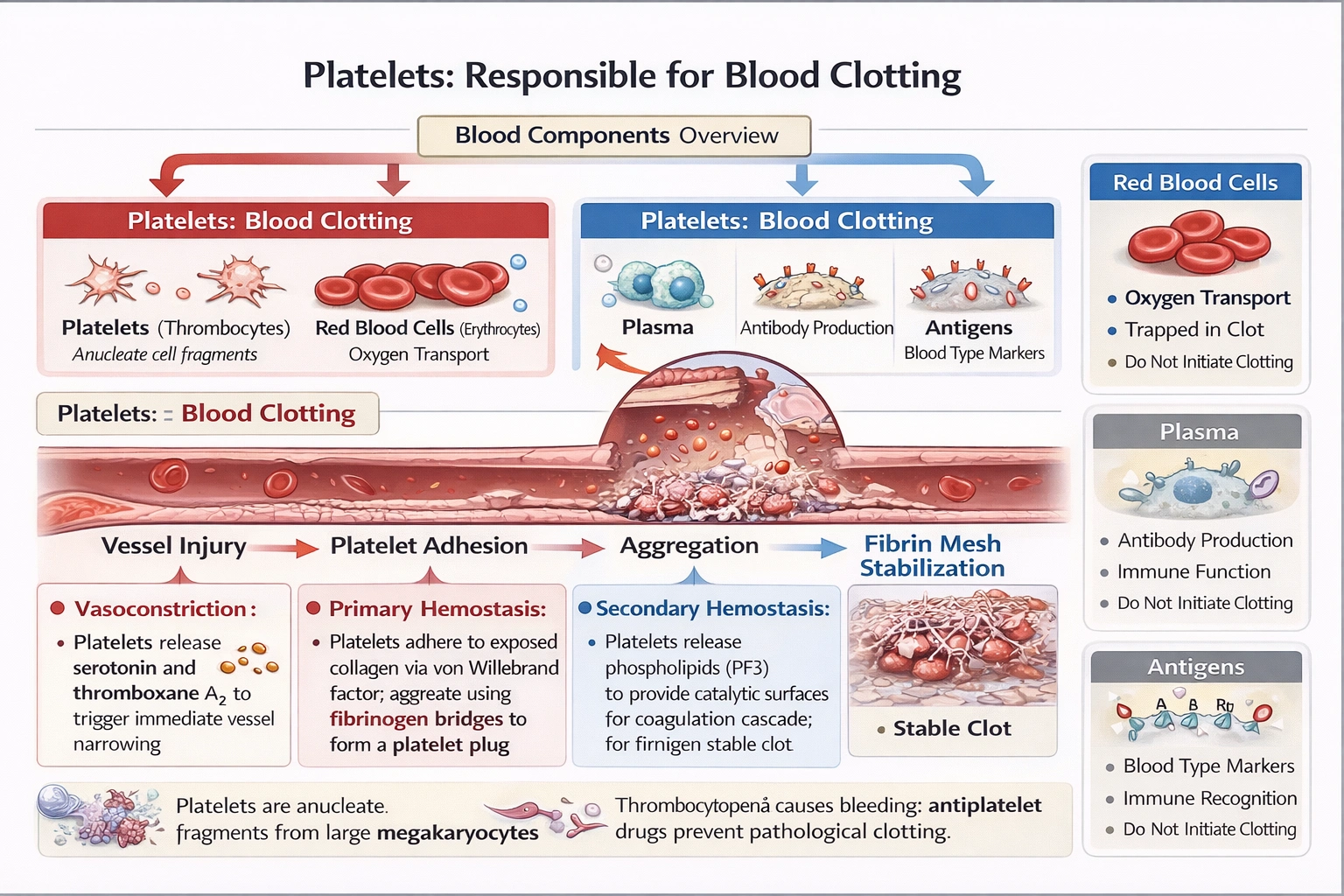
Platelets (thrombocytes) serve as the primary cellular mediators of hemostasis—adhering to damaged endothelium, aggregating to form temporary plugs, and releasing clotting factors that catalyze fibrin mesh formation for stable clot development.
Platelets function as anucleate cytoplasmic fragments derived from megakaryocytes, containing granules with ADP, serotonin, thromboxane A₂, and clotting factors that amplify the coagulation cascade upon vascular injury—initiating the critical first response to prevent blood loss.
A) Platelets
Platelets execute three hemostatic phases:
1. Vasoconstriction: Release serotonin and thromboxane Aâ‚‚ causing immediate vessel narrowing
2. Primary hemostasis: Adhere to exposed collagen via von Willebrand factor, then aggregate through fibrinogen bridges forming platelet plugs within seconds
3. Secondary hemostasis: Release phospholipids (PF3) providing catalytic surfaces for coagulation cascade complexes (tenase, prothrombinase) that convert fibrinogen to fibrin mesh stabilizing the plug
Thrombocytopenia (low platelet count) causes petechiae and prolonged bleeding despite normal coagulation factors—demonstrating platelets' indispensable role. Antiplatelet drugs (aspirin, clopidogrel) prevent pathological clotting by inhibiting platelet activation pathways.
B) Red blood cells
Erythrocytes transport oxygen via hemoglobin but play minimal direct roles in clotting. They become trapped within fibrin meshes after clot formation but don't initiate or catalyze coagulation. Polycythemia (excess RBCs) may increase blood viscosity and thrombosis risk indirectly, but RBCs lack clotting factor storage, adhesion molecules, or aggregation capabilities essential for hemostasis initiation.
C) Antigens
Antigens are molecular markers (e.g., ABO blood group antigens on RBC surfaces) that trigger immune responses when foreign—but don't participate in clotting mechanics. Blood type compatibility affects transfusion safety but not intrinsic coagulation ability; type O individuals clot normally despite lacking A/B antigens. Antigens represent immunological identifiers, not hemostatic components.
D) Plasma cells
Plasma cells are antibody-secreting B lymphocyte derivatives functioning in adaptive immunity—not hemostasis. They produce immunoglobulins targeting pathogens but contain no clotting factors or platelet-like granules. Plasma (the liquid blood component) contains clotting factors, but plasma cells are distinct cellular elements of immune tissue—confusing "plasma" with "plasma cells" represents a critical terminology error.
Conclusion:
Platelets provide the indispensable cellular foundation for hemostasis through rapid adhesion, aggregation, and coagulation cascade activation—making them chiefly responsible for clotting initiation and amplification. While plasma clotting factors (fibrinogen, prothrombin) execute the biochemical cascade, they require platelet phospholipid surfaces for efficient reaction kinetics. Option A correctly identifies platelets as the primary clotting component, distinguishing them from oxygen carriers (B), immune markers (C), or antibody producers (D). This understanding proves clinically vital—platelet transfusions treat thrombocytopenic bleeding, while antiplatelet therapy prevents thrombotic events in cardiovascular disease, reflecting platelets' central hemostatic role.
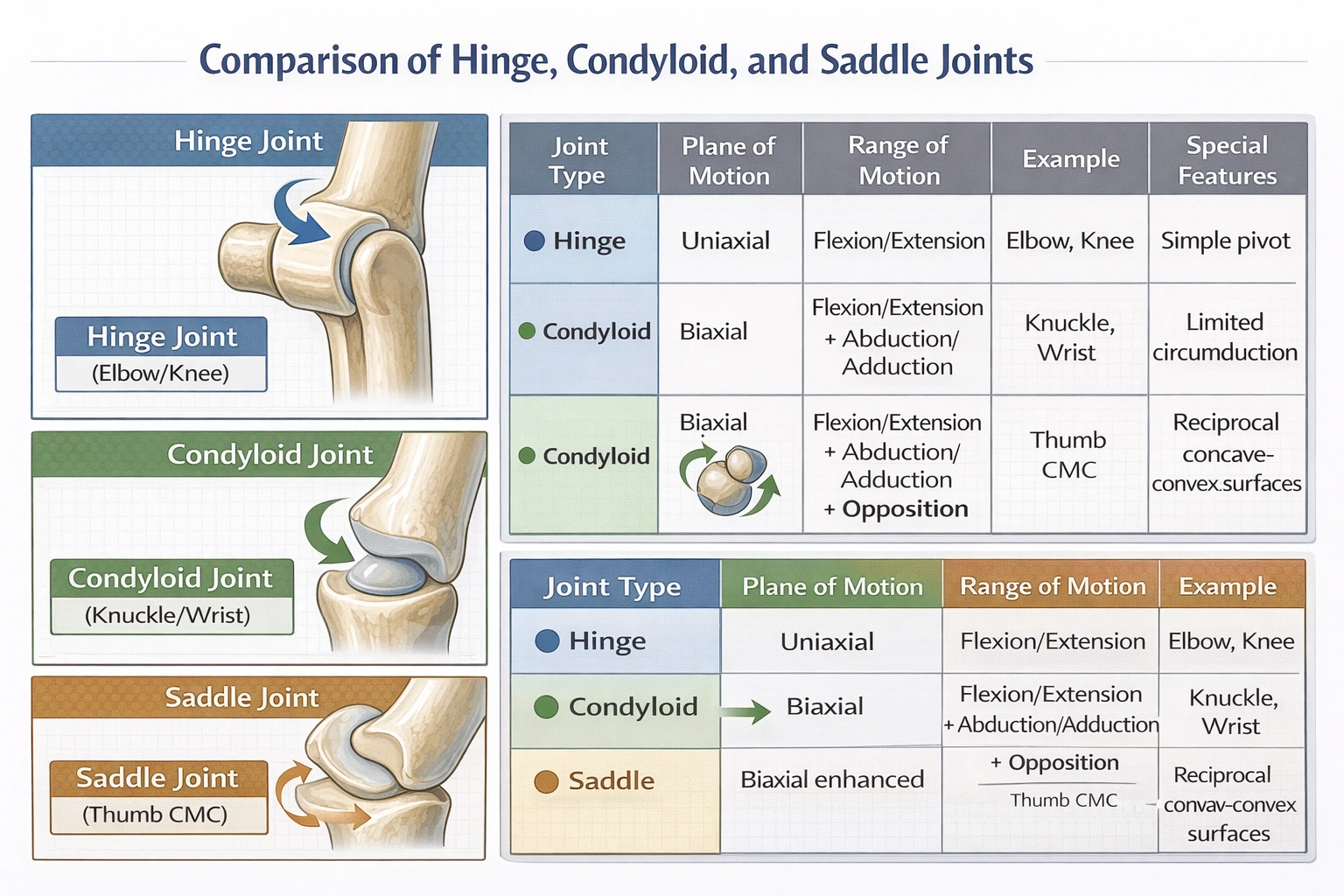
Which of the following lists of joint types is in the correct order for increasing amounts of permitted motion (least mobile to most mobile)?
A.
Hinge, condyloid, saddle
B. Saddle, hinge, condyloid
C. Saddle, condyloid, hinge
D. Hinge, saddle, condyloid
Rationale
Hinge joints permit the least motion (uniaxial movement in a single plane), condyloid joints allow intermediate mobility (biaxial movement in two planes), and saddle joints provide the greatest freedom among these three types (biaxial movement with enhanced range enabling unique opposition movements)—creating the progression hinge → condyloid → saddle for increasing mobility.
Joint mobility correlates with structural complexity: simpler hinge mechanisms restrict motion to single-plane rotation, while increasingly complex articular surface geometries permit multiplanar movement through complementary concave-convex relationships.
A) Hinge, condyloid, saddle
• Hinge joints (e.g., elbow, knee interphalangeal joints) feature convex cylindrical surfaces fitting into concave troughs—permitting only flexion and extension in a single sagittal plane (uniaxial). Range typically 0–150° with no rotation or lateral movement.
• Condyloid joints (e.g., wrist radiocarpal joint, metacarpophalangeal "knuckle" joints) possess oval convex surfaces articulating with elliptical cavities—enabling flexion/extension plus abduction/adduction in two perpendicular planes (biaxial), with limited circumduction but no axial rotation. Typical ranges: 80° flexion, 70° extension, 20° radial/ulnar deviation at wrist.
• Saddle joints (e.g., carpometacarpal joint of thumb) feature reciprocally concave-convex surfaces resembling interlocking saddles—permitting flexion/extension, abduction/adduction, AND opposition (unique combination movement bringing thumb across palm). This reciprocal geometry provides greater angular freedom than condyloid joints despite both being biaxial—thumb opposition enables precision grip impossible at condyloid joints.
This progression reflects increasing functional versatility: hinge joints provide stable levers for force transmission, condyloid joints enable planar manipulation, and saddle joints (particularly the thumb) facilitate opposable precision grip critical for tool use.
B) Saddle, hinge, condyloid
This sequence incorrectly places the most mobile joint type (saddle) first and intermediate mobility (condyloid) last—reversing the actual mobility gradient. Saddle joints demonstrably permit greater functional range than hinges (which allow only single-plane motion) and condyloid joints (lacking opposition capability).
C) Saddle, condyloid, hinge
This option ranks joints from most to least mobile—opposite of the requested "least mobile to most mobile" sequence. While anatomically accurate in relative rankings, it violates the directional requirement specified in the question.
D) Hinge, saddle, condyloid
This sequence incorrectly places saddle joints before condyloid joints in mobility ranking. While both are biaxial, saddle joints' reciprocal surface geometry permits opposition—a movement condyloid joints cannot execute—granting functionally greater mobility. The thumb's saddle joint enables 50° opposition critical for precision handling, whereas condyloid knuckle joints lack this capability despite similar angular ranges in primary planes.
Conclusion:
Joint mobility progresses systematically with structural complexity: hinge (uniaxial, single plane) → condyloid (biaxial, two planes without opposition) → saddle (biaxial with enhanced freedom enabling opposition). Option A correctly orders these types from least to most mobile—reflecting fundamental biomechanical principles where articular surface geometry directly determines movement possibilities. This understanding proves essential for clinical applications: hinge joint replacements prioritize stability over mobility, while saddle joint prosthetics (thumb CMC) must preserve opposition capability for functional hand use. Misranking joint mobility could lead to inappropriate surgical planning or rehabilitation protocols that fail to respect inherent anatomical constraints and capabilities.
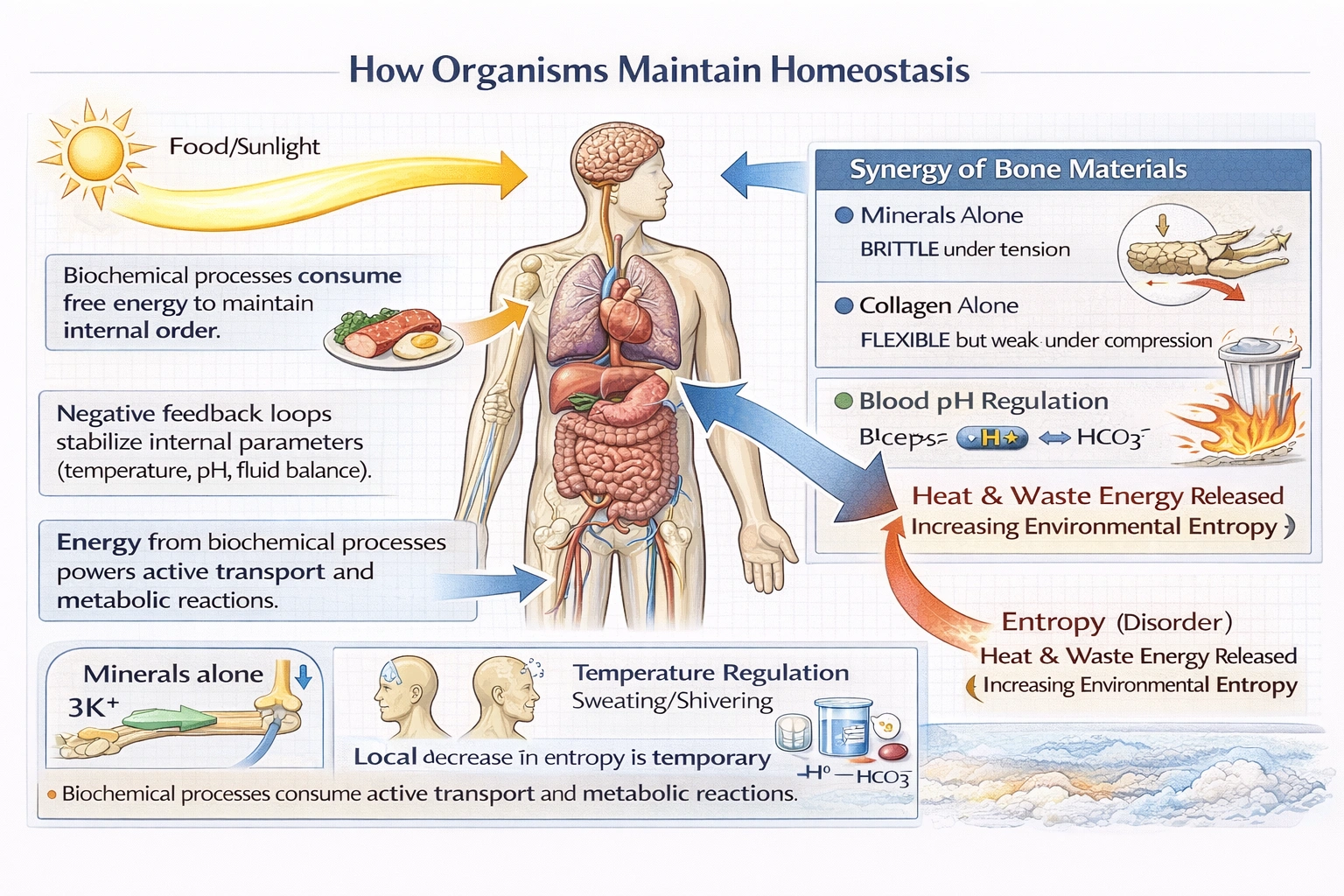
How do organisms maintain homeostasis?
A.
They increase their body temperature blood pH and fluid balance.
B. They undergo biochemical processes and absorb energy to increase entropy.
C. They undergo biochemical processes to maintain the order of their external environment.
D. They use free energy and matter via biochemical processes to work against entropy.
Rationale
Organisms maintain homeostasis by continuously expending free energy (from food or sunlight) through metabolic processes to create and sustain internal order—temporarily resisting the universal tendency toward entropy increase as mandated by the second law of thermodynamics.
Homeostasis represents a dynamic steady state requiring constant energy input to maintain concentration gradients, temperature differentials, and molecular organization against dissipative forces—making living systems localized pockets of decreasing entropy sustained by increasing entropy in their surroundings.
A) They increase their body temperature, blood pH, and fluid balance
Homeostasis maintains stable internal conditions within narrow ranges—not continuous increases. Body temperature is regulated around 37°C (not perpetually increased); blood pH is maintained near 7.4 (not progressively elevated); fluid balance is kept constant through intake/excretion equilibrium. "Increasing" these parameters would cause pathology (hyperthermia, alkalosis, fluid overload)—opposite to homeostatic goals. Homeostasis implies stability through negative feedback, not unidirectional change.
B) They undergo biochemical processes and absorb energy to increase entropy
The second law of thermodynamics states that total entropy (disorder) of universe always increases. Organisms locally decrease internal entropy (creating order) while increasing environmental entropy by greater amounts—net universal entropy still rises. Absorbing energy enables organisms to build complex molecules (decreasing local entropy), but this requires dissipating more energy as heat (increasing environmental entropy). Organisms don't absorb energy to increase entropy—they absorb energy to decrease local entropy while unavoidably increasing universal entropy as a thermodynamic necessity.
C) They undergo biochemical processes to maintain the order of their external environment
Organisms primarily regulate their internal environment (blood glucose, temperature, pH)—not the external environment. While ecosystems exhibit emergent order, individual organisms don't expend energy to organize external surroundings as a homeostatic mechanism. Beavers building dams or earthworms aerating soil represent niche construction, not homeostasis per se. Homeostasis specifically refers to internal physiological stability—external environment regulation is ecologically significant but thermodynamically distinct from organismal homeostasis.
D) They use free energy and matter via biochemical processes to work against entropy
Organisms consume free energy (chemical energy in food with negative ΔG) to power endergonic reactions that build complex structures (proteins from amino acids, concentration gradients via active transport). This creates localized order (decreased entropy) within the organism. However, the second law isn't violated because energy transformations are inefficient—approximately 60% of food energy dissipates as heat, increasing environmental entropy by more than the internal entropy decrease. The net universal entropy still increases, satisfying thermodynamics while permitting temporary local order maintenance. Homeostasis exemplifies this principle: sodium-potassium pumps use ATP to maintain ionic gradients (order), but ATP hydrolysis releases heat (disorder) exceeding the gradient's order value.
Conclusion:
Homeostasis requires continuous energy expenditure to maintain internal order against entropic decay—a temporary local defiance of entropy increase enabled by greater entropy production in the surroundings. Option D correctly describes this thermodynamic reality: organisms use free energy to power processes that sustain internal organization (working against local entropy) while unavoidably increasing universal entropy through heat dissipation. Options A, B, and C misrepresent homeostasis as unidirectional parameter increase, entropy promotion, or external environment regulation—fundamentally misunderstanding both homeostatic mechanisms and thermodynamic principles governing living systems. This understanding proves essential for comprehending metabolism's purpose (energy capture for order maintenance), aging (gradual entropy increase overcoming regulatory capacity), and ecosystem energetics (energy flow enabling biological organization).
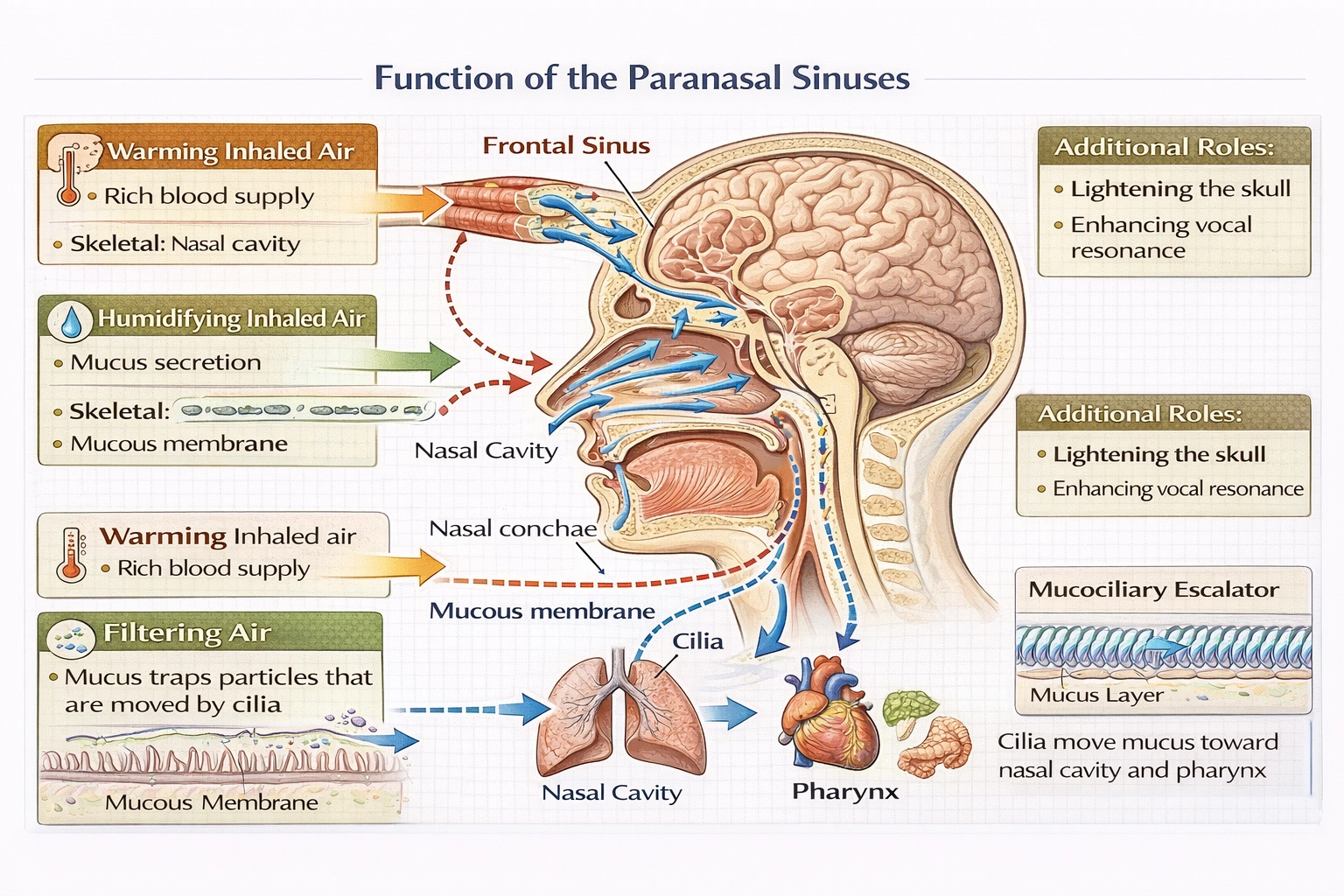
What is the function of the sinuses?
A.
To trap the many airborne pathogens
B. To direct air down the trachea rather than the esophagus
C. To warm, humidify, and filter air
D. To sweep away pathogens and direct them toward the top of the trachea
Rationale
The paranasal sinuses contribute to warming, humidifying, and filtering inhaled air, working in concert with nasal conchae and mucosa to condition respiratory air before it reaches delicate lung tissues while also reducing skull weight and enhancing vocal resonance.
Sinuses function as extensions of the nasal cavity lined with ciliated pseudostratified columnar epithelium and mucus-producing goblet cells that participate in the upper respiratory tract's air-conditioning system—though their contribution is secondary to the nasal passages themselves.
A) To trap the many airborne pathogens
Pathogen trapping occurs primarily in the nasal cavity via mucus adhesion and ciliary clearance—not specifically in sinuses. Sinuses connect to nasal passages through small ostia that limit direct airflow; most inhaled air passes through nasal meatuses rather than entering sinuses significantly. While sinus mucosa contains immune cells, pathogen trapping isn't their primary function—nasal conchae and pharyngeal tonsils serve this role more directly. Sinus infections (sinusitis) actually represent pathological states where trapped pathogens cause inflammation—not evidence of designed trapping function.
B) To direct air down the trachea rather than the esophagus
Airway/esophagus separation is accomplished by the epiglottis and laryngeal structures during swallowing—not sinuses. The epiglottis covers the laryngeal inlet during deglutition, directing food/liquid toward the esophagus while maintaining air passage through the trachea. Sinuses are air-filled cavities in skull bones with no involvement in airway protection or directional guidance during swallowing.
C) To warm, humidify, and filter air
Sinuses contribute to respiratory air conditioning through:
• Warming: Rich vascularization of sinus mucosa transfers body heat to inhaled air
• Humidifying: Mucous secretions add moisture to prevent desiccation of lower respiratory tract
• Filtering: Mucus traps larger particles; ciliary action moves debris toward nasal cavity for expulsion
Though nasal conchae provide the primary surface area for these functions, sinuses augment the process by increasing total mucosal surface area and creating turbulence that enhances contact between air and conditioning surfaces. Sinuses also lighten the skull (reducing neck muscle burden) and act as resonance chambers for vocalization—secondary functions supporting their evolutionary retention.
D) To sweep away pathogens and direct them toward the top of the trachea
Mucociliary clearance moves trapped particles toward the pharynx (not trachea) for swallowing or expectoration—following the "mucociliary escalator" from peripheral airways upward to throat. Sinuses drain into nasal passages via ostia, with cilia moving mucus toward these drainage openings—not toward the trachea. Directing debris toward the trachea would deposit contaminants deeper into lungs—counterproductive to respiratory defense. This option reverses the actual clearance direction and misattributes the mechanism to sinuses rather than the broader respiratory epithelium.
Conclusion:
Sinuses primarily assist in respiratory air conditioning (warming, humidifying, filtering) as extensions of the nasal cavity's mucosal surface—while providing secondary benefits of skull lightening and vocal resonance. Option C correctly identifies their respiratory contribution, distinguishing it from pathogen trapping as a primary function (A), airway protection mechanisms (B), or misdirected clearance pathways (D). Understanding sinus function proves clinically relevant: sinus ostia blockage during colds impairs drainage causing pressure/pain, and chronic sinusitis disrupts normal air conditioning contributing to lower respiratory irritation. The sinuses' role in air preparation exemplifies upper respiratory tract integration—where multiple structures collaborate to protect delicate alveolar surfaces from temperature extremes, desiccation, and particulate damage.
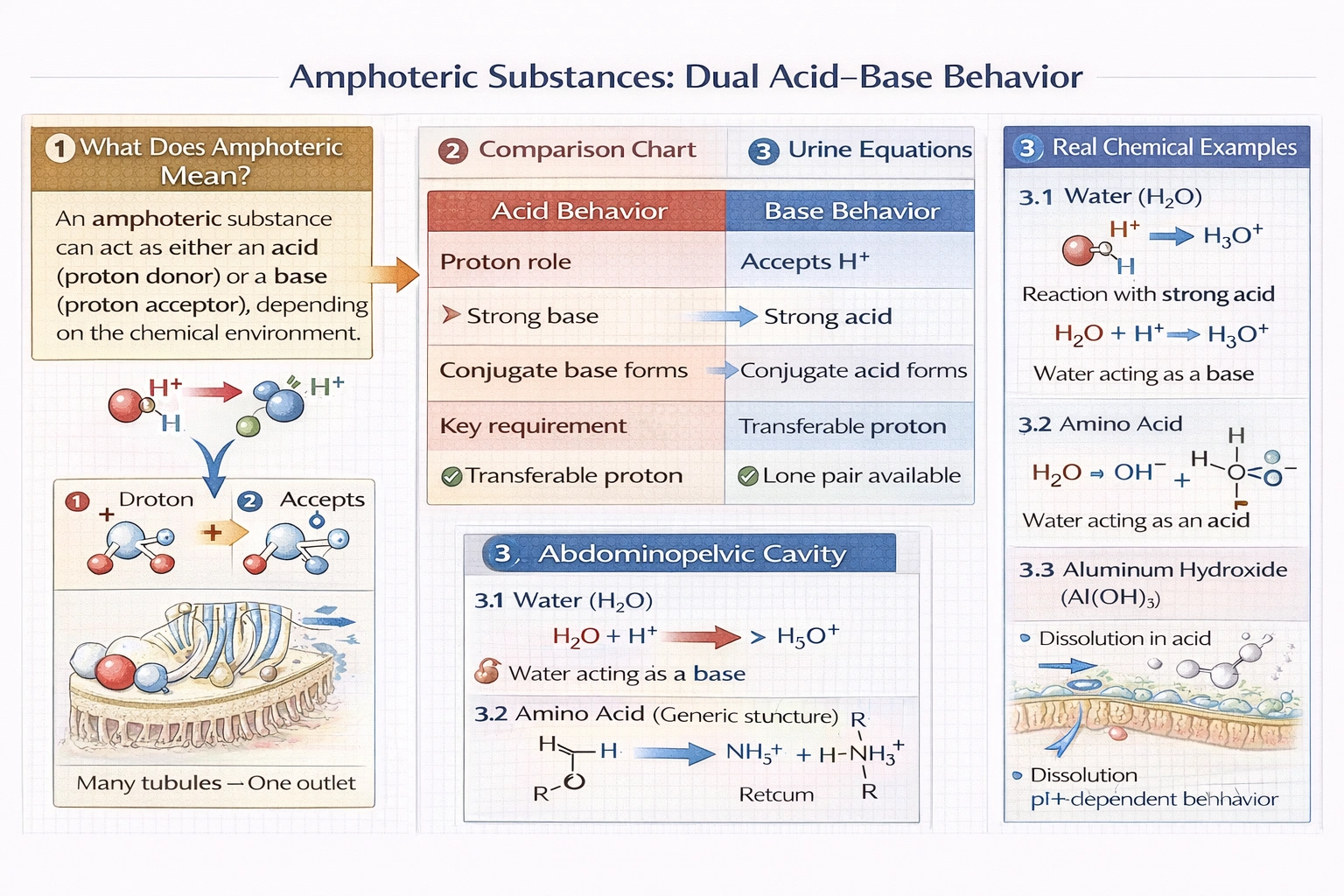
Which of the following best defines the term amphoteric?
A.
A substance that conducts electricity due to ionization when dissolved in a solvent
B. A substance that can act as an acid or a base depending on the properties of the solute
C. A substance that, according to the Brønsted-Lowry Acid-Base Theory, is a proton-donor
D. A substance that donates its proton and forms its conjugate base in a neutralization reaction
Rationale
An amphoteric substance can function as either an acid (proton donor) or a base (proton acceptor) depending on the chemical environment—exemplified by water, amino acids, and aluminum hydroxide that react differently with strong acids versus strong bases.
Amphoterism arises from molecular structures containing both proton-donating groups (e.g., -OH, -COOH) and proton-accepting sites (e.g., lone pairs on O, N)—enabling dual reactivity that depends on the relative strength of surrounding acids or bases rather than inherent fixed behavior.
A) A substance that conducts electricity due to ionization when dissolved in a solvent
This defines an electrolyte—not an amphoteric substance. While many amphoteric compounds ionize in solution (e.g., water autoionizes to H₃O⺠and OHâ»), ionization and electrical conductivity aren't defining characteristics of amphoterism. Many electrolytes aren't amphoteric (NaCl conducts but can't act as acid/base), and some amphoteric substances show minimal ionization in certain solvents. Conductivity relates to charge carrier presence, not dual acid-base behavior.
B) A substance that can act as an acid or a base depending on the properties of the solute
Correction: "depending on the properties of the solvent or surrounding species"—amphoterism depends on reaction partners, not solute properties (the amphoteric substance is the solute). Water demonstrates classic amphoterism:
• With strong base (NHâ‚‚â»): Hâ‚‚O + NH₂⻠→ OH + NH₃ (water acts as acid, donating Hâº)
• With strong acid (HCl): Hâ‚‚O + HCl → H₃O⺠+ Clâ» (water acts as base, accepting Hâº)
Amino acids exhibit amphoterism through carboxyl group (acidic) and amino group (basic), forming zwitterions at physiological pH. Aluminum hydroxide dissolves in both strong acids (Al(OH)₃ + 3HCl → AlCl₃ + 3H₂O) and strong bases (Al(OH)₃ + NaOH → Na[Al(OH)₄])—demonstrating dual reactivity defining amphoterism.
C) A substance that, according to the Brønsted-Lowry Acid-Base Theory, is a proton-donor
This defines a Brønsted-Lowry acid exclusively—not an amphoteric substance. Amphoteric compounds can act as either proton donors or acceptors depending on context. Labeling them solely as proton donors ignores their base behavior capability—fundamentally misrepresenting amphoterism's dual nature.
D) A substance that donates its proton and forms its conjugate base in a neutralization reaction
This describes acid behavior in a specific reaction type—not amphoterism's defining characteristic. Amphoteric substances may accept protons in neutralization reactions (acting as bases) rather than donating them. For example, water accepting H⺠from HCl forms H₃Oâºâ€”water acts as base, not acid. Amphoterism requires capacity for both behaviors, not just proton donation in neutralization contexts.
Conclusion:
Amphoterism is defined by context-dependent dual acid-base behavior—functioning as proton donor with strong bases and proton acceptor with strong acids. Option B captures this essential characteristic despite minor wording imprecision ("solute" should be "solvent/reaction partner"). Options A, C, and D describe electrolyte behavior, acid-only function, or specific reaction roles—none capturing the defining dual reactivity of amphoteric substances. Understanding amphoterism proves essential for predicting chemical behavior: amino acid buffering capacity, aluminum's corrosion resistance in varied pH environments, and water's universal solvent properties all stem from amphoterism. This concept bridges acid-base theories and explains why certain substances resist pH changes or dissolve in both acidic and basic solutions—critical for biochemistry, environmental science, and industrial chemistry applications.
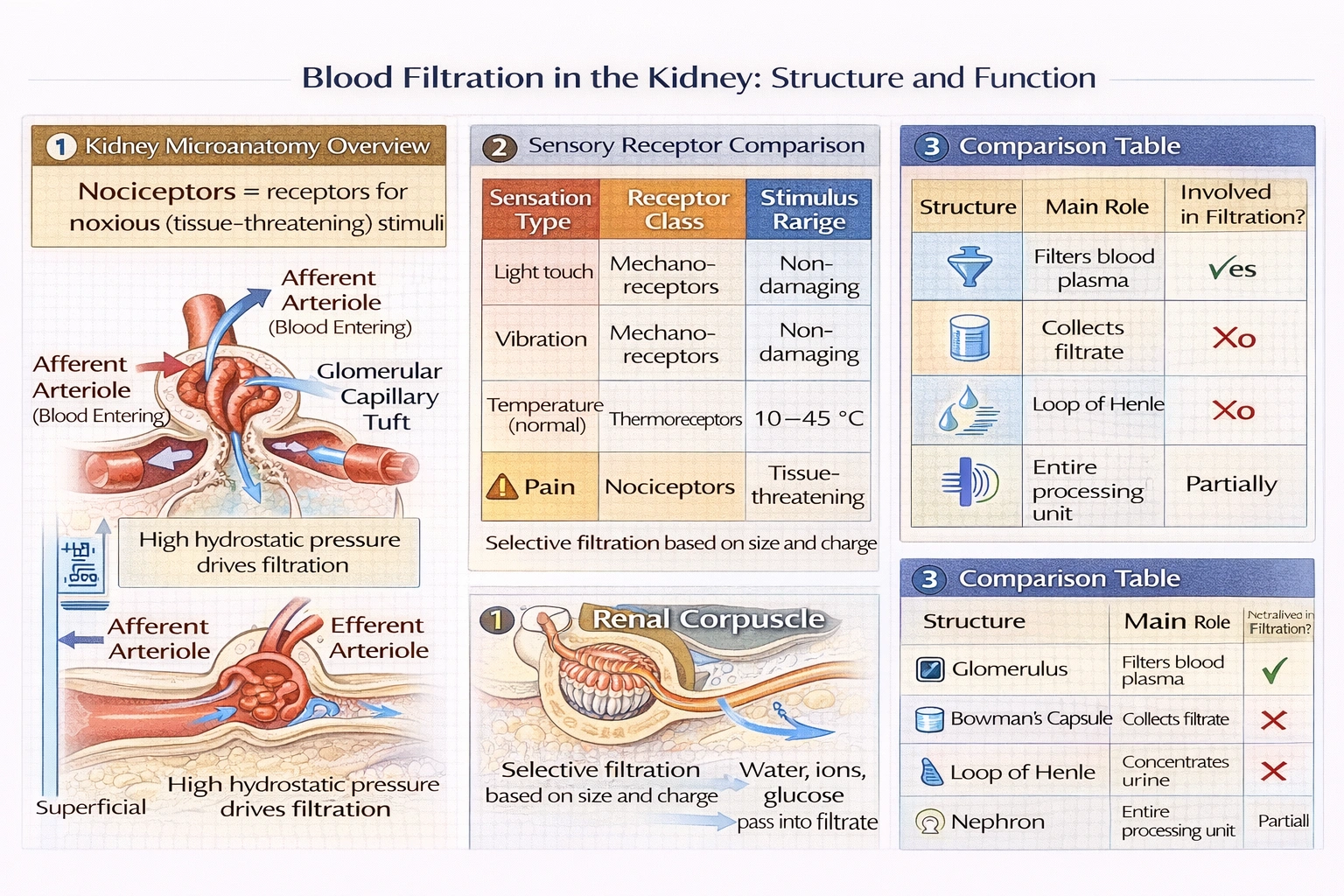
A cluster of capillaries that functions as the main filter of the blood entering the kidney is known as which of the following?
A.
The Bowman's capsule
B. The Loop of Henle
C. The glomerulus
D. The nephron
Rationale
The glomerulus is a tuft of fenestrated capillaries enclosed within Bowman's capsule, where blood plasma undergoes ultrafiltration driven by hydrostatic pressure to form the initial filtrate that enters the renal tubule system for processing into urine.
Glomerular filtration represents the first step in urine formation, with approximately 180 liters of plasma filtered daily through the glomerular basement membrane acting as a selective barrier based on molecular size and charge retaining cells and large proteins while permitting water, ions, and small solutes to pass.
A) The Bowman's capsule
Bowman's capsule is the double-walled epithelial cup surrounding the glomerulus receiving the filtered fluid (filtrate) but not performing filtration itself. Its visceral layer consists of podocytes with foot processes that wrap around glomerular capillaries, contributing to the filtration barrier's final layer. Bowman's capsule functions as the filtrate collection chamber, not the filtering structure—confusing container with filter mechanism.
B) The Loop of Henle
The loop of Henle functions in urine concentration through countercurrent multiplication establishing the medullary osmotic gradient essential for water conservation. Located distal to the glomerulus in the nephron sequence, it modifies already-formed filtrate through selective reabsorption but plays no role in initial blood filtration. Its descending and ascending limbs create the gradient enabling collecting duct water reabsorption under ADH control—distinct from filtration mechanics.
C) The glomerulus
The glomerulus consists of:
• Fenestrated endothelium: Capillary walls with 70–90 nm pores permitting plasma passage while blocking blood cells
• Basement membrane: Gel-like acellular layer of collagen IV and glycoproteins acting as size/charge barrier (blocks molecules >8 nm or negatively charged proteins)
• Podocyte slit diaphragms: Final filtration barrier between podocyte foot processes with nephrin proteins forming 4–14 nm pores
Blood enters via afferent arteriole (high pressure ~55 mmHg) and exits via efferent arteriole maintaining high intraglomerular pressure essential for filtration. This unique arrangement (arteriole-capillary-arteriole) distinguishes glomerular circulation from systemic capillary beds and enables continuous high-pressure filtration independent of systemic pressure fluctuations.
D) The nephron
The nephron is the functional unit of the kidney comprising multiple structures: renal corpuscle (glomerulus + Bowman's capsule), proximal tubule, loop of Henle, distal tubule, and collecting duct system. While filtration occurs within the nephron, the nephron itself isn't the filtering structure—it's the entire processing unit. Attributing filtration specifically to "the nephron" lacks anatomical precision; the glomerulus performs filtration while other nephron segments handle reabsorption/secretion.
Conclusion:
The glomerulus uniquely serves as the kidney's primary blood filtration structure—its specialized capillary architecture and high-pressure circulation enabling continuous plasma ultrafiltration. Option C correctly identifies this structure, distinguishing it from the filtrate collector (A), concentration apparatus (B), or entire functional unit (D). Understanding glomerular function proves essential clinically: glomerular damage (glomerulonephritis) causes proteinuria/hematuria due to barrier disruption, while reduced glomerular filtration rate (GFR) indicates kidney dysfunction requiring intervention. The glomerulus's high-pressure filtration design represents a remarkable evolutionary adaptation—enabling massive daily plasma processing (180 L) with minimal energy expenditure compared to active transport mechanisms.
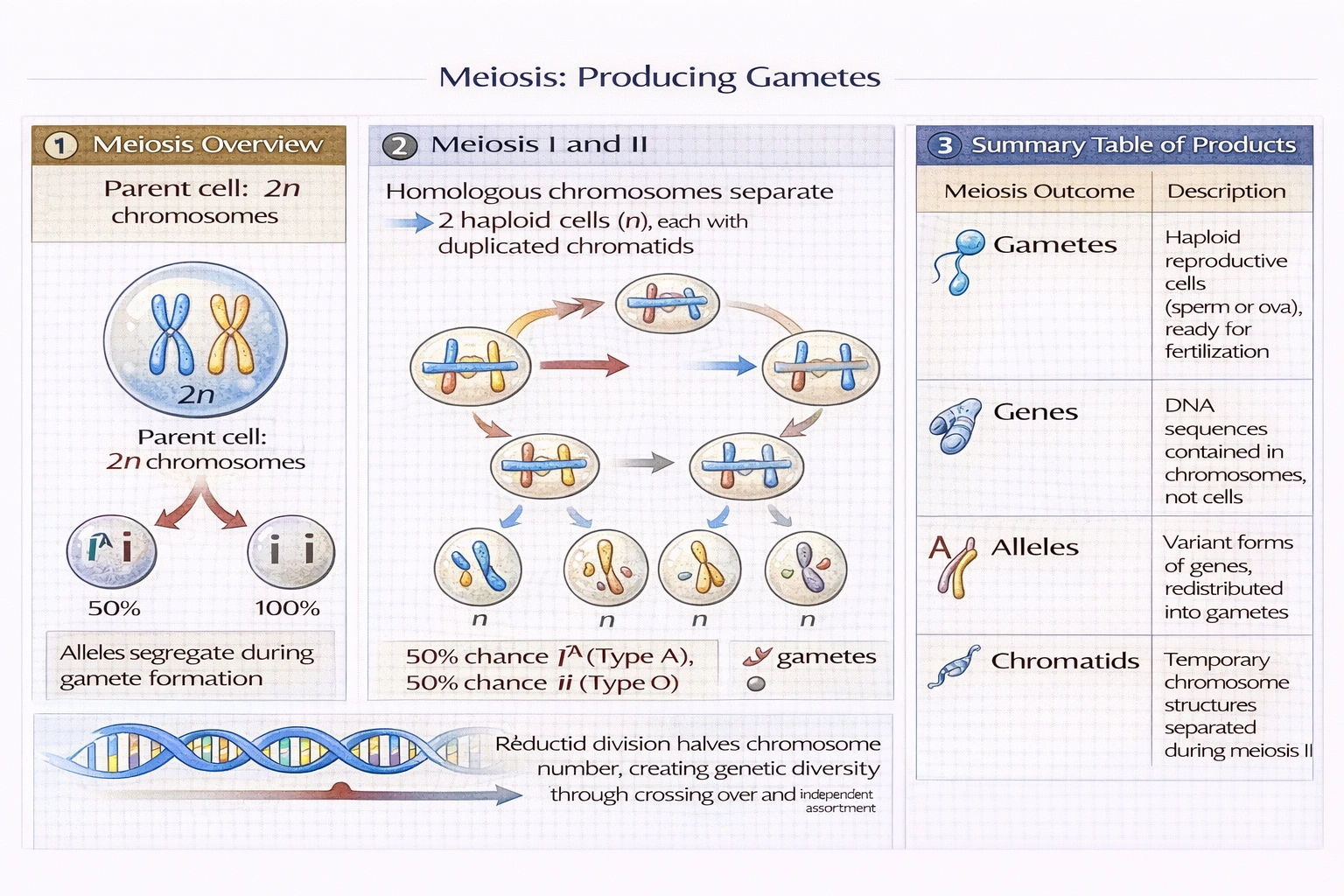
Which of the choices below are the reproductive cells produced by meiosis?
A.
Genes
B. Alleles
C. Chromatids
D. Gametes
Rationale
Gametes are the reproductive cells produced by meiosis—spermatozoa in males and ova in females are the haploid reproductive cells produced through meiosis that fuse during fertilization to restore diploidy and initiate new organism development.
Meiosis reduces chromosome number from diploid (2n) to haploid (n) through two sequential divisions without intervening DNA replication—ensuring fertilization restores species-typical chromosome number while generating genetic diversity through crossing over and independent assortment.
A) Genes
Genes are DNA sequences encoding functional products (proteins or RNAs)—not cells produced by meiosis. Genes reside on chromosomes within cells but aren't cellular products themselves. Meiosis distributes existing genes into gametes but doesn't "produce" genes as entities—genes replicate during S phase before meiosis begins. Confusing genetic information units with cellular products represents a fundamental category error.
B) Alleles
Alleles are variant forms of genes (e.g., Iᴬ versus i for ABO blood group)—molecular variants rather than cells. Meiosis segregates alleles into different gametes according to Mendel's law of segregation, but doesn't produce alleles themselves. Alleles arise through mutation and exist as DNA sequence differences; meiosis merely redistributes pre-existing allelic variants into haploid cells.
C) Chromatids
Chromatids are duplicated chromosome copies held together at centromeres after S phase—temporary structures that separate during anaphase of mitosis or meiosis II. While chromatid separation occurs during meiosis, chromatids aren't the end products—meiosis produces whole cells (gametes) containing single chromatids that become individual chromosomes after separation. Chromatids represent structural components, not cellular reproductive products.
D) Gametes
Gametes are the definitive meiotic products:
• Spermatozoa (male gametes): Produced in testes through spermatogenesis—one diploid spermatogonium yields four functional sperm
• Ova (female gametes): Produced in ovaries through oogenesis—one diploid oogonium yields one functional ovum plus polar bodies
Gametes contain half the somatic chromosome number (23 in humans), enabling diploid restoration at fertilization (23 + 23 = 46). Their haploid state results directly from meiotic reduction division—distinguishing them from mitotic products (diploid somatic cells). Gametes also carry unique genetic combinations due to crossing over (prophase I) and independent assortment (metaphase I)—providing the variation essential for evolution and selective breeding.
Conclusion:
Gametes represent the specialized haploid cells produced exclusively through meiosis for sexual reproduction—distinguished from genes/alleles (molecular information units) and chromatids (temporary chromosome structures). Option D correctly identifies gametes as meiosis' cellular products, while other options confuse molecular/genetic concepts with cellular outcomes. Understanding this distinction proves fundamental to reproductive biology: gamete production defects cause infertility, gamete fusion initiates embryogenesis, and gamete genetic diversity underlies population adaptation—making gametes the essential cellular link between generations in sexually reproducing organisms.
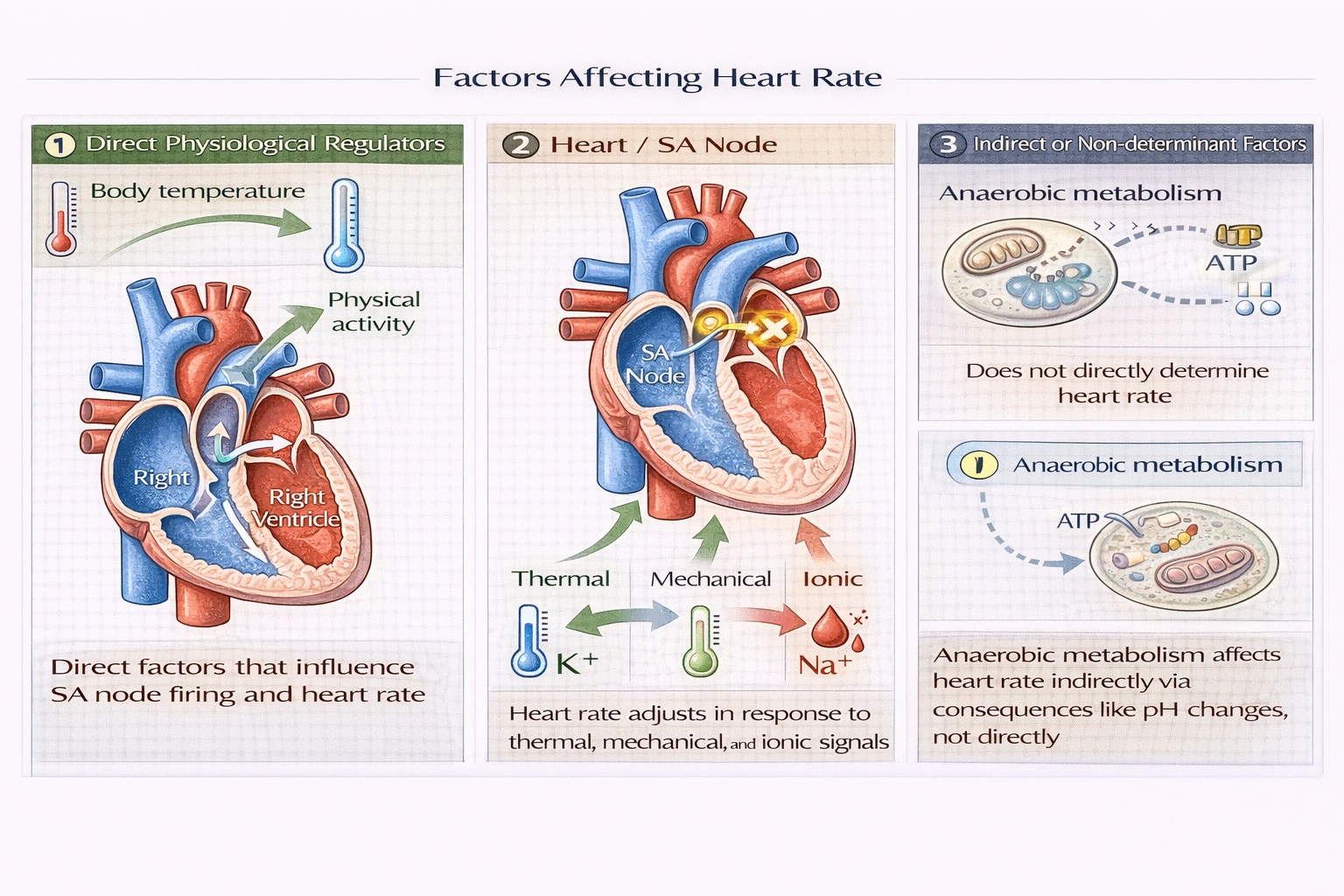
Which of the following does not help determine heart rate?
A.
body temperature
B. physical activity
C. concentration of ions
D. anaerobic cellular metabolism
Rationale
Anaerobic cellular metabolism does not directly determine heart rate. Heart rate regulation involves autonomic nervous input, hormonal signals, and direct physiological feedback mechanisms—but anaerobic metabolism represents a cellular energy state rather than a regulatory signal influencing sinoatrial node firing frequency.
A) body temperature
Body temperature directly modulates heart rate through thermoregulatory mechanisms: for every 1°C increase in core temperature, heart rate rises approximately 10 beats per minute to enhance convective heat loss via increased blood flow to skin. Hypothermia conversely slows heart rate—demonstrated clinically during therapeutic hypothermia where cooling to 33°C reduces heart rate by 20–30 bpm. Temperature-sensitive ion channels in the sinoatrial node mediate this direct thermal effect on pacemaker depolarization rate.
B) physical activity
Physical activity increases heart rate through multiple integrated mechanisms: central command signals from motor cortex, baroreceptor feedback responding to blood pressure changes, chemoreceptor detection of rising CO₂/falling pH, and circulating catecholamines (epinephrine/norepinephrine). During maximal exercise, heart rate can triple from resting levels (60 → 180 bpm) to increase cardiac output proportionally with oxygen demand—demonstrating activity's powerful regulatory influence.
C) concentration of ions
Ion concentrations critically regulate heart rate through effects on sinoatrial node action potentials: hyperkalemia (elevated Kâº) depolarizes pacemaker cells, initially increasing then decreasing heart rate as severe elevation causes conduction blocks; hypocalcemia prolongs depolarization, slowing heart rate; calcium channel blockers therapeutically reduce heart rate by inhibiting Ca²⺠influx during pacemaker potential. Electrolyte imbalances directly alter membrane potential dynamics governing automaticity.
D) anaerobic cellular metabolism
Anaerobic metabolism describes ATP production via glycolysis without oxygen—occurring in tissues during oxygen deficit—but doesn't signal heart rate changes. Heart rate responds to the consequences of anaerobic metabolism (e.g., lactic acid lowering pH detected by chemoreceptors, or CO₂ accumulation), not the metabolic state itself. Anaerobic pathways operate locally within cells without generating systemic signals that directly modulate sinoatrial node activity—making this a cellular process rather than a heart rate determinant.
Conclusion:
Heart rate regulation integrates thermal, mechanical, chemical, and neural inputs that directly influence pacemaker cell electrophysiology—while anaerobic metabolism represents an intracellular energy pathway without inherent signaling capacity for cardiac rhythm control. Options A, B, and C describe genuine regulatory factors with documented physiological mechanisms, whereas D confuses metabolic state with regulatory signal. Understanding this distinction proves clinically relevant: lactic acidosis from anaerobic metabolism indirectly increases heart rate via chemoreceptor activation, but the metabolism itself isn't the determinant—preventing misattribution of causality in critical care scenarios.
Which of the following terms matches the definition: an abnormally slow heartbeat.
A.
tachycardia
B. bradycardia
C. fibrillation
D. myocardial infarct
Rationale
Bradycardia describes an abnormally slow heartbeat. Bradycardia is clinically defined as a resting heart rate below 60 beats per minute in adults—though athletic individuals may exhibit physiological bradycardia (40–50 bpm) without symptoms, while pathological bradycardia causes dizziness, syncope, or fatigue due to inadequate cardiac output.
A) tachycardia
Tachycardia denotes an abnormally rapid heart rate exceeding 100 beats per minute at rest—opposite to the slow rate described in the question. Mechanisms include sinus tachycardia (physiological response to stress/exercise), atrial fibrillation, or ventricular tachycardia. Tachycardia reduces diastolic filling time, potentially decreasing stroke volume despite increased rate—a compensatory mechanism that becomes maladaptive when sustained.
B) bradycardia
Bradycardia originates from Greek bradys (slow) + kardia (heart). Causes include:
• Sinus node dysfunction: Sick sinus syndrome with impaired pacemaker automaticity
• Conduction blocks: AV nodal blocks (first, second, or third degree) delaying or preventing atrial impulses from reaching ventricles
• Medications: Beta-blockers, calcium channel blockers, digoxin
• Physiological: Vagal tone elevation in athletes or during sleep
Symptomatic bradycardia requires intervention (atropine, pacing) when cardiac output falls below metabolic demands—demonstrating its clinical significance as a rhythm disturbance requiring recognition and management.
C) fibrillation
Fibrillation describes rapid, disorganized electrical activity causing ineffective quivering rather than coordinated contraction—atrial fibrillation (400–600 bpm atrial rate) or ventricular fibrillation (chaotic ventricular activity causing cardiac arrest). Fibrillation involves rapid rates with loss of mechanical function—not slow rates. Ventricular fibrillation is immediately life-threatening requiring defibrillation, while atrial fibrillation increases stroke risk but maintains some cardiac output.
D) myocardial infarct
Myocardial infarction (heart attack) refers to cardiac muscle necrosis from prolonged coronary ischemia—potentially causing bradycardia if the inferior wall (supplied by right coronary artery) is affected and vagal stimulation occurs, but infarction itself is tissue death, not a rhythm description. Infarction may produce various arrhythmias (bradycardia, tachycardia, fibrillation) depending on location and extent—making it a pathological condition rather than a specific rhythm term.
Conclusion:
Bradycardia specifically denotes abnormally slow heart rates—distinguished from rapid rhythms (tachycardia, fibrillation) and structural damage (infarction). Option B provides the precise terminology matching the definition, essential for accurate clinical communication where rhythm identification guides emergency interventions (e.g., pacing for symptomatic bradycardia versus defibrillation for fibrillation). Mislabeling rhythms could lead to dangerous treatment errors—administering beta-blockers for presumed tachycardia when bradycardia is actually present would exacerbate hemodynamic compromise.
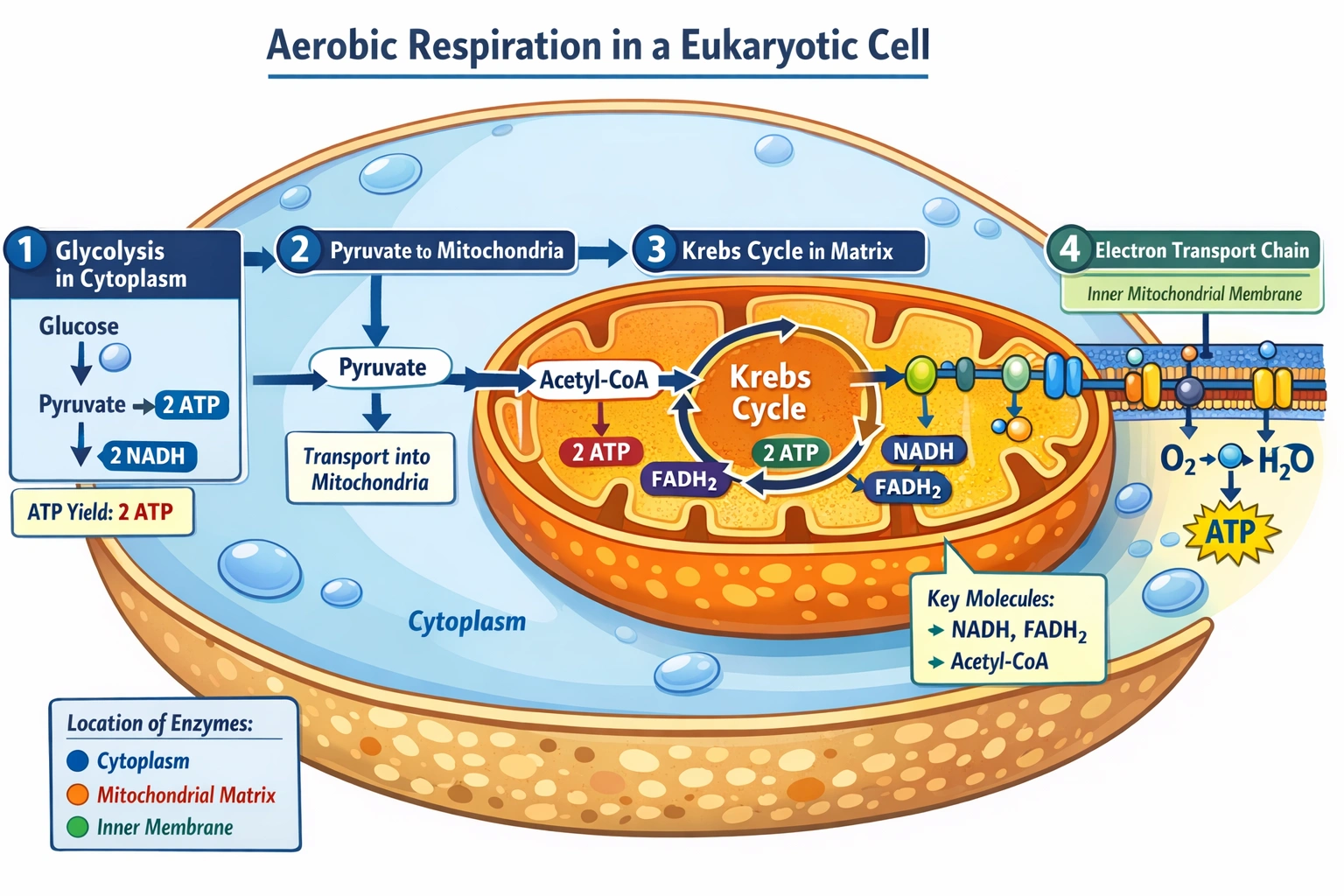
Aerobic respiration in cells occurs in the
A.
cytoplasm
B. nucleus
C. mitochondria
D. cell membrane
Rationale
Aerobic respiration occurs in the mitochondria. Mitochondria serve as the primary sites for aerobic ATP production through the Krebs cycle (in the matrix) and oxidative phosphorylation (across the inner membrane)—utilizing oxygen as the final electron acceptor to generate approximately 30–32 ATP molecules per glucose molecule, vastly exceeding anaerobic yield.
A) cytoplasm
The cytoplasm hosts glycolysis—the anaerobic breakdown of glucose to pyruvate yielding 2 ATP net—but cannot perform aerobic respiration lacking oxygen-dependent enzymes and membrane systems. Pyruvate produced in cytoplasm must enter mitochondria for aerobic processing; without mitochondrial uptake, pyruvate converts to lactate (anaerobic metabolism) even in oxygen presence if mitochondrial function is impaired. Cytoplasmic metabolism alone cannot utilize oxygen for ATP synthesis.
B) nucleus
The nucleus contains genetic material and directs protein synthesis but lacks respiratory enzymes or membrane systems for energy production. Nuclear processes consume ATP rather than generating it—transcription, DNA replication, and repair all require energy input from cytoplasmic/mitochondrial sources. No components of the electron transport chain or Krebs cycle enzymes localize to the nucleus—making it metabolically incapable of respiration.
C) mitochondria
Mitochondria execute aerobic respiration through compartmentalized processes:
• Pyruvate dehydrogenase complex (matrix): Converts pyruvate to acetyl-CoA
• Krebs cycle (matrix): Oxidizes acetyl-CoA producing NADH, FADH₂, and GTP
• Electron transport chain (inner membrane): Transfers electrons through complexes I–IV, pumping protons into intermembrane space
• ATP synthase (inner membrane): Uses proton gradient to phosphorylate ADP → ATP
This organization enables chemiosmotic coupling—oxygen accepts electrons at complex IV forming water, while the proton gradient drives ATP synthesis. Mitochondrial cristae increase surface area for maximal ATP production—approximately 90% of cellular ATP derives from this organelle in aerobic tissues.
D) cell membrane
The cell membrane regulates transport and signaling but lacks respiratory enzyme complexes. Prokaryotes perform respiration across plasma membranes lacking mitochondria, but eukaryotic cells evolved mitochondria specifically to compartmentalize aerobic metabolism—separating it from cytoplasmic processes. Eukaryotic plasma membranes contain no electron transport chain components; attempting respiration here would expose the cell to damaging reactive oxygen species without protective mitochondrial antioxidant systems.
Conclusion:
Mitochondria uniquely provide the structural and enzymatic machinery for efficient aerobic ATP production—compartmentalizing oxygen-dependent reactions to maximize yield while minimizing oxidative damage. Option C correctly identifies this organelle, distinguishing eukaryotic energy metabolism from prokaryotic systems. Understanding mitochondrial respiration proves essential clinically: mitochondrial disorders cause multi-system diseases (MELAS, Leigh syndrome), cyanide poisoning inhibits complex IV causing histotoxic hypoxia, and cancer cells often shift to anaerobic metabolism (Warburg effect)—all highlighting mitochondria's central role in cellular energetics and pathology.
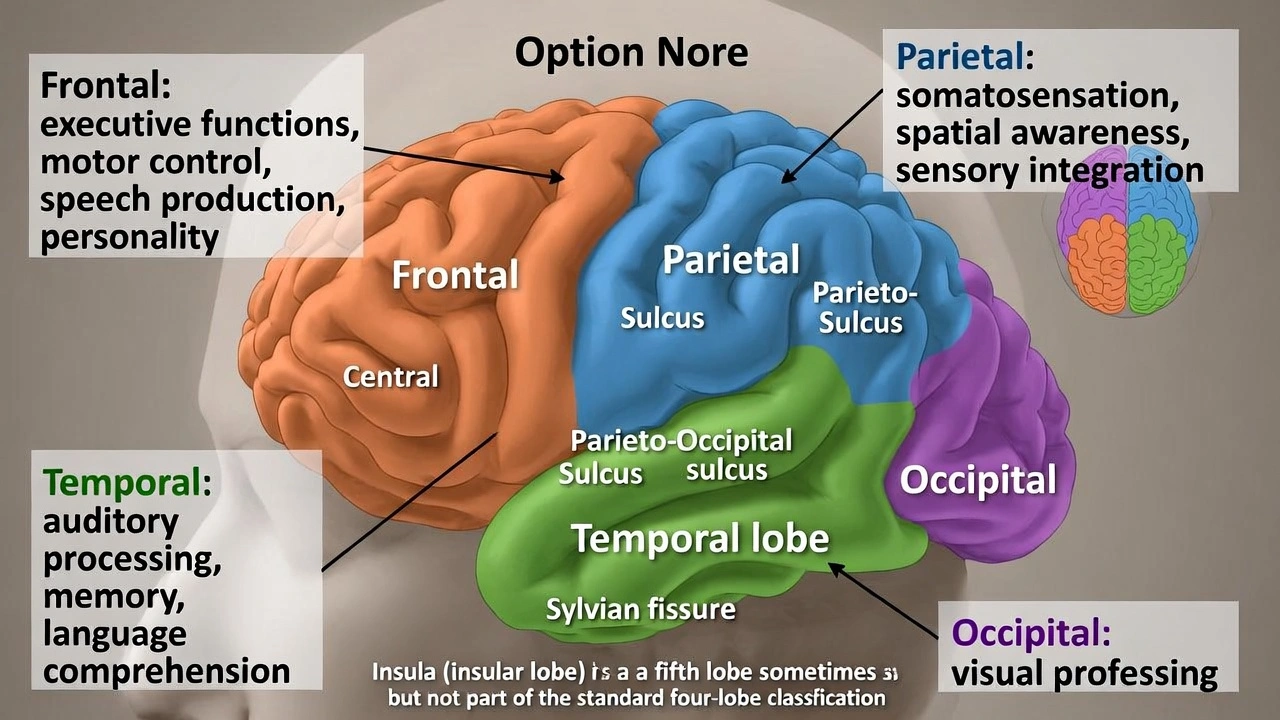
The brain is made of ___ lobes.
A.
2
B. 3
C. 4
D. 5
Rationale
The brain contains four lobes per cerebral hemisphere. Each cerebral hemisphere is divided into four anatomically and functionally distinct lobes—frontal, parietal, temporal, and occipital—separated by prominent sulci and fissures that organize specialized neural processing regions within the cerebral cortex.
A) 2
Two-lobed division (anterior/posterior) represents an oversimplified historical model abandoned with advances in neuroanatomy. Modern neuroimaging and functional mapping confirm four distinct lobes with specialized roles; no contemporary anatomical classification recognizes only two lobes.
B) 3
Three-lobed models occasionally appear in simplified educational contexts but lack anatomical accuracy. The Sylvian fissure clearly separates temporal lobe from frontal/parietal lobes, and the parieto-occipital sulcus demarcates occipital lobe—creating four distinct regions visible on gross examination and confirmed by cytoarchitectonic studies (Brodmann areas).
C) 4
The four lobes per hemisphere are:
• Frontal lobe: Anterior to central sulcus; governs executive functions, motor control, speech production (Broca's area), and personality
• Parietal lobe: Between central sulcus and parieto-occipital sulcus; processes somatosensation, spatial awareness, and integrates sensory modalities
• Temporal lobe: Inferior to Sylvian fissure; handles auditory processing, memory formation (hippocampus), and language comprehension (Wernicke's area)
• Occipital lobe: Posterior pole; dedicated to visual processing
This quadripartite organization is universal in human neuroanatomy, visible in standard brain atlases and essential for clinical localization of neurological deficits.
D) 5
Five lobes sometimes referenced include the insular lobe (deep to Sylvian fissure) as a fifth lobe—but the insula is typically considered part of the cerebral cortex rather than a primary lobe in standard four-lobe classification. Limbic lobe references also exist but represent functional rather than anatomical divisions. Standard anatomical teaching recognizes four primary lobes per hemisphere.
Conclusion:
Four lobes per cerebral hemisphere represent the universally accepted anatomical division in human neuroanatomy—critical for clinical neurology where lesion localization depends on lobar organization (e.g., occipital stroke causing visual field defects, frontal tumor producing personality changes). Option C provides the standard anatomical count essential for medical education and neurological diagnosis.
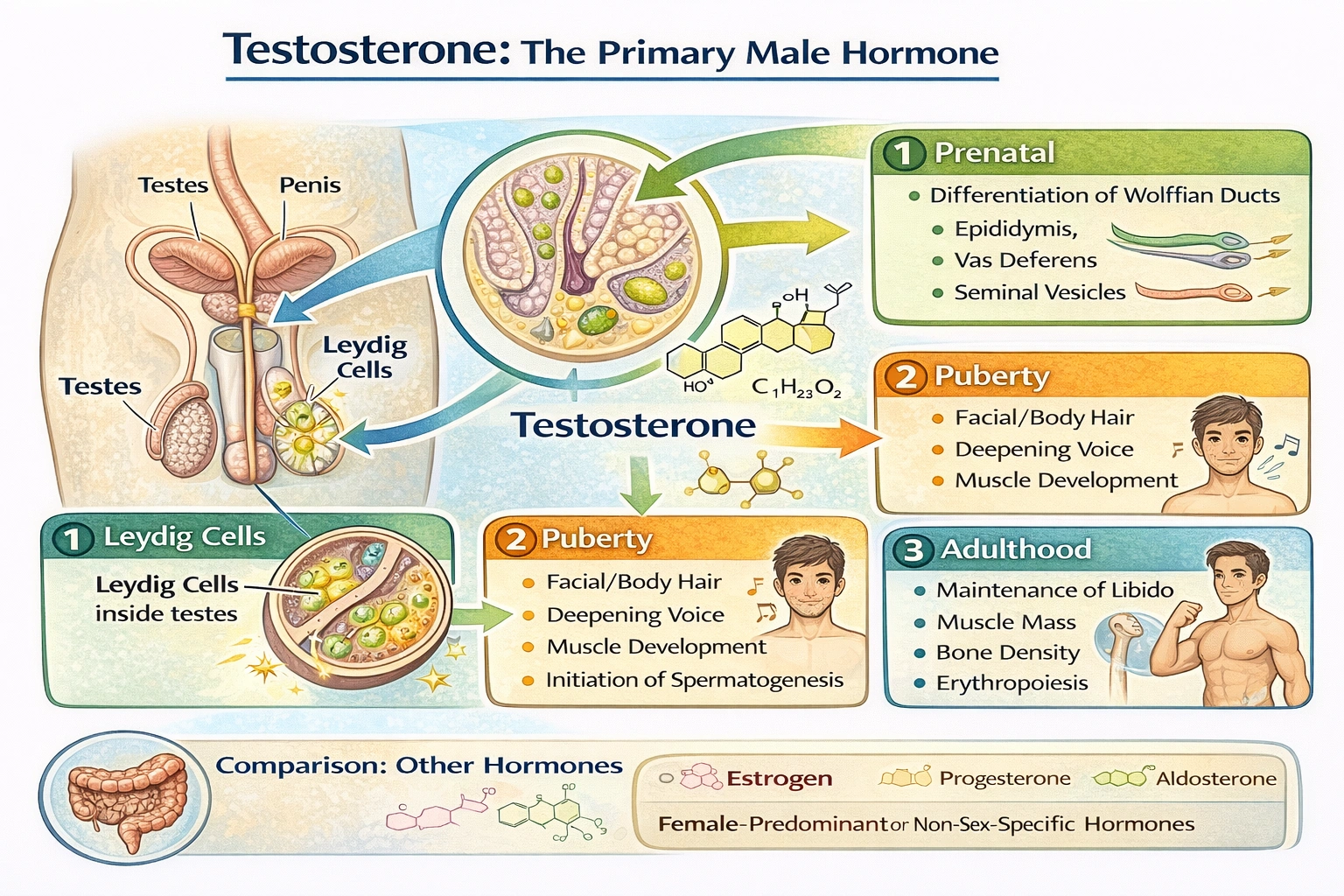
___ is the most important male hormone.
A.
estrogen
B. aldosterone
C. progesterone
D. testosterone
Rationale
Testosterone is the primary male hormone. Testosterone, an androgen produced primarily by Leydig cells in testes (and minimally by adrenal cortex), drives male sexual development, spermatogenesis, secondary sexual characteristics, muscle mass maintenance, and libido—serving as the principal hormonal regulator of male physiology throughout life.
A) estrogen
Estrogen represents the primary female sex hormone (though present in males at low levels), produced mainly by ovaries. In males, estrogen derives from testosterone aromatization and regulates bone density and libido—but doesn't drive male development or function. Administering estrogen to males causes feminization (gynecomastia, reduced body hair), demonstrating its non-masculinizing role.
B) aldosterone
Aldosterone is a mineralocorticoid hormone regulating sodium/potassium balance and blood pressure—produced by adrenal cortex in both sexes. While essential for electrolyte homeostasis, it lacks sex-specific functions and doesn't influence male development, reproduction, or secondary sexual characteristics.
C) progesterone
Progesterone functions primarily in female reproductive physiology—preparing endometrium for implantation and maintaining pregnancy. Males produce minimal progesterone as a testosterone precursor, but it plays no significant role in male development or function. Elevated progesterone in males may suppress gonadotropins and reduce testosterone production.
D) testosterone
Testosterone orchestrates male development through:
• Prenatal: Differentiation of Wolffian ducts into epididymis, vas deferens, seminal vesicles
• Puberty: Deepening voice, facial/body hair growth, muscle development, spermatogenesis initiation
• Adulthood: Maintenance of libido, muscle mass, bone density, and erythropoiesis
Normal male serum levels range 300–1000 ng/dL; deficiency causes hypogonadism (fatigue, reduced muscle mass, infertility), while exogenous administration treats these conditions—demonstrating testosterone's central role in male physiology.
Conclusion:
Testosterone uniquely serves as the principal androgen governing male sexual differentiation, reproductive function, and secondary sexual characteristics—distinguishing it from female-predominant hormones (A, C) or non-sex-specific regulators (B). Option D correctly identifies this hormone essential for understanding male endocrinology, treating hypogonadism, and managing conditions like prostate cancer where androgen signaling drives pathology.
Auditory impulses are interpreted in the ___ lobes.
A.
frontal
B. parietal
C. temporal
D. occipital
Rationale
Auditory impulses are interpreted in the temporal lobes. The temporal lobes contain the primary auditory cortex (Brodmann areas 41–42) within Heschl's gyri on the superior temporal plane, where sound frequency, intensity, and location are processed—enabling speech comprehension in Wernicke's area (posterior superior temporal gyrus) and complex sound recognition.
A) frontal
Frontal lobes govern executive functions (planning, decision-making), voluntary motor control (primary motor cortex), speech production (Broca's area in dominant hemisphere), and personality regulation. While frontal regions participate in attention to auditory stimuli and speech initiation, they don't perform primary sound interpretation—damage causes expressive aphasia or executive dysfunction without deafness.
B) parietal
Parietal lobes process somatosensory information (primary somatosensory cortex), spatial orientation, and sensory integration (e.g., combining touch with vision). The angular gyrus contributes to reading and number processing, but parietal regions don't interpret auditory signals—damage causes neglect syndromes or agraphesthesia without auditory deficits.
C) temporal
Temporal lobe auditory processing follows a hierarchical pathway:
• Primary auditory cortex: Receives input from medial geniculate nucleus via auditory radiations; analyzes basic sound features (frequency, intensity)
• Auditory association cortex: Surrounds primary area; integrates sound patterns into meaningful units (words, melodies)
• Wernicke's area: Dominant hemisphere posterior superior temporal gyrus; comprehends spoken language
• Non-dominant temporal lobe: Processes musical pitch, environmental sounds, and emotional tone
Unilateral temporal lobe damage causes contralateral hearing deficits and receptive aphasia (if dominant hemisphere affected); bilateral damage produces cortical deafness despite intact peripheral hearing.
D) occipital
Occipital lobes exclusively process visual information—primary visual cortex (V1) in calcarine fissure receives input from lateral geniculate nucleus, with surrounding areas analyzing motion, color, and form. Occipital damage causes visual field defects or cortical blindness but preserves all auditory function—demonstrating strict modality segregation in cortical processing.
Conclusion:
Temporal lobes serve as the dedicated cortical region for auditory processing—from basic sound detection to complex language comprehension. Option C correctly identifies this localization, essential for neurological diagnosis: temporal lobe lesions produce specific auditory deficits (word deafness, music agnosia) distinct from frontal (expressive language problems), parietal (sensory integration issues), or occipital (visual deficits) lesions. Understanding this functional anatomy guides localization of stroke, tumor, or epileptic foci based on symptom profiles.

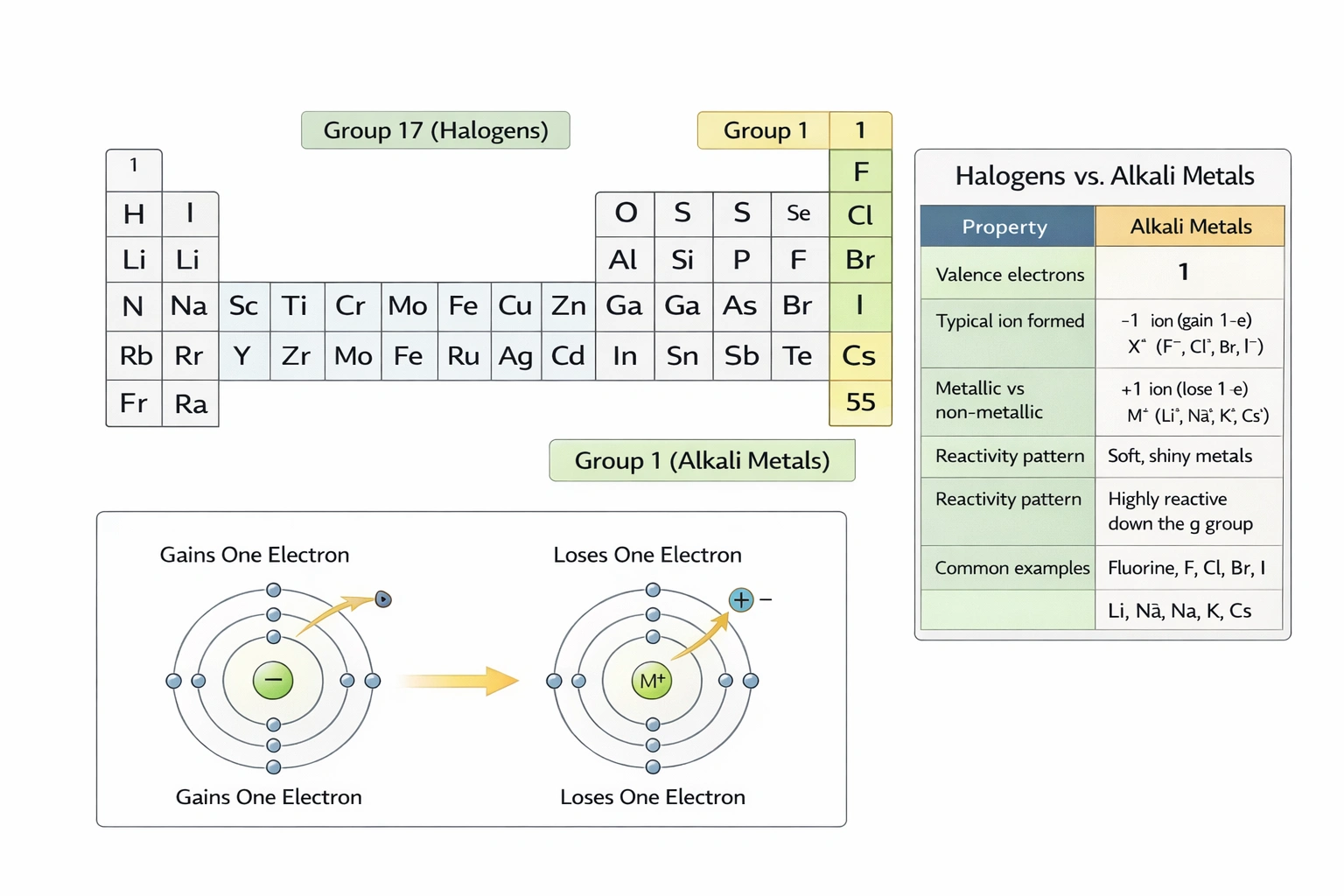
Which of the following elements are not halogens?
A.
Chlorine
B. Bromine
C. Iodine
D. Cesium
Rationale
Cesium is not a halogen. Halogens constitute group 17 (VIIA) of the periodic table—fluorine, chlorine, bromine, iodine, and astatine—characterized by seven valence electrons and high reactivity seeking one additional electron to achieve stable octet configuration, whereas cesium belongs to group 1 (alkali metals) with one valence electron.
A) Chlorine
Chlorine (Cl, atomic number 17) is a classic halogen—greenish-yellow gas at room temperature forming diatomic molecules (Clâ‚‚). It readily gains one electron to form chloride ions (Clâ»), participating in ionic compounds (NaCl) and covalent bonds (HCl). Chlorine's high electronegativity (3.16) and electron affinity exemplify halogen reactivity—used in water purification, PVC production, and bleach manufacturing.
B) Bromine
Bromine (Br, atomic number 35) is the only liquid halogen at room temperature—reddish-brown volatile liquid forming Brâ‚‚ molecules. Like other halogens, it gains one electron to form bromide ions (Brâ»), appearing in sedatives (potassium bromide), flame retardants, and photographic emulsions. Its intermediate reactivity between chlorine and iodine follows halogen group trends.
C) Iodine
Iodine (I, atomic number 53) is a dark purple solid halogen subliming to violet vapor—forming Iâ‚‚ molecules. It gains one electron to form iodide ions (Iâ»), essential for thyroid hormone synthesis (T3/T4). Iodine's lower reactivity compared to lighter halogens follows periodic trends—used in antiseptics, contrast media, and iodized salt to prevent deficiency disorders.
D) Cesium
Cesium (Cs, atomic number 55) is an alkali metal in group 1—not a halogen. It possesses one valence electron that it readily donates to form Cs⺠cations—opposite to halogen electron-accepting behavior. Cesium is soft, silvery-gold metal with extremely low ionization energy (376 kJ/mol), reacting explosively with water and air. Its chemistry centers on +1 oxidation state compounds (CsCl), contrasting with halogens' -1 state dominance. Cesium's position in period 6, group 1 places it far from halogen group 17 both chemically and physically.
Conclusion:
Halogens share defining characteristics: group 17 placement, seven valence electrons, diatomic molecular forms, high electronegativity, and tendency to form -1 anions—properties cesium fundamentally lacks as a group 1 alkali metal that readily loses electrons. Option D correctly identifies the non-halogen element, essential for understanding periodic table organization and predicting chemical behavior: halogens form salts with alkali metals (NaCl, KBr, CsI), but cesium itself belongs to the metal family that reacts with halogens rather than being one.
Compounds with various structures but the same molecular formula are called ___
A.
polar compounds
B. isomers
C. variables
D. transient compounds
Rationale
Isomers share the same molecular formula but have different structures. Isomers represent distinct compounds with identical atom counts and types but differing arrangements—creating unique physical properties, chemical reactivities, and biological activities despite identical molecular formulas, classified into structural isomers (different connectivity) and stereoisomers (different spatial orientation).
A) polar compounds
Polarity describes uneven electron distribution creating molecular dipoles—dependent on bond electronegativity differences and molecular geometry. Polar compounds may or may not be isomers; polarity is an electronic property unrelated to structural variation among same-formula compounds. Two isomers can differ in polarity (e.g., cis/trans dichloroethene), but polarity itself doesn't define isomerism.
B) isomers
Isomer classification includes:
• Structural isomers: Different atom connectivity (e.g., n-butane vs. isobutane, both Câ‚„Hâ‚â‚€)
• Stereoisomers: Identical connectivity but different spatial arrangement
o Geometric isomers: Restricted rotation (cis/trans alkenes, e.g., cis-2-butene vs. trans-2-butene)
o Optical isomers: Mirror-image chirality (enantiomers like D/L glucose)
Isomerism profoundly impacts biological function: thalidomide enantiomers differ in teratogenicity; glucose/fructose (structural isomers, both C₆Hâ‚â‚‚O₆) have distinct metabolic pathways—demonstrating how identical formulas yield functionally distinct compounds.
C) variables
"Variables" is a mathematical/statistical term describing quantities that change—having no application in chemical nomenclature for compound classification. No chemical concept uses "variables" to describe structural variations among compounds, making this option terminologically inappropriate.
D) transient compounds
Transient compounds refer to short-lived reaction intermediates (carbocations, radicals) that exist fleetingly during chemical transformations—not stable compounds with defined structures. Isomers are typically stable, isolable compounds—not transient species. Transience describes kinetic instability rather than structural variation among same-formula compounds.
Conclusion:
Isomerism represents a fundamental chemical concept where identical molecular formulas yield distinct compounds through structural or spatial variations—critical for understanding drug action, metabolic pathways, and material properties. Option B correctly identifies isomers as the precise term for this phenomenon, essential for pharmaceutical development (where isomer purity affects drug safety) and analytical chemistry (requiring separation techniques like chiral chromatography). Confusing isomerism with polarity or other properties would impair prediction of compound behavior despite identical composition.
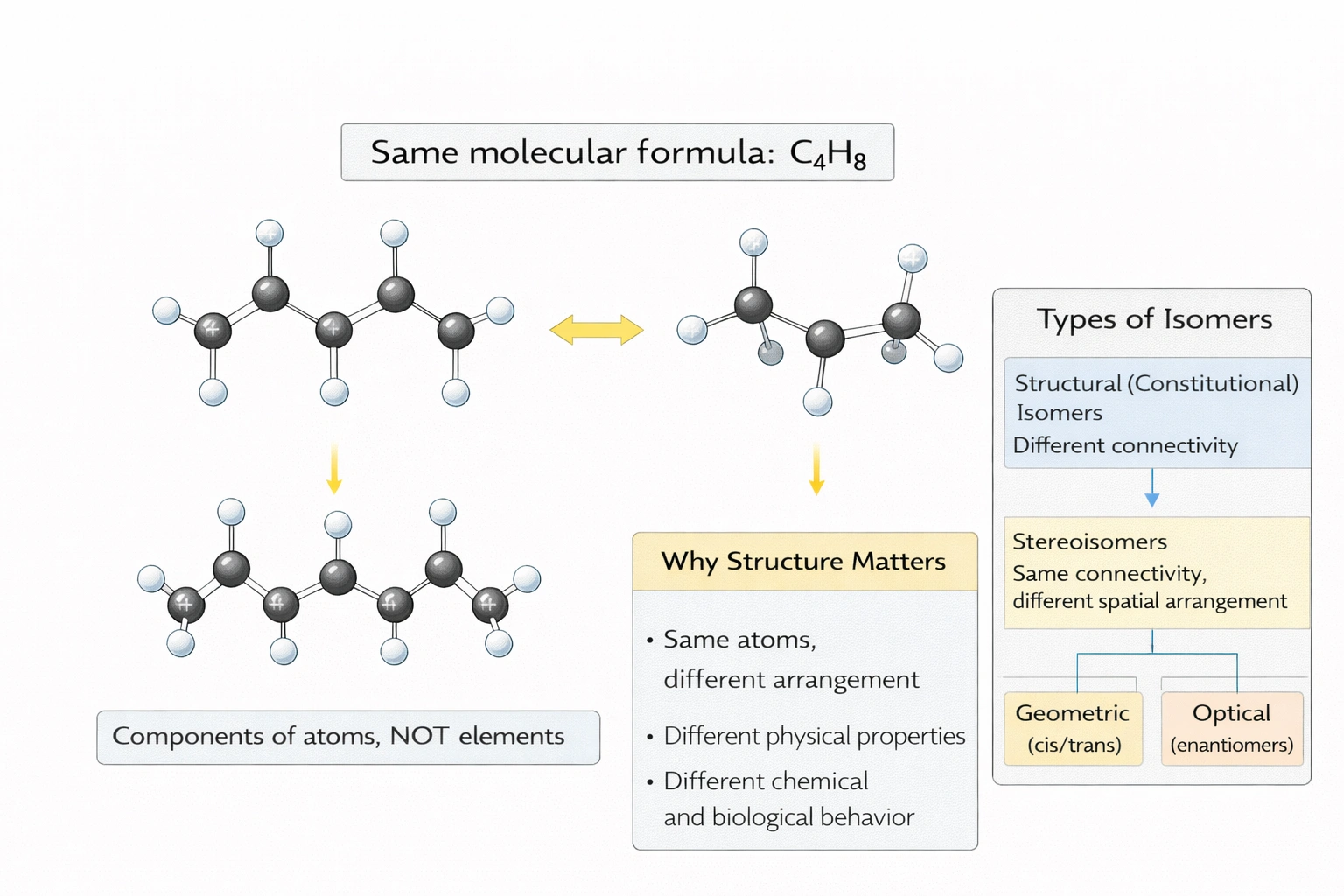
Which of the following terms matches the definition: energy is conserved with every process.
A.
1st Law of Thermodynamics
B. 2nd Law of Thermodynamics
C. 3rd Law of Thermodynamics
D. 4th Law of Thermodynamics
Rationale
The 1st Law of Thermodynamics states that energy is conserved. The first law establishes that energy cannot be created or destroyed in isolated systems—only transformed between forms (kinetic, potential, thermal, chemical) or transferred as work or heat—with the total energy of the universe remaining constant, expressed mathematically as ΔU = Q - W where U is internal energy, Q is heat added, and W is work done by the system.
A) 1st Law of Thermodynamics
The first law formalizes energy conservation:
• Isolated systems: Total energy constant (ΔU = 0)
• Closed systems: Energy change equals heat transfer minus work output (ΔU = Q - W)
• Open systems: Includes mass flow energy terms
Examples: In cellular respiration, chemical energy in glucose converts to ATP (chemical), heat, and mechanical work—total energy conserved. Perpetual motion machines of the first kind (creating energy) violate this law and are impossible—demonstrating its universal applicability across physical, chemical, and biological processes.
B) 2nd Law of Thermodynamics
The second law addresses entropy (disorder) increase in spontaneous processes—not energy conservation. It states that isolated systems evolve toward maximum entropy (ΔS_universe > 0 for spontaneous processes), explaining why heat flows hot→cold spontaneously but not reverse without work input. While energy is conserved (first law), its quality degrades toward less usable forms (second law)—two distinct principles.
C) 3rd Law of Thermodynamics
The third law states that entropy of a perfect crystal approaches zero as temperature approaches absolute zero—providing an absolute entropy reference point. It doesn't address energy conservation but enables calculation of absolute entropy values for thermodynamic tables—critical for predicting reaction spontaneity but unrelated to energy preservation.
D) 4th Law of Thermodynamics
No scientifically recognized "fourth law" exists in thermodynamics—this option represents a distractor. Some non-standard proposals (e.g., maximum entropy production principle) exist in specialized literature but lack universal acceptance as fundamental thermodynamic laws. Standard thermodynamics recognizes only three laws.
Conclusion:
The first law exclusively embodies energy conservation—fundamental to all physical sciences and engineering. Option A correctly identifies this principle, distinguishing it from entropy concepts (B, C) or non-existent laws (D). Understanding this law prevents impossible design attempts (perpetual motion machines) and enables energy accounting in complex systems—from metabolic pathways to power plant efficiency calculations—where tracking energy transformations while recognizing total conservation is essential for accurate analysis.
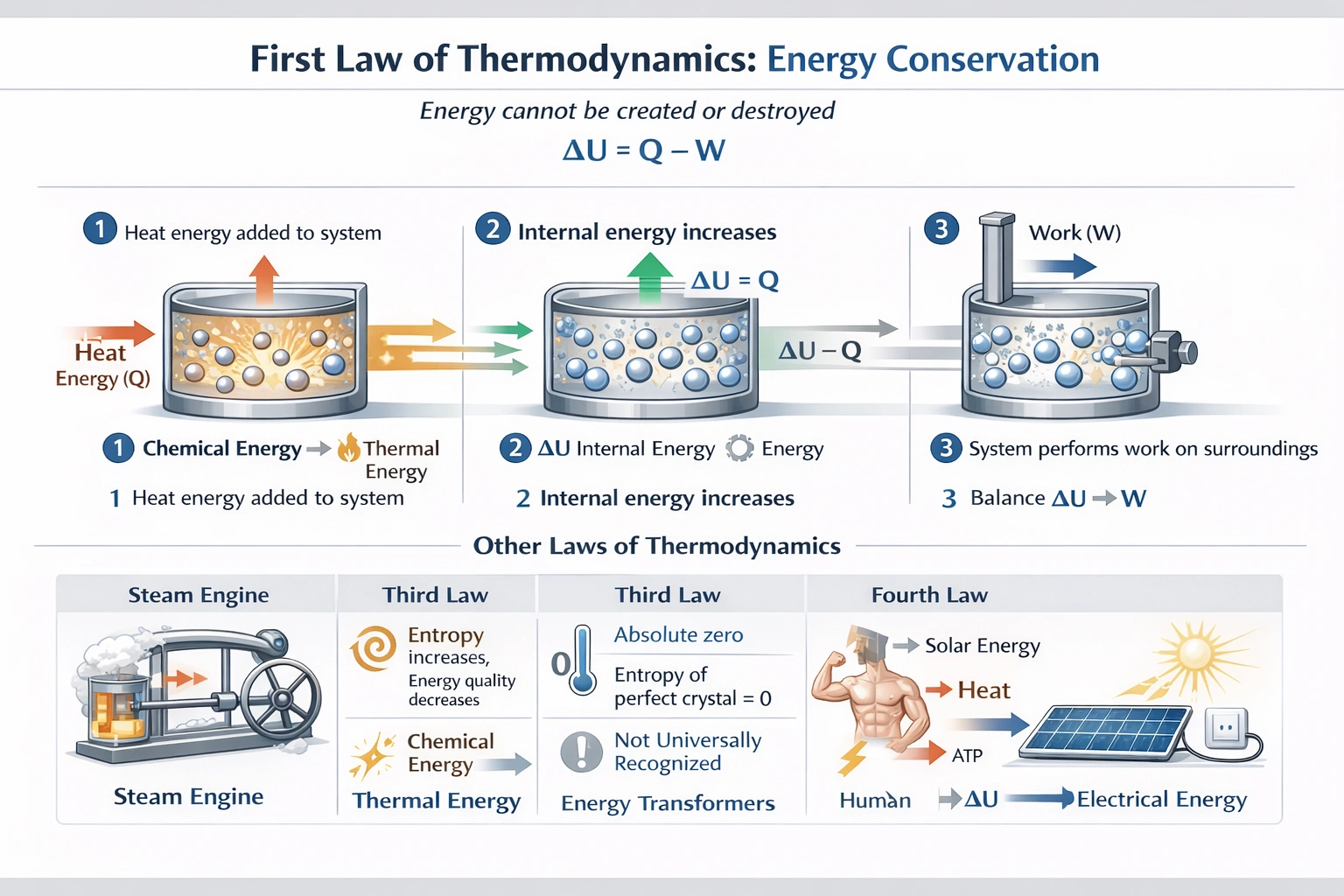
Using the 2nd Law of Newton identify the formula that is applicable.
A.
F = ma
B. Speed = Distance/Time
C. Power = F × D
D. Watts = Voltage × Amperes
Rationale
Newton's second law is expressed as F = ma. Newton's second law of motion states that the net force acting on an object equals its mass multiplied by its acceleration—quantifying how forces cause changes in motion with directionality (vector relationship) where acceleration occurs in the force's direction.
A) F = ma
This equation defines Newton's second law precisely:
• F: Net force vector (newtons, N)
• m: Mass scalar (kilograms, kg)
• a: Acceleration vector (m/s²)
The law establishes causality: forces produce acceleration proportional to magnitude and inversely proportional to mass (a = F/m). Examples: 10 N force on 2 kg mass produces 5 m/s² acceleration; same force on 5 kg mass yields 2 m/s²—demonstrating mass as inertia measure resisting acceleration. This vector equation underpins classical mechanics, engineering dynamics, and biomechanics analysis.
B) Speed = Distance/Time
This defines average speed—a kinematic relationship describing motion magnitude without direction. It derives from motion definitions rather than force dynamics, representing descriptive kinematics rather than causal force-motion relationships central to Newton's laws. While useful for motion description, it doesn't express force-acceleration causality.
C) Power = F × D
This misstates the power formula—correct form is Power = Work/Time = (Force × Distance)/Time. Power measures energy transfer rate (watts = joules/second), not direct force-distance product. While related to mechanics, this represents energy concepts rather than Newton's force-acceleration principle—confusing work/power with fundamental motion laws.
D) Watts = Voltage × Amperes
This expresses electrical power (P = VI)—relating to electromagnetism rather than mechanical motion laws. While watts measure power universally, this equation belongs to electrical engineering physics, not Newtonian mechanics governing force and motion relationships. Newton's laws address mechanical systems, not electromagnetic phenomena.
Conclusion:
F = ma exclusively represents Newton's second law—establishing the foundational relationship between force, mass, and acceleration that enables prediction of motion under applied forces. Option A provides the precise mathematical expression essential for physics problem-solving: calculating required forces for desired accelerations (rocket propulsion), determining braking distances (automotive safety), or analyzing biomechanical stresses (sports medicine). Confusing this with kinematic definitions (B), energy concepts (C), or electrical relationships (D) would impair application of Newtonian mechanics to real-world motion problems.
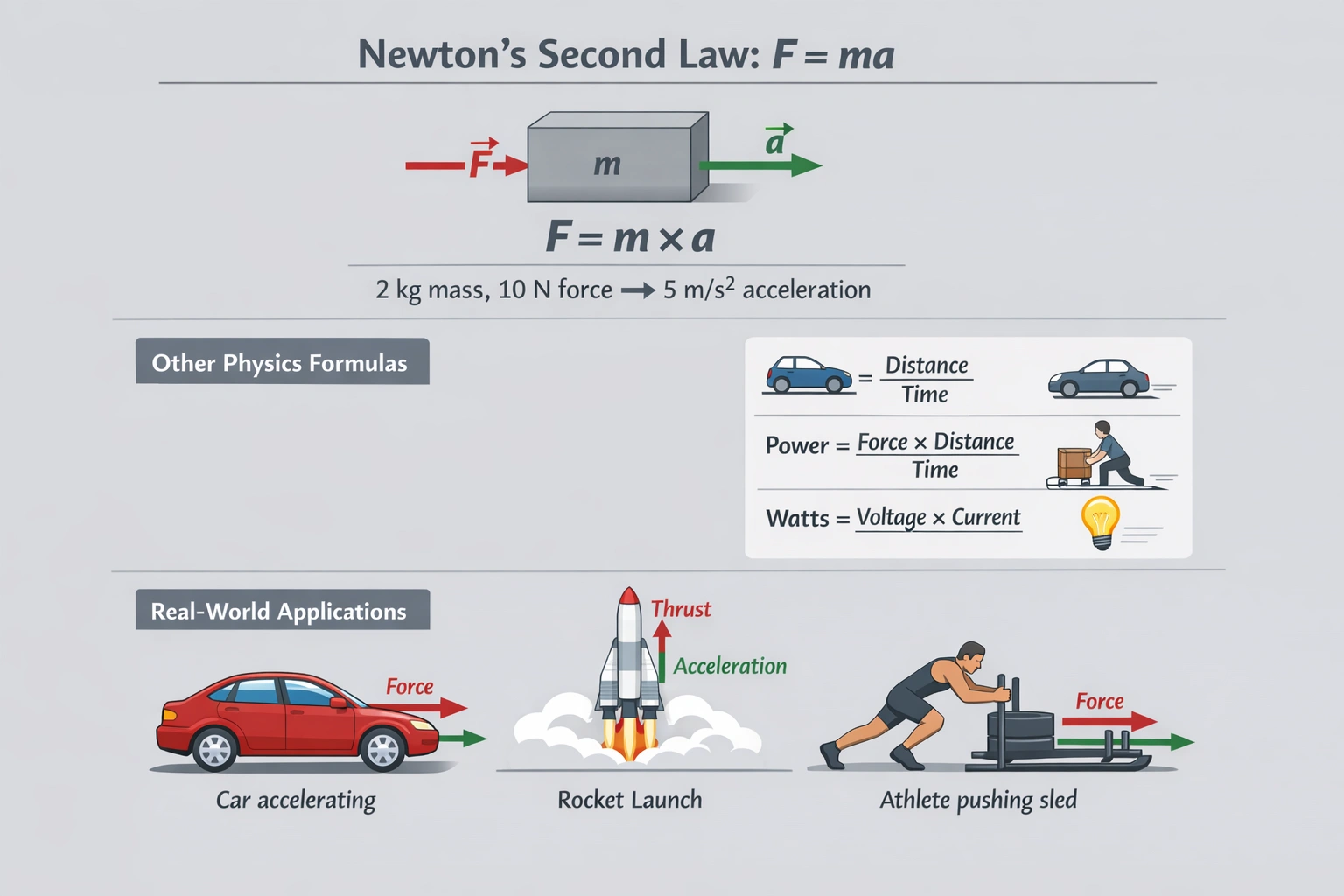
The chart above shows the average snowfall in inches for a town on Michigans Upper Peninsula during the months November through April. Which of the following can be concluded based on the information provided in the chart?
A.
April is not a good month to go skiing in the Upper Peninsula
B. Snowfall blocks the sunshine and reduces the number of sunny days
C. The fewest sunny days occur in the months with the heaviest snowfall
D. There is no connection between the amount of snowfall and the number of sunny days
Rationale
The fewest sunny days occur in the months with the heaviest snowfall. The graph shows an inverse relationship between snowfall and sunny days with the highest snowfall occurring in December and January while the lowest number of sunny days coincides with these same months demonstrating that maximum snowfall correlates with minimum sunny days.
A) April is not a good month to go skiing in the Upper Peninsula
While April shows minimal snowfall making it poor for skiing this conclusion is not directly supported by the data. The chart shows only snowfall and sunny days but does not address skiing conditions which depend on snow quality snowpack depth and other factors not measured in the graph. The conclusion makes an inference beyond the specific data presented.
B) Snowfall blocks the sunshine and reduces the number of sunny days
This statement implies causation which the data cannot prove. The graph shows correlation between high snowfall and low sunny days but does not establish that snowfall causes reduced sunshine. Snowfall and sunshine are both influenced by weather patterns but the graph cannot determine causal relationships. Only correlational analysis is possible from this data.
C) The fewest sunny days occur in the months with the heaviest snowfall
This conclusion directly matches the graph data:
• Highest snowfall occurs in December and January (peaking around 30 inches)
• Fewest sunny days occur in December and January (dropping to approximately 10 days)
• As snowfall decreases from January to April sunny days increase
This inverse relationship demonstrates that months with heaviest snowfall correspond to the fewest sunny days without implying causation.
D) There is no connection between the amount of snowfall and the number of sunny days
This conclusion contradicts the clear inverse relationship shown in the graph. The data demonstrates a strong negative correlation where higher snowfall corresponds to fewer sunny days. The lines move in opposite directions across all months indicating a consistent relationship.
Conclusion:
The graph clearly shows an inverse relationship between snowfall and sunny days with December and January having both the highest snowfall and lowest sunny days. Option C correctly identifies this direct observation from the data without making unsupported causal claims. The correlation is evident throughout the six-month period with no exceptions making it the only valid conclusion directly supported by the information presented.
The two criteria for classifying epithelial tissue are cell layers and ___
A.
cell composition
B. cell absorption
C. cell shape
D. cell stratification
Rationale
Cell shape completes the classification criteria for epithelial tissue. Epithelial tissues are systematically categorized using two structural characteristics: the number of cell layers (simple versus stratified) and the shape of the cells (squamous cuboidal or columnar) creating a precise naming system that reflects both architecture and function across different body locations.
A) cell composition
Cell composition refers to molecular or biochemical makeup rather than morphological features used in histological classification. All epithelial cells share similar basic composition with plasma membranes cytoplasm and nuclei differing primarily in shape arrangement and specialized surface modifications rather than fundamental compositional differences. Composition does not provide the visual criteria pathologists and histologists use to identify and name epithelial types under microscopy.
B) cell absorption
Absorption represents a functional capability of certain epithelia particularly in the digestive and renal systems but is not a structural classification criterion. Many epithelial types do not absorb substances: secretory epithelia produce materials protective epithelia resist abrasion and absorption capacity varies within cell shape categories making it unsuitable as a universal classification parameter. Histological classification relies on observable structure not physiological function.
C) cell shape
Cell shape provides the second essential classification dimension alongside layering:
• Squamous: Flat scale-like cells with horizontally flattened nuclei found in areas requiring diffusion or minimal friction (blood vessels alveoli)
• Cuboidal: Cube-shaped cells with spherical central nuclei involved in secretion and absorption (kidney tubules glandular ducts)
• Columnar: Tall rectangular cells with elongated nuclei often featuring microvilli or cilia for absorption or movement (intestinal lining respiratory tract)
Combined with layering (simple squamous simple cuboidal stratified squamous etc.) these two criteria generate the complete epithelial classification system used universally in histology.
D) cell stratification
Stratification describes the number of cell layers already identified as the first classification criterion in the question statement. Using stratification as both criteria would be redundant. Simple epithelia have one layer stratified have multiple layers and pseudostratified appear layered but actually consist of a single layer with nuclei at varying heights. Stratification cannot serve as the second independent criterion when it defines the first dimension.
Conclusion:
Epithelial classification requires two independent structural parameters: layering and cell shape to generate precise tissue identification. Simple squamous versus stratified squamous versus simple columnar each describes distinct tissues with different locations and functions. Option C correctly identifies cell shape as the complementary criterion to layering forming the foundation of histological epithelial taxonomy. This dual-parameter system enables accurate tissue identification essential for diagnosing diseases like carcinomas that retain epithelial shape characteristics even when malignant.

On average, how many protons does one atom of zinc (Zn) have?
A.
30
B. 35
C. 35.359
D. 65.39
Rationale
Zinc atoms always contain 30 protons. The atomic number of an element directly equals its proton count which is fixed for all atoms of that element regardless of isotope and is the defining characteristic of the element.
A) 30
Zinc's atomic number is 30 which means all zinc atoms contain exactly 30 protons. This value is fixed and unchanging for all zinc atoms regardless of isotope or atomic mass. The proton count defines the element's identity and chemical properties making it invariant across all zinc atoms.
B) 35
This option represents the atomic number of bromine not zinc. Zinc has atomic number 30 while bromine has atomic number 35. Confusing these values represents a fundamental misunderstanding of periodic table organization where each element has a unique atomic number corresponding to its proton count.
C) 35.359
This value has no basis in zinc's atomic structure. The proton count must be an integer as protons are discrete particles. This option appears to be a miscalculation or misreading of zinc's atomic mass (65.39) and is not a meaningful value for proton count.
D) 65.39
This option represents zinc's atomic mass not the proton count. The atomic mass reflects the weighted average of all zinc isotopes including protons and neutrons while the proton count remains fixed at 30 for all zinc atoms. The atomic mass varies slightly across isotopes but the proton count never changes.
Conclusion:
The number of protons in an atom equals its atomic number which is fixed for all atoms of a given element. Zinc has atomic number 30 meaning all zinc atoms contain exactly 30 protons regardless of isotope. Option A correctly identifies this invariant proton count essential for understanding elemental identity and atomic structure.

In the development of genetic traits, one gene must match to one ___ for the traits to develop correctly.
A.
Codon
B. Ribosome
C. Amino acid
D. Chromosome
Rationale
One codon matches to one amino acid during protein synthesis. The genetic code operates through codon-amino acid correspondence where each three-nucleotide codon in mRNA specifies a single amino acid to be incorporated into a growing polypeptide chain ensuring accurate translation of genetic information into functional proteins.
A) Codon
A codon is a sequence of three nucleotides in mRNA that specifies a particular amino acid during translation. The statement "one gene must match to one codon" is inaccurate because a single gene contains hundreds or thousands of codons not just one. Each gene codes for an entire protein sequence composed of many amino acids linked together making this option incorrect for completing the sentence.
B) Ribosome
Ribosomes are cellular structures composed of rRNA and proteins that serve as the site of protein synthesis. They facilitate the matching between codons and amino acids but do not represent the matching unit themselves. One ribosome translates many codons during a single protein synthesis event and multiple ribosomes can translate the same mRNA simultaneously making ribosomes machinery rather than matching units.
C) Amino acid
The statement "one gene must match to one amino acid" is scientifically inaccurate because a single gene typically codes for a polypeptide containing dozens to hundreds of amino acids not just one. The classic "one gene-one enzyme" hypothesis was refined to recognize that genes code for entire polypeptide chains not individual amino acids. One codon matches to one amino acid but one gene matches to many amino acids arranged in a specific sequence.
D) Chromosome
Chromosomes are structures containing hundreds to thousands of genes along with regulatory elements and non-coding DNA. One chromosome contains many genes not a one-to-one relationship with individual genes. Chromosomes represent the packaging level above genes not the matching unit for trait development.
Conclusion:
The question appears to contain imprecise wording but based on standard genetic principles the fundamental matching relationship in protein synthesis is between one codon and one amino acid. Option A represents the structural unit (codon) that participates in this matching relationship though the phrasing "one gene must match to one codon" remains scientifically inaccurate. The genetic code's triplet nature where three nucleotides specify one amino acid forms the basis of accurate protein synthesis essential for proper trait development.
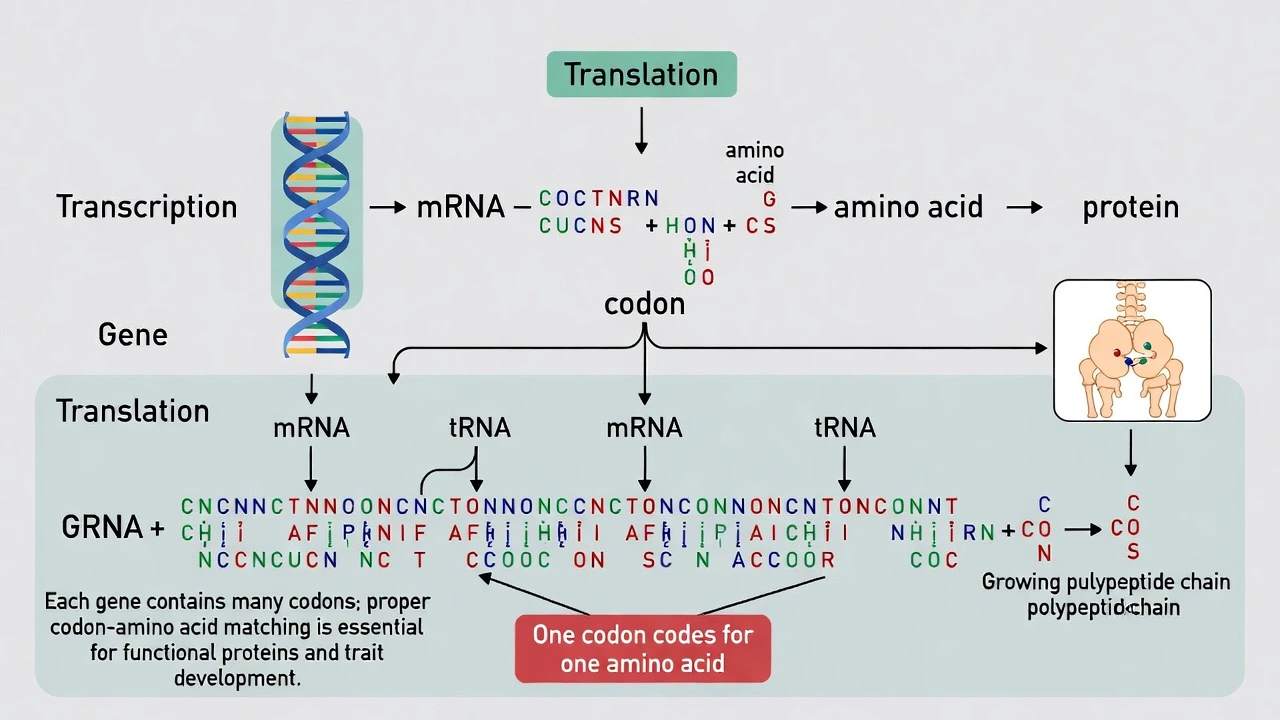
During the anaphase of mitosis, the ___, originally in pairs, separate from their daughters and move to the opposite ends (or poles) of the cell.
A.
Chromosomes
B. Spindle fibers
C. Centrioles
D. Nuclear membranes
Rationale
Chromosomes separate during anaphase of mitosis. During anaphase sister chromatids which were previously joined at the centromere separate and are pulled toward opposite spindle poles by shortening kinetochore microtubules transforming each chromatid into an individual daughter chromosome.
A) Chromosomes
At the start of anaphase each chromosome consists of two identical sister chromatids joined at the centromere. When the anaphase-promoting complex triggers separase activation cohesin proteins binding the chromatids degrade allowing them to separate. Each separated chromatid is now considered an individual chromosome. These daughter chromosomes move toward opposite poles pulled by depolymerizing microtubules attached to their kinetochores completing the equal distribution of genetic material.
B) Spindle fibers
Spindle fibers (microtubules) do not separate during anaphase but rather facilitate chromosome movement. Kinetochore microtubules shorten pulling chromosomes toward poles while polar microtubules lengthen pushing poles apart. The spindle apparatus remains intact throughout anaphase disassembling only during telophase. Spindle fibers serve as the machinery for separation not the entities being separated.
C) Centrioles
Centrioles duplicate during interphase and migrate to opposite poles during prophase where they organize spindle formation. They remain at the poles throughout mitosis without separating further. Centrioles anchor the spindle poles but do not themselves separate into daughter components during anaphase.
D) Nuclear membranes
Nuclear membranes break down during prophase before chromosomes align and begin reassembling during telophase after chromosomes reach the poles. No nuclear membrane exists during anaphase to undergo separation. The nuclear envelope must disassemble to allow spindle access to chromosomes and reassembles only after chromosome segregation completes.
Conclusion:
Anaphase is defined by the separation of sister chromatids (now called daughter chromosomes) which move to opposite cell poles ensuring each future daughter cell receives an identical set of genetic material. Option A correctly identifies chromosomes as the structures undergoing separation distinguishing them from the spindle machinery that moves them centrioles that anchor poles or nuclear membranes that disassemble earlier in mitosis. This precise chromosome segregation mechanism maintains genetic fidelity across cell generations.
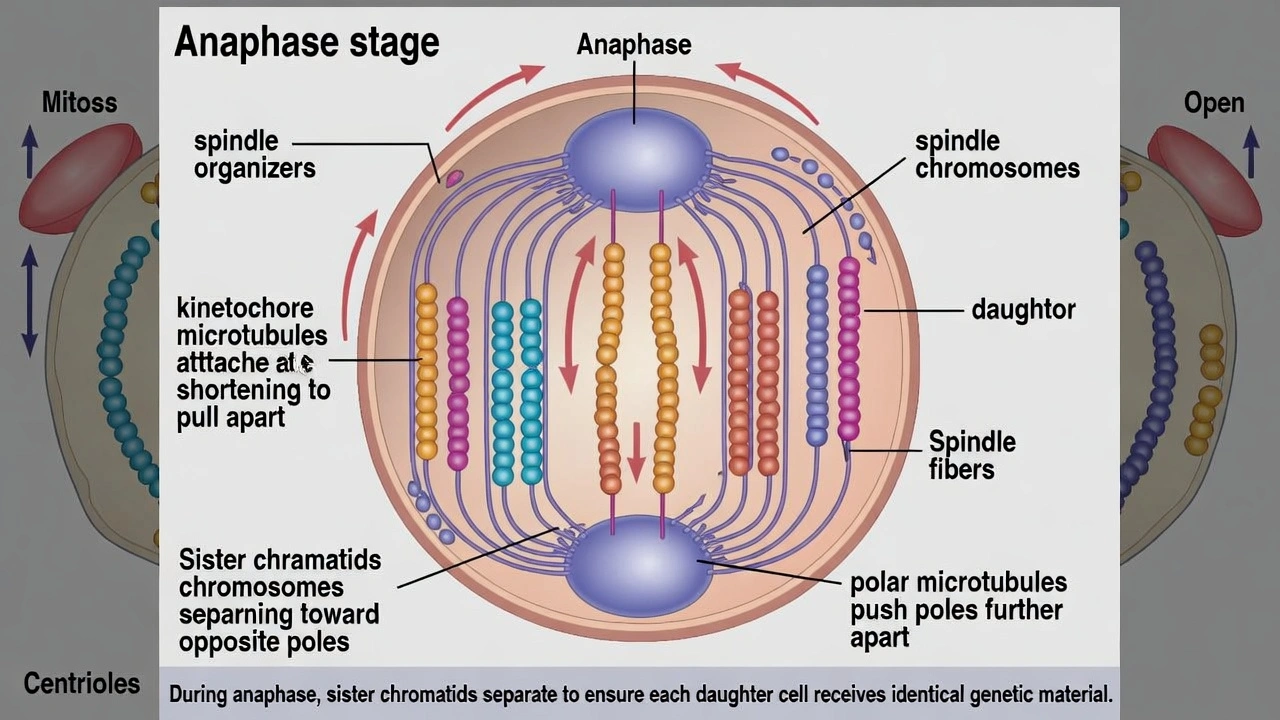
A triple beam balance would show the units of measurement in which form?
A.
Liters
B. Grams
C. Meters
D. Gallons
Rationale
Triple beam balances display measurements in grams. Triple beam balances measure mass by comparing an unknown mass to known counterweights sliding along three calibrated beams with different measurement ranges typically measuring from 0.1 grams to 610 grams with precision to 0.1 gram.
A) Liters
Liters measure volume not mass representing the space occupied by a substance. One liter of water has a mass of approximately 1000 grams but one liter of oil has less mass due to lower density. Triple beam balances cannot measure volume directly requiring graduated cylinders or volumetric flasks for liquid volume measurement.
B) Grams
Triple beam balances measure mass in grams through a mechanical lever system. The three beams typically measure:
• Rear beam: 0-100 grams in 10-gram increments
• Middle beam: 0-500 grams in 100-gram increments
• Front beam: 0-10 grams in 0.1-gram increments
The sum of positions on all three beams gives the total mass in grams. This mechanical measurement system requires no electricity and remains accurate with proper calibration.
C) Meters
Meters measure length or distance in the metric system representing linear dimensions not mass. Rulers meter sticks and tape measures display measurements in meters or centimeters. Triple beam balances have no capability to measure linear dimensions making meters an inappropriate unit for this instrument.
D) Gallons
Gallons measure volume in the imperial system primarily used in the United States. One US gallon equals approximately 3.785 liters. Triple beam balances measure mass not volume and use metric units (grams) not imperial units (gallons). Volume-to-mass conversion requires knowledge of density making direct gallon measurement impossible on a balance.
Conclusion:
Triple beam balances are mechanical instruments designed specifically for mass measurement in grams (the standard metric unit for mass). Option B correctly identifies this unit distinguishing mass measurement from volume (liters gallons) or length (meters) measurements requiring different instruments. Understanding appropriate measurement units and instruments proves essential for accurate scientific data collection and avoiding unit conversion errors in experimental work.
Which of the following do catalysts alter to control the rate of a chemical reaction?
A.
Substrate energy
B. Activation energy
C. Inhibitor energy
D. Promoter energy
Rationale
Catalysts lower the activation energy of chemical reactions. Activation energy represents the energy barrier that reactant molecules must overcome to transform into products. Catalysts provide alternative reaction pathways with reduced activation energy enabling more molecular collisions to succeed at a given temperature without being consumed in the process.
A) Substrate energy
Substrate energy refers to the inherent chemical potential energy stored in reactant molecules. Catalysts do not alter this intrinsic energy content which is determined by molecular structure and bonding. The overall energy difference between reactants and products (ΔG) remains unchanged by catalysts; only the pathway between them is modified.
B) Activation energy
Catalysts function by stabilizing the transition state of a reaction through temporary interactions with reactants creating an alternative pathway requiring less energy input. The activation energy reduction enables a greater proportion of molecular collisions to possess sufficient energy for successful reaction at a given temperature. For example catalase reduces hydrogen peroxide decomposition activation energy from 75 kJ/mol to 23 kJ/mol increasing reaction rate by 10â¹ fold at physiological temperatures. Catalysts emerge unchanged after the reaction allowing single catalyst molecules to facilitate numerous reaction cycles.
C) Inhibitor energy
Inhibitors are substances that decrease reaction rates often by binding to catalysts (enzymes) and blocking active sites or altering conformation. No scientific concept called "inhibitor energy" exists as a reaction parameter. Catalysts counteract inhibition but do not alter a nonexistent "inhibitor energy" parameter.
D) Promoter energy
Promoters enhance catalyst efficiency in some industrial processes but "promoter energy" is not a recognized thermodynamic parameter. Catalysts themselves function by altering activation energy not by modifying hypothetical promoter energy values.
Conclusion:
Activation energy reduction represents the universal mechanism by which all catalysts operate across biological (enzymatic) and industrial contexts. Option B correctly identifies this fundamental principle distinguishing catalysts from reactants (that alter substrate concentrations) or products (that shift equilibrium). Catalysts accelerate reactions without affecting thermodynamic equilibrium positions making activation energy modification their exclusive mode of action essential for understanding reaction kinetics in chemistry biology and industrial processes.
What function do ribosomes serve within the cell?
A.
Ribosomes are responsible for cell movement
B. Ribosomes aid in protein synthesis
C. Ribosomes help protect the cell from its environment
D. Ribosomes have enzymes that help with digestion
Rationale
Ribosomes aid in protein synthesis. Ribosomes function as molecular machines that translate messenger RNA sequences into polypeptide chains by facilitating codon-anticodon recognition between mRNA and transfer RNA while catalyzing peptide bond formation between amino acids to construct proteins according to genetic instructions.
A) Ribosomes are responsible for cell movement
Cell movement depends on cytoskeletal elements like microtubules actin filaments and motor proteins such as dynein kinesin and myosin. Ribosomes lack motility functions and remain either free in cytoplasm or bound to endoplasmic reticulum without involvement in cellular locomotion or organelle transport.
B) Ribosomes aid in protein synthesis
Ribosomes consist of large and small subunits composed of ribosomal RNA and proteins forming three tRNA binding sites: A (aminoacyl) P (peptidyl) and E (exit). During translation the small subunit binds mRNA while tRNA molecules deliver specific amino acids matching codons. The large subunit contains peptidyl transferase activity catalyzing covalent bonds between adjacent amino acids extending the polypeptide chain until a stop codon terminates synthesis.
C) Ribosomes help protect the cell from its environment
Cellular protection comes from the plasma membrane cell wall (in plants and bacteria) specialized structures like tight junctions or immune cells. Ribosomes operate internally synthesizing proteins but provide no barrier function against environmental threats pathogens or physical damage.
D) Ribosomes have enzymes that help with digestion
Digestive enzymes (like proteases lipases and amylases) are proteins synthesized by ribosomes but ribosomes themselves lack enzymatic digestive capabilities. Lysosomes contain hydrolytic enzymes for intracellular digestion while the digestive system secretes enzymes produced by ribosomes in specialized cells.
Conclusion:
Protein synthesis represents ribosomes' exclusive and essential cellular function translating genetic information into functional polypeptides required for virtually all cellular processes. Option B correctly identifies this fundamental role distinguishing ribosomes from motility structures (cytoskeletal elements) protective barriers or digestive organelles. Understanding ribosome function proves critical for comprehending gene expression antibiotic mechanisms that target bacterial ribosomes and diseases caused by translation errors.

In the suburban neighborhood of Northwoods, there have been large populations of deer, and residents have complained about them eating flowers and garden plants. What would be a logical explanation, based on observations, for the large increase in the deer population over the last two seasons?
A.
Increased quantity of food sources
B. Decreased population of a natural predator
C. Deer migration from surrounding areas
D. Increase in hunting licenses sold
Rationale
Decreased population of natural predators likely explains the deer population increase. Predator-prey relationships follow ecological principles where predator removal or decline typically causes prey population expansion due to reduced mortality rates allowing more individuals to survive to reproductive age and produce offspring without top-down population control.
A) Increased quantity of food sources
While suburban gardens provide additional food sources this represents a secondary factor. Food availability typically limits populations only when severely restricted. Deer populations in suburban areas often exceed carrying capacity based on natural forage alone suggesting predation rather than food limitation normally controls their numbers.
B) Decreased population of a natural predator
Predators like wolves coyotes and mountain lions naturally regulate deer populations through predation. Removing these controls allows deer numbers to increase rapidly as birth rates exceed mortality rates without predation pressure. Historical examples include Kaibab Plateau deer explosion after predator eradication and eastern US deer overpopulation following wolf extirpation. This represents the most direct ecological mechanism for sudden population increases.
C) Deer migration from surrounding areas
Migration might contribute to local population increases but cannot explain regional population growth across multiple areas simultaneously. Deer migration typically responds to seasonal resource availability or habitat disturbance not sustained multi-season increases. Migration redistributes existing populations rather than increasing total numbers.
D) Increase in hunting licenses sold
Increased hunting would decrease deer populations not increase them. Hunting serves as human-imposed population control with higher harvest rates reducing population sizes. This option contradicts basic population dynamics principles where increased mortality reduces population growth.
Conclusion:
Predator removal represents the most ecologically sound explanation for rapid deer population increases in suburban environments where natural predators have been eliminated through human development. Option B correctly identifies this trophic cascade mechanism fundamental to wildlife management understanding that predator absence often causes herbivore overpopulation with cascading ecosystem effects including vegetation damage biodiversity loss and increased vehicle collisions.
What is the speed of a wave with a frequency of 12 Hz and a wavelength of 3 meters?
A.
12 meters per second
B. 36 meters per second
C. 4 meters per second
D. 0.25 meters per second
Rationale
The wave speed is 36 meters per second. Wave speed equals frequency multiplied by wavelength according to the fundamental wave equation v = fλ where v represents velocity f represents frequency in hertz (cycles per second) and λ represents wavelength in meters.
A) 12 meters per second
This value equals the frequency alone without multiplying by wavelength or represents wavelength divided by frequency (3 ÷ 12 = 0.25) not the correct calculation. Applying v = fλ requires multiplication not identity with one parameter.
B) 36 meters per second
Substituting values into the wave equation v = fλ gives v = 12 Hz × 3 m = 36 m/s. This calculation correctly applies the fundamental relationship where each cycle spans 3 meters and 12 cycles pass a fixed point each second producing propagation at 36 meters per second. The units verify correctly: hertz × meters = cycles/second × meters = meters/second.
C) 4 meters per second
This value results from incorrectly dividing frequency by wavelength (12 ÷ 3 = 4) rather than multiplying. The inverse relationship would apply only if solving for frequency given speed and wavelength (f = v/λ) not for speed calculation.
D) 0.25 meters per second
This value comes from dividing wavelength by frequency (3 ÷ 12 = 0.25) representing the period (the time for one cycle) not the wave speed. Period T = 1/f = 1/12 second per cycle but speed requires distance per time not time per cycle.
Conclusion:
The wave equation v = fλ provides the direct relationship between propagation speed frequency and wavelength with multiplication yielding speed. Option B correctly applies this fundamental principle essential for understanding wave phenomena across physics from sound waves to electromagnetic radiation where frequency and wavelength inversely relate at constant speed within a given medium.

Which of the following structures has the lowest blood pressure?
A.
arteries
B. arteriole
C. venule
D. vein
Rationale
Veins have the lowest blood pressure in the circulatory system. Blood pressure progressively decreases throughout the circulatory pathway from aortic exit (approximately 100 mmHg systolic) to vena cava entry (approximately 2-5 mmHg) with veins exhibiting the lowest pressures due to energy dissipation through resistance vessels and distance from the heart's pumping action.
A) arteries
Systemic arteries maintain high pressure (systolic 90-120 mmHg diastolic 60-80 mmHg) to propel blood through the vascular tree. Elastic arteries like the aorta dampen pulsatile flow while muscular arteries distribute blood to organs. High arterial pressure ensures adequate perfusion pressure reaches capillary beds despite downstream resistance.
B) arteriole
Arterioles represent the primary resistance vessels where the steepest pressure drop occurs (from approximately 85 mmHg at arteriole entrance to 30 mmHg at exit). Their smooth muscle walls constrict or dilate to regulate flow into capillary beds. This resistance function makes arterioles critical for blood pressure regulation but they still maintain substantially higher pressure than venous structures.
C) venule
Venules collect blood from capillaries at pressures around 10-15 mmHg slightly higher than larger veins. They represent the transition from microcirculation to venous return with thin walls and minimal smooth muscle. Pressure continues declining as blood moves toward the heart but venules maintain higher pressure than veins proper.
D) vein
Veins operate under the lowest pressures in the circulatory system (vena cava approximately 2-5 mmHg). This low pressure necessitates specialized adaptations for venous return: one-way valves prevent backflow skeletal muscle pumps compress veins during movement and respiratory pumps create thoracic pressure gradients. These mechanisms overcome gravity and low pressure to return blood to the heart against minimal driving pressure.
Conclusion:
Blood pressure follows a continuous decline from arteries through arterioles capillaries venules to veins with veins exhibiting the lowest values essential for understanding circulatory physiology. Option D correctly identifies veins as the lowest pressure structures explaining why venous insufficiency causes edema and why veins require valves and external compression mechanisms for effective return flow unlike high-pressure arterial systems.
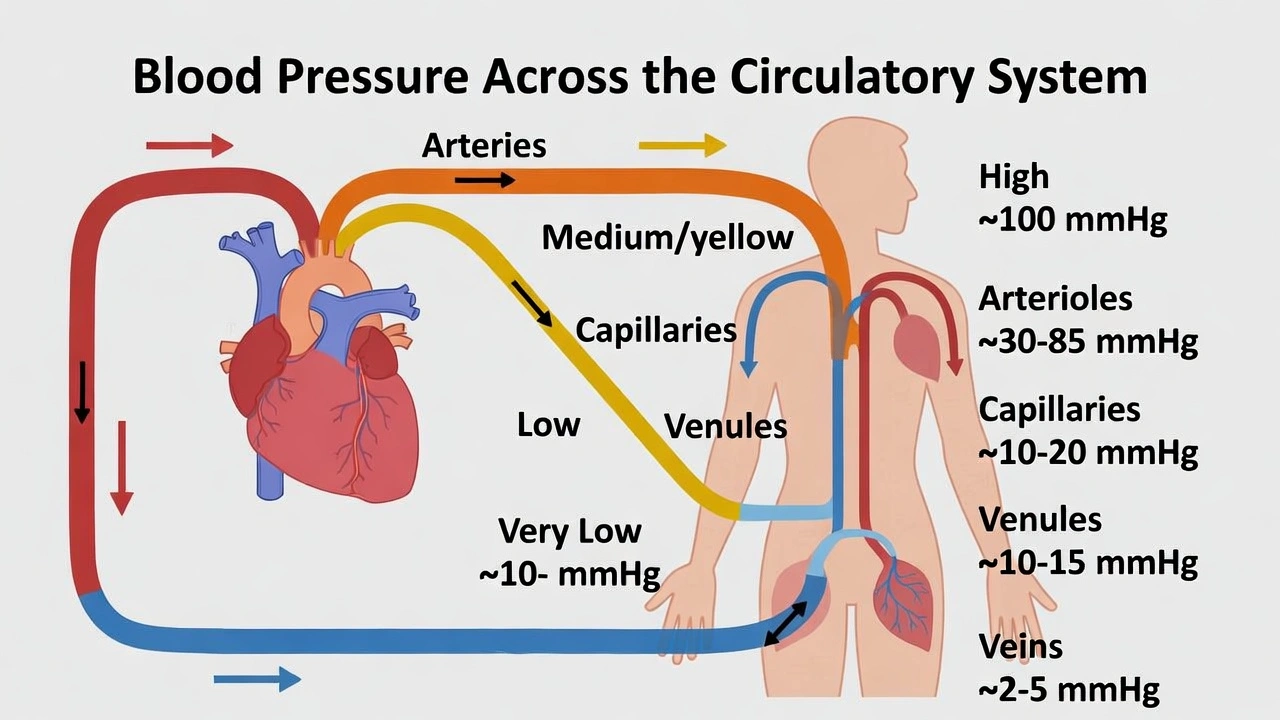
A cyclist is riding over a hill. At what point is his potential energy greatest?
A.
at the base of the hill
B. halfway up the hill
C. at the very top of the hill
D. on the way down the hill
Rationale
Potential energy is greatest at the very top of the hill. Gravitational potential energy equals mass times gravitational acceleration times height (PE = mgh) making it directly proportional to vertical elevation above a reference point with maximum height corresponding to maximum potential energy.
A) at the base of the hill
The base represents the lowest elevation point in the scenario where gravitational potential energy reaches its minimum value. Kinetic energy may be maximal here if the cyclist has accelerated downhill but potential energy depends solely on height not speed.
B) halfway up the hill
Halfway elevation produces intermediate potential energy values between minimum at the base and maximum at the summit. Since potential energy scales linearly with height halfway up yields approximately half the maximum potential energy not the greatest value.
C) at the very top of the hill
The summit represents the maximum vertical displacement from the reference point (typically the base or sea level) creating the greatest gravitational potential energy. At this point the cyclist possesses maximum capacity to convert potential energy into kinetic energy during descent. This principle enables roller coasters to use initial elevation for subsequent motion without additional propulsion.
D) on the way down the hill
Descending positions have decreasing elevation and therefore decreasing potential energy as height diminishes. Potential energy continuously converts to kinetic energy during descent making any downhill position lower in potential energy than the summit.
Conclusion:
Gravitational potential energy depends exclusively on vertical height with maximum elevation producing maximum potential energy. Option C correctly identifies the hilltop as the point of greatest potential energy essential for understanding energy transformations in mechanics sports physiology and engineering systems where elevation changes drive motion through potential-kinetic energy conversion.
The atomic number of an element is defined by:
A.
The total number of protons and electrons it contains
B. The total number of electrons it contains
C. The total number of protons it contains
D. The total number of neutrons it contains
Rationale
The atomic number equals the number of protons in an atom's nucleus. The atomic number uniquely identifies each chemical element determining its position in the periodic table chemical properties and electron configuration in neutral atoms though ionization can alter electron count without changing elemental identity.
A) The total number of protons and electrons it contains
This sum varies with ionization state. Neutral atoms have equal protons and electrons but ions gain or lose electrons changing this total while remaining the same element. A sodium atom (Na) and sodium ion (Naâº) both have atomic number 11 despite different electron counts making proton-electron sum an unreliable identifier.
B) The total number of electrons it contains
Electron count changes during chemical reactions and ionization without altering elemental identity. Chlorine atoms gain electrons to form Clâ» ions but remain chlorine with atomic number 17. The variable nature of electron count disqualifies it as the defining characteristic of an element.
C) The total number of protons it contains
Proton count remains constant for all atoms of a given element regardless of isotope or ionization state. Carbon always has 6 protons whether as ¹²C ¹³C ¹â´C isotopes or as Câ´âº ions. Changing proton count transforms the atom into a different element through nuclear reactions not chemical processes. This invariance makes proton number the fundamental defining property of elements.
D) The total number of neutrons it contains
Neutron count varies among isotopes of the same element. Carbon-12 has 6 neutrons carbon-13 has 7 and carbon-14 has 8 yet all remain carbon with atomic number 6. Neutron variation affects nuclear stability and mass but not elemental identity or chemical behavior.
Conclusion:
Proton number exclusively defines elemental identity remaining invariant across isotopes ions and chemical states. Option C correctly identifies this fundamental principle essential for understanding periodic table organization nuclear reactions that transmute elements and why chemical properties depend on proton-determined electron configurations rather than neutron or electron variations.
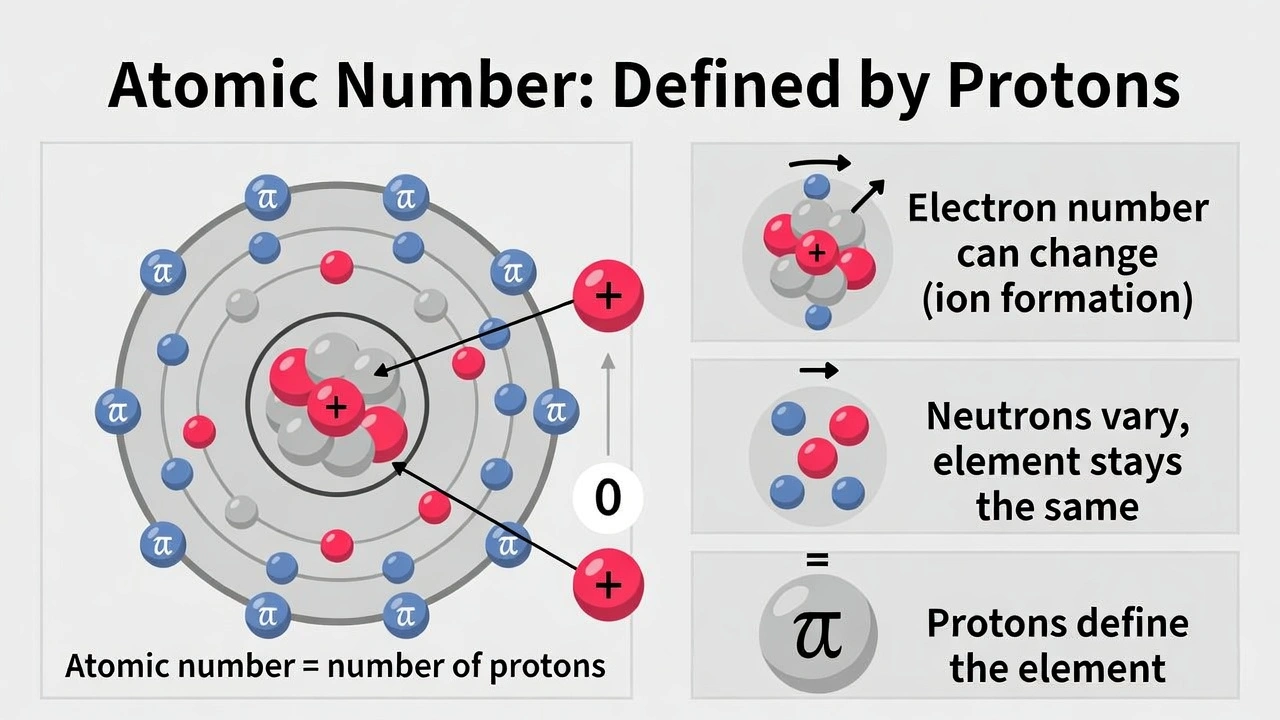
NEX Exams
NEX Quizzes
3 Practice Tests
NEX Quizzes
3 Practice Tests
NEX Quizzes
3 Practice Tests
NEX Quizzes
3 Practice Tests