How many electrons does a neutral atom of iodine have?
-
A
53
-
B
74
-
C
126
-
D
127
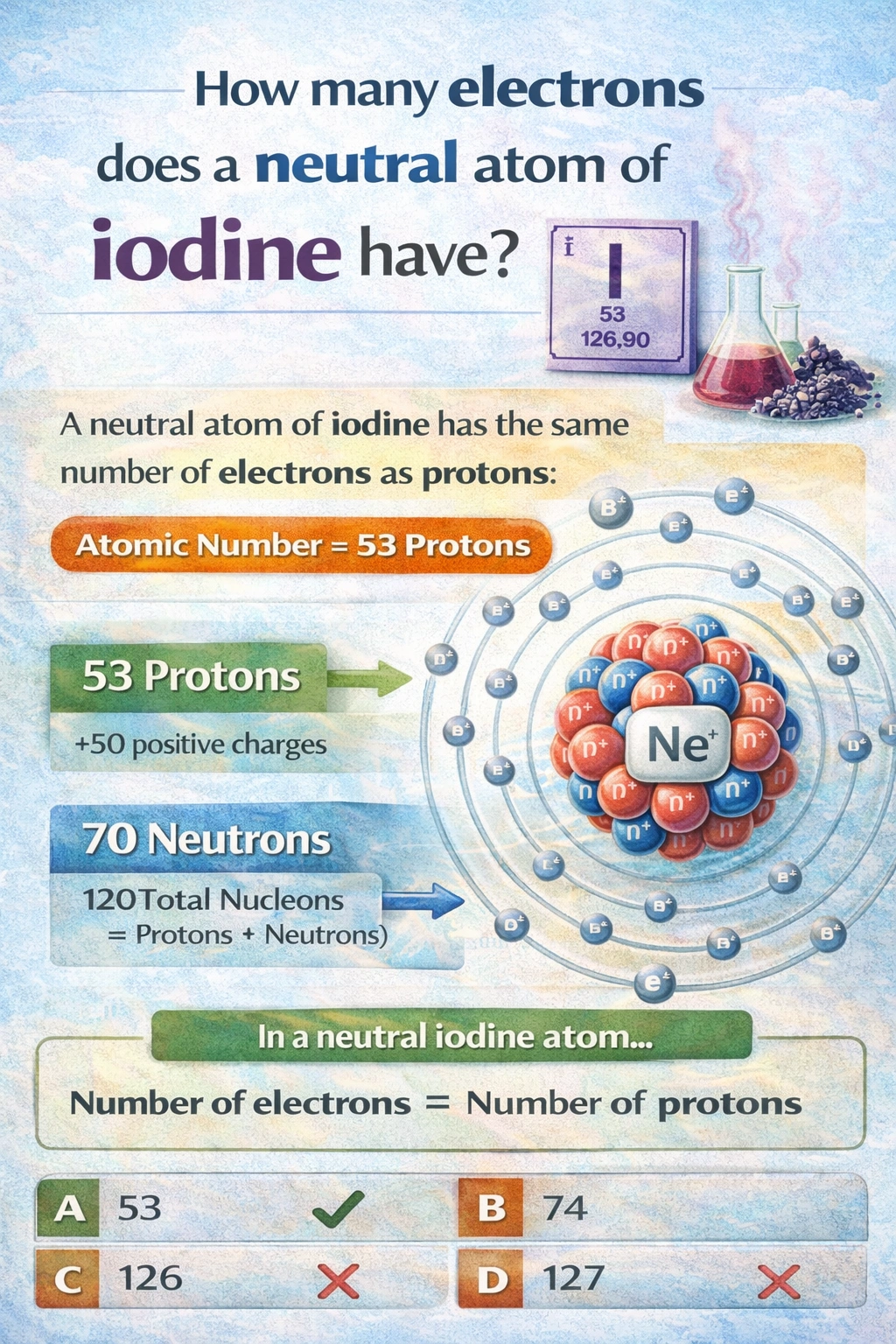
A neutral atom of iodine has 53 electrons.
The number of electrons in a neutral atom is equal to its atomic number, which is the number of protons in the nucleus. This equality ensures the atom has no net electrical charge.
A) 53
Iodine (I) has an atomic number of 53. This means every iodine atom contains 53 protons in its nucleus. For the atom to be electrically neutral, it must also contain 53 electrons to balance the positive charge of the 53 protons. This is the fundamental rule governing neutral atoms.
B) 74
The number 74 is the atomic number of tungsten (W), not iodine. It may be mistakenly associated with the neutron count of a common iodine isotope or confused with another element. It does not correspond to the proton count of iodine.
C) 126
The number 126 is approximately double iodine's atomic number and is close to the neutron count for some of its heavier isotopes. For example, iodine-127 has 74 neutrons (127-53=74). The number 126 is not a standard value for iodine's subatomic particles and is certainly not its electron count in a neutral atom.
D) 127
The number 127 is the mass number of the most common stable isotope of iodine, Iodine-127. The mass number is the sum of protons and neutrons (53 protons + 74 neutrons = 127). This is a measure of the atom's approximate mass, not its electron count.
Conclusion:
The defining characteristic of an element is its atomic number (proton count). In a neutral atom, the electron count must match the proton count to achieve charge balance. Iodine's atomic number is 53, so a neutral iodine atom possesses 53 electrons.
Topic Flashcards
Click to FlipWhat is the atomic number of iodine, and what does this number represent?
53. It represents the number of protons in the nucleus of every iodine atom.
For any neutral atom, how is the number of electrons related to the number of protons?
hey are equal. The equal positive (protons) and negative (electrons) charges cancel out, giving the atom a net charge of zero.
How many electrons does a neutral atom of iodine-127 (the most common isotope) have?
53 electrons. The isotope mass (127) changes the neutron count, not the proton or electron count in a neutral atom.
In the option "127" for the electron count, what does the number 127 actually refer to for an iodine atom?
The mass number of the iodine-127 isotope (53 protons + 74 neutrons = 127).
If an iodine atom gained one extra electron, how many electrons would it have, and what would its charge be?
54 electrons. It would be a negatively charged ion (an anion) with a charge of -1.