HESI A2 CHEMISTRY PRACTICE EXAM
This HESI A2 Chemistry practice exam provides a full quiz experience with questions that simulate the real exam. It is useful for measuring your preparedness and identifying weak areas in chemistry before test day.
Topics Covered
Atomic Theory
Chemical Reactions
Periodic Trends
Acids and Bases
Chemical Calculations
00:00
A substance with a pH of 3 is how many times more acidic than a substance with a pH of 5?
A.
8
B. 2
C. 100
D. 1,000
Rationale
A substance with pH 3 is 100 times more acidic than a substance with pH 5.
The pH scale operates on a logarithmic base-10 system where each whole number decrease represents a tenfold increase in hydrogen ion concentration. A two-unit difference between pH values therefore corresponds to 10s², yielding a hundredfold difference in acidity.
A) 8
The value 8 does not correspond to any standard calculation within the logarithmic pH framework. It may arise from arbitrary arithmetic operations such as averaging or linear interpolation between pH values. The pH scale does not permit simple addition, subtraction, or averaging to determine acidity ratios. Logarithmic relationships require exponential calculations rather than linear manipulations. This value lacks mathematical or chemical basis in pH comparisons.
B) 2
This value results from subtracting the pH numbers directly (5 ˆ’ 3 = 2), treating the scale as linear. The pH scale is logarithmic, not linear, so numerical subtraction does not reflect actual concentration differences. A one-unit pH change already represents a tenfold acidity difference, not a single unit. Linear subtraction fundamentally misrepresents the exponential nature of hydrogen ion concentration changes.
C) 100
The pH scale is defined as the negative base-10 logarithm of hydrogen ion concentration ([H?]). A decrease of one pH unit corresponds to a tenfold increase in [H?]; a two-unit decrease corresponds to 10s². Between pH 5 and pH 3, the hydrogen ion concentration increases by a factor of 10 x 10, or 100. This exponential relationship is intrinsic to logarithmic measurement systems used in chemistry and biology.
D) 1,000
This value corresponds to a three-unit pH difference (10³ = 1,000), not the two-unit difference present in the question. It would apply when comparing pH 2 to pH 5 or pH 3 to pH 6. Applying a three-unit exponent to a two-unit gap overestimates acidity by one full order of magnitude. The calculation misaligns the exponent with the actual numerical difference between the given pH values.
Conclusion
The logarithmic nature of the pH scale dictates that acidity differences scale exponentially with each unit change. A two-unit separation between pH 3 and pH 5 produces a 10s², or 100-fold, increase in hydrogen ion concentration. Values derived from linear operations or incorrect exponents fail to reflect this fundamental mathematical relationship. The hundredfold difference represents the precise quantitative relationship between these two pH values.

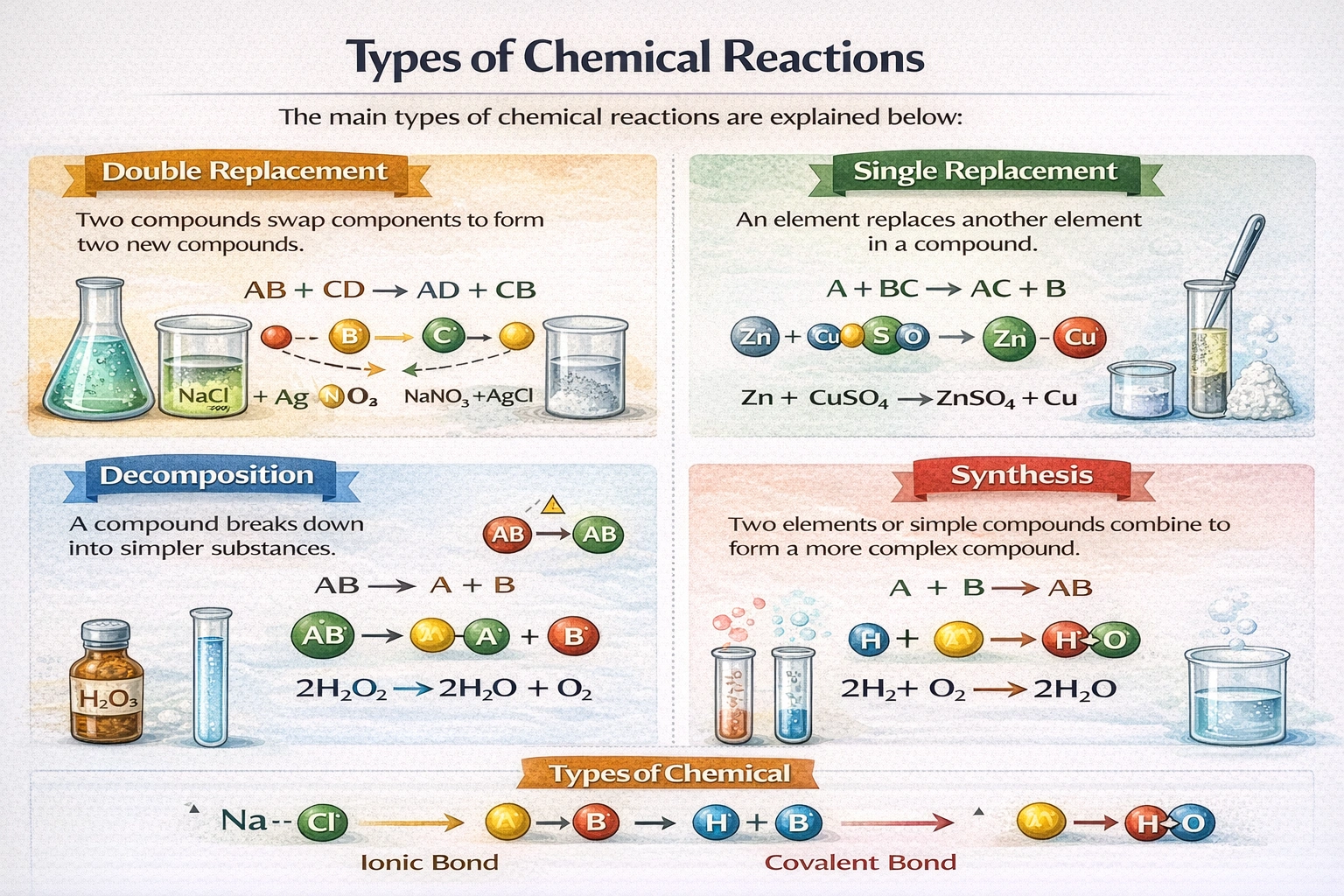
Al(NO?)? + H?SO? ? Al?(SO?)? + HNO? is an example of which kind of reaction?
A.
Decomposition reaction
B. Synthesis reaction
C. Single replacement reaction
D. Double replacement reaction
Rationale
Double replacement reactions involve two ionic compounds exchanging ions to form two new compounds.
In this equation, aluminum ions from aluminum nitrate combine with sulfate ions from sulfuric acid to form aluminum sulfate, while hydrogen ions pair with nitrate ions to form nitric acid. This partner-swapping mechanism defines the double replacement reaction class.
A) Decomposition reaction
Decomposition reactions begin with a single reactant that breaks apart into multiple simpler products. The general form follows AB ? A + B or similar fragmentation patterns. This equation starts with two distinct reactants rather than one. Both products remain compound-level substances without elemental formation. The reaction direction moves toward recombination rather than breakdown.
B) Synthesis reaction
Synthesis reactions combine two or more reactants into a single unified product. The characteristic pattern follows A + B ? AB or extensions with multiple inputs yielding one output. This equation produces two separate compounds as products rather than consolidating into one substance. Aluminum sulfate and nitric acid form independently without merging. The two-reactant, two-product stoichiometry contradicts synthesis requirements.
C) Single replacement reaction
Single replacement reactions require one elemental reactant displacing another within a compound. The standard form follows A + BC ? AC + B where A represents a free element. Both reactants in this equation exist as compounds, Al(NO?)? and H?SO?, without elemental participants. Aluminum does not enter as free metal nor does hydrogen appear as H? gas. The mechanism involves mutual ion exchange rather than unidirectional displacement.
D) Double replacement reaction
Double replacement reactions follow the pattern AB + CD ? AD + CB where cations and anions exchange partners. Aluminum nitrate provides Al³? and NO?? ions while sulfuric acid supplies H? and SO?s²? ions. The Al³? ions combine with SO?s²? to form Al?(SO?)? and H? ions combine with NO?? to form HNO?. Both ionic pairs reorganize simultaneously without elemental involvement. Precipitation, gas formation, or acid-base neutralization often drive these exchanges.
Conclusion
The reaction demonstrates simultaneous exchange of cation-anion partners between two ionic compounds. Aluminum and hydrogen ions swap their original anionic associations to generate new compound pairings. This bidirectional recombination without elemental reactants or single-product formation aligns precisely with double replacement mechanics. The stoichiometric rearrangement confirms this reaction class beyond alternative classifications.
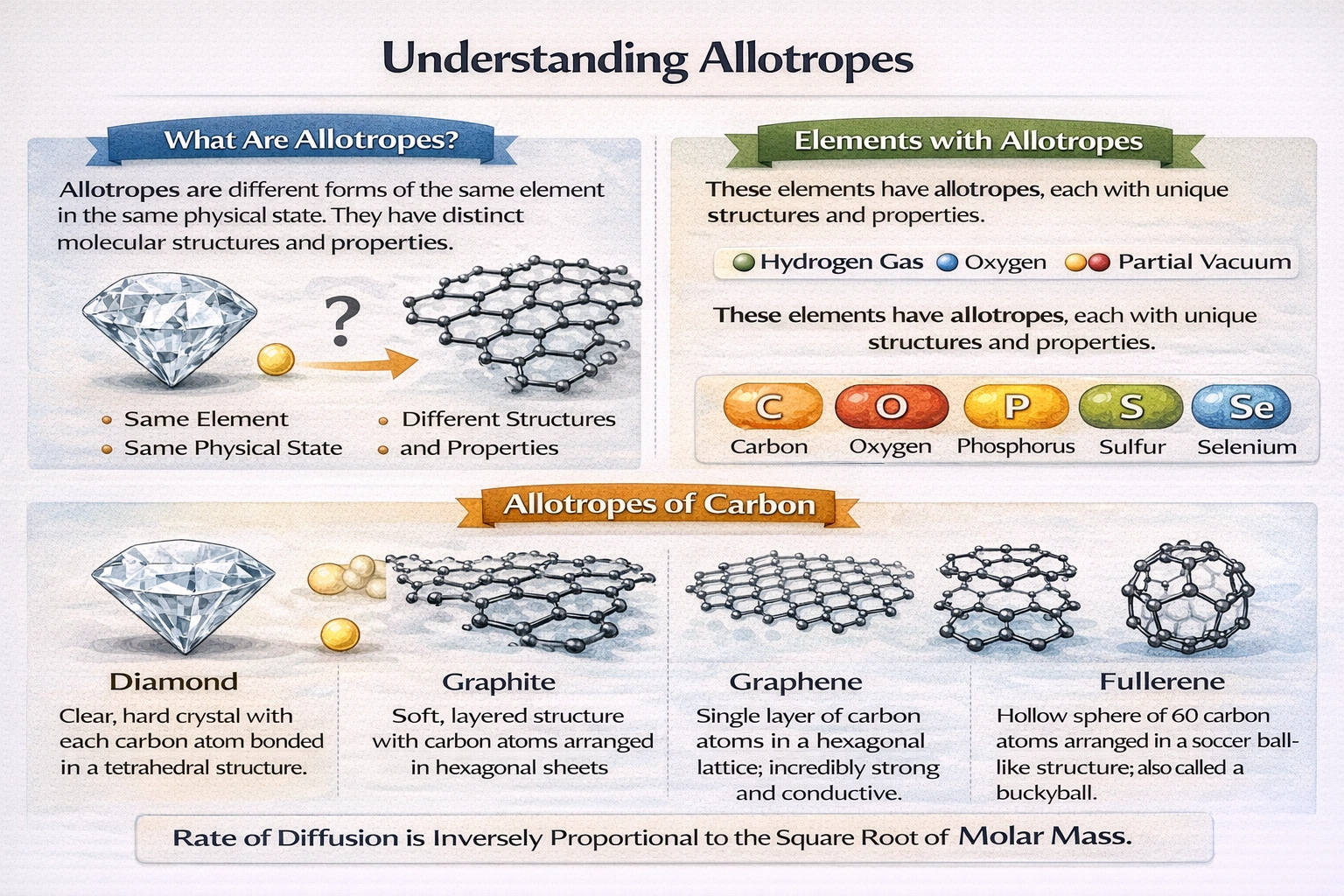
Which of the following is not an allotrope of carbon?
A.
Diamonds
B. Graphite
C. Fluorine
D. Buckminsterfullerene
Rationale
Fluorine is not an allotrope of carbon. Allotropes represent different structural arrangements of atoms within the same element, not distinct chemical elements. Fluorine exists as a separate element with atomic number 9, fundamentally different from carbon which has atomic number 6.
A) Diamonds
Diamond consists entirely of carbon atoms arranged in a tetrahedral lattice where each carbon forms four strong covalent bonds. This three-dimensional network produces exceptional hardness and thermal conductivity. The structure lacks free electrons, making diamond an electrical insulator despite its thermal properties. Diamond represents one of carbon's most thermodynamically stable allotropes under standard conditions.
B) Graphite
Graphite comprises carbon atoms organized in stacked hexagonal layers with each atom bonded to three neighbors. Weak van der Waals forces between layers allow easy slippage, producing lubricating properties. Delocalized electrons within each layer enable electrical conductivity parallel to the planes. This layered architecture creates anisotropic physical properties distinct from diamond's isotropic behavior.
C) Fluorine
Fluorine is a chemical element with symbol F and atomic number 9, residing in Group 17 of the periodic table as a halogen. It exists naturally as diatomic F? molecules with a pale yellow gas appearance. Fluorine possesses seven valence electrons and exhibits the highest electronegativity of all elements. Its atomic structure and chemical behavior bear no relationship to carbon or carbon-based molecular arrangements.
D) Buckminsterfullerene
Buckminsterfullerene (C‚†‚€) forms a closed spherical structure of 60 carbon atoms arranged in pentagons and hexagons resembling a soccer ball. Each carbon atom bonds to three others with partial double-bond character distributed across the cage. This molecular allotrope dissolves in organic solvents unlike diamond or graphite. Discovered in 1985, it launched the field of nanoscale carbon materials including carbon nanotubes and graphene.
Conclusion
Allotropes must consist of identical elements arranged in different structural configurations. Diamond, graphite, and buckminsterfullerene all comprise pure carbon in distinct bonding geometries. Fluorine represents an entirely separate element with different atomic number, electron configuration, and chemical properties. Only fluorine fails the fundamental requirement of being composed of carbon atoms.
Blood with a pH of 1.3 indicates what about the blood sample?
A.
It is strongly acidic.
B. It is strongly basic.
C. It is weakly acidic.
D. It is weakly basic.
Rationale
A blood sample with a pH of 1.3 is strongly acidic. The pH scale spans from 0 to 14, with values below 7 indicating acidity and lower numbers representing stronger acidity. A pH of 1.3 corresponds to a hydrogen ion concentration of approximately 5x10?s² M, about 5 million times greater than that of neutral water (pH 7). This level of acidity is comparable to gastric acid or battery acid and is incompatible with life, as normal human blood maintains a tightly regulated pH between 7.35 and 7.45.
A) It is strongly acidic
This is correct. pH 1.3 falls in the range of strong acids (pH < 2), indicating nearly complete dissociation of a strong acid like HCl or H?SO? in the sample. Such extreme acidity would denature proteins, disrupt cellular function, and destroy biological structures. While no living organism can sustain blood at this pH, the question assesses the chemical interpretation of the value, not physiological plausibility.
B) It is strongly basic
Basic solutions have pH > 7, with strong bases like NaOH yielding pH values of 12-14. A pH of 1.3 is on the opposite end of the scale and reflects high [H?], not high [OH?]. This option fundamentally misreads the pH scale.
C) It is weakly acidic
Weakly acidic solutions (e.g., coffee, milk) have pH values between 4 and 6. These result from partial dissociation of weak acids like acetic or carbonic acid. pH 1.3 is far more acidic, representing a 1,000- to 10,000-fold higher [H?] than weakly acidic solutions, and thus cannot be classified as "weak."
D) It is weakly basic
Weak bases (e.g., baking soda solution) have pH values between 8 and 10. A pH of 1.3 is not only non-basic but extremely acidic, making this choice chemically nonsensical.
Conclusion
The pH scale is logarithmic and directional: lower values = more acidic. A pH of 1.3 unambiguously indicates strong acidity, regardless of biological context. Thus, A) It is strongly acidic is the correct answer.
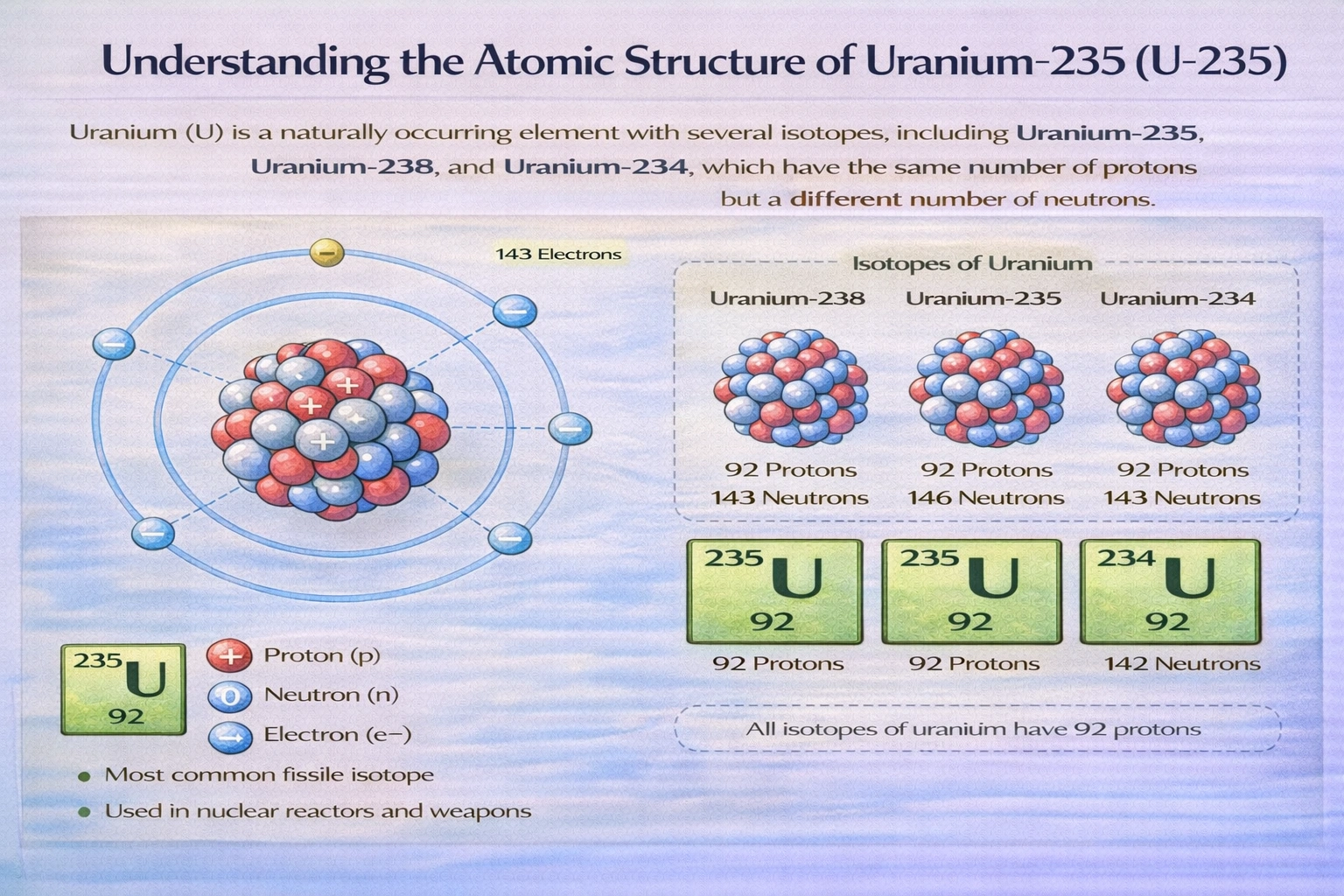
How many neutrons are in an atom of uranium-235?
A.
92
B. 125
C. 143
D. 235
Rationale
An atom of uranium-235 contains 143 neutrons. The mass number 235 represents the total count of protons and neutrons in the nucleus. Uranium's atomic number of 92 fixes the proton count across all isotopes, leaving 143 neutrons to complete the mass number.
A) 92
The value 92 represents uranium's atomic number, defining the proton count in every uranium isotope. Protons determine elemental identity and remain constant regardless of isotopic variation. Neutron count varies between isotopes while proton count stays fixed at 92. This number describes nuclear charge carriers, not neutral particles within the nucleus.
B) 125
The value 125 does not correspond to any standard calculation for uranium-235's composition. Subtracting atomic number 92 from mass number 235 yields 143, not 125. No common uranium isotope possesses exactly 125 neutrons, uranium-217 would contain this count but is highly unstable and not naturally occurring. This value lacks mathematical or isotopic basis for the given nuclide.
C) 143
Uranium-235 has a mass number of 235, representing the sum of protons and neutrons. With 92 protons fixed by uranium's atomic number, subtraction yields 235 ˆ’ 92 = 143 neutrons. This neutron count distinguishes uranium-235 from uranium-238, which contains 146 neutrons. The 143-neutron configuration contributes to this isotope's nuclear instability and fissionability under thermal neutron bombardment.
D) 235
The value 235 represents the mass number, the combined total of protons and neutrons, not neutrons alone. Mass number serves as isotopic notation (e.g., uranium-235) but requires subtraction of atomic number to isolate neutron count. Confusing mass number with neutron count ignores the proton contribution inherent to all atomic nuclei. This value describes the entire nucleon population rather than its neutron subset.
Conclusion
Neutron count derives from the relationship: neutrons = mass number ˆ’ atomic number. For uranium-235, this calculation yields 235 ˆ’ 92 = 143 neutrons. The atomic number remains invariant across isotopes while neutron count varies to produce distinct nuclear species. This fundamental arithmetic underpins isotopic identification and nuclear property prediction across the periodic table.
Which ion would you expect to dominate in water solutions of bases?
A.
MgCl?
B. 2HCl
C. H?
D. OH?
Rationale
Hydroxide (OH?) ions dominate in aqueous solutions of bases. According to the Arrhenius definition, bases increase hydroxide ion concentration when dissolved in water through dissociation or reaction with water molecules. Elevated OH? concentration directly produces pH values greater than 7 and governs characteristic base behavior including proton acceptance and reaction with acids.
A) MgCl?
Magnesium chloride is a neutral ionic compound, not an ion itself. It dissociates completely in water to form Mgs²? cations and Cl? anions. Neither ion hydrolyzes significantly to alter solution pH under standard conditions. The resulting solution remains near neutral with no dominant basic species present. This compound represents a salt derived from strong acid and base neutralization rather than a base itself.
B) 2HCl
Hydrochloric acid is a strong acid that dissociates to produce H? (hydronium) ions and chloride ions in aqueous solution. The coefficient "2" represents stoichiometric quantity and does not define an ion species. HCl solutions exhibit acidic character with pH values below 7 due to high H? concentration. Acidic solutions fundamentally oppose basic character and cannot dominate in base-containing systems.
C) H?
Hydrogen ions exist in water primarily as hydronium ions (H?O?) through proton hydration. These ions define acidic solutions and decrease in concentration as basicity increases. The ion product of water (Kw = [H?][OH?] = 1.0 x 10?²´ at 25°C) dictates an inverse relationship between H? and OH? concentrations. Elevated H? levels suppress basic properties and characterize proton-donating species rather than bases.
D) OH?
Hydroxide ions form when Arrhenius bases such as NaOH or KOH dissociate in water, releasing OH? directly into solution. Weak bases like ammonia generate OH? indirectly through proton acceptance from water molecules. OH? concentration determines solution basicity, drives neutralization reactions with acids to form water, and creates the slippery tactile sensation characteristic of basic solutions. This ion remains the universal indicator of basic aqueous environments across chemical classification systems.
Conclusion
Basic solutions are defined by hydroxide ion predominance relative to hydrogen ions. OH? concentration directly correlates with base strength and pH elevation above neutrality. While alternative base definitions exist (Brønsted-Lowry, Lewis), aqueous basicity universally manifests through elevated OH? levels. This ion serves as the quantitative and qualitative marker distinguishing basic solutions from acidic or neutral systems.

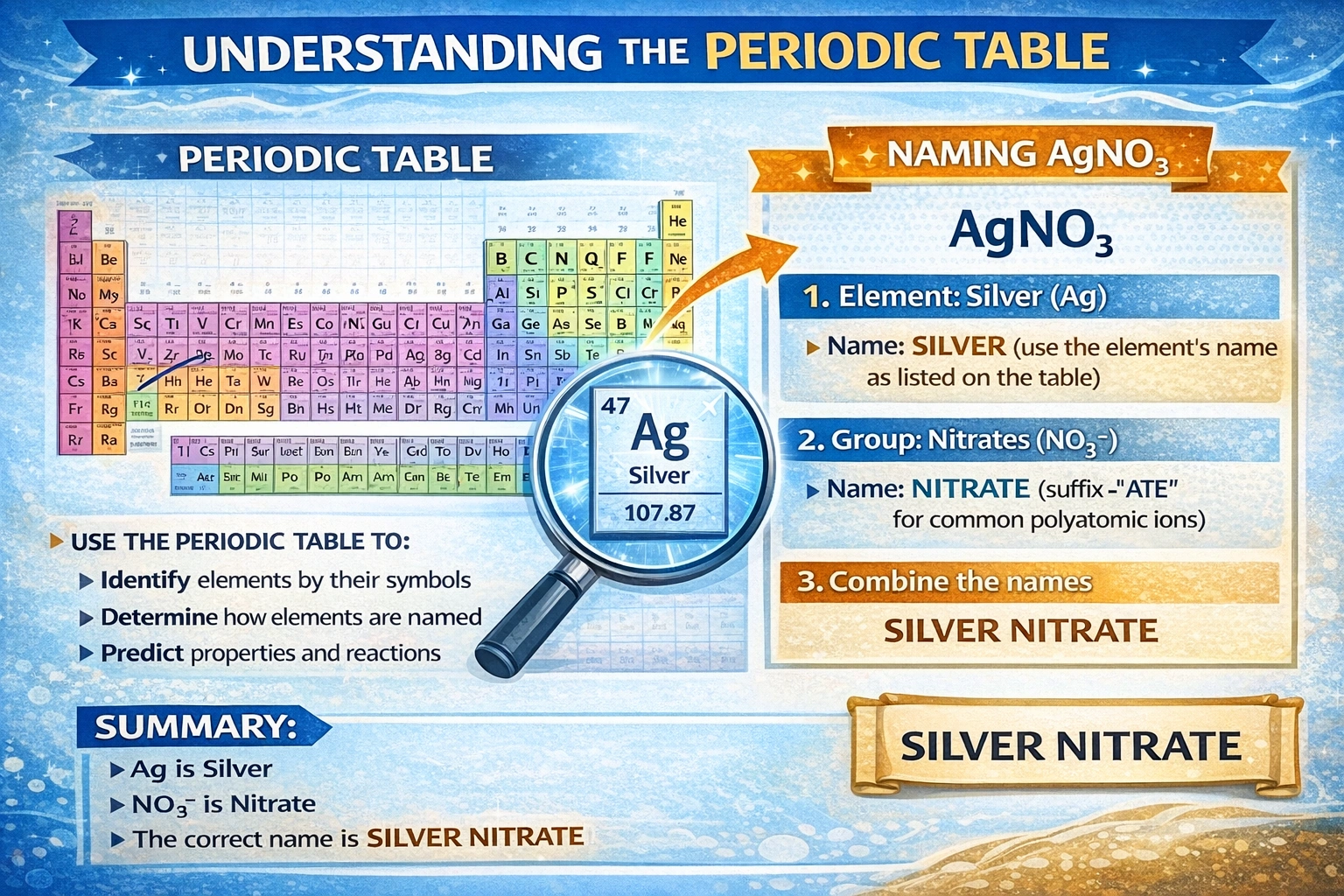
What is the correct name of AgNO??
A.
Argent nitrous
B. Argent oxide
C. Silver nitrite
D. Silver nitrate
Rationale
Silver nitrate is the correct IUPAC name for AgNO?. The compound consists of the silver cation (Ag?) paired with the nitrate anion (NO??). Standard nomenclature combines the cation name "silver" with the anion name "nitrate" to form the complete compound designation.
A) Argent nitrous
"Argent" derives from the Latin argentum and represents an archaic term for silver occasionally seen in historical contexts. "Nitrous" incorrectly refers to the nitrite ion (NO??) or nitrogen oxides with lower oxidation states. The compound AgNO? contains the nitrate ion (NO??), not nitrite. Modern chemical nomenclature requires "silver" rather than "argent" for systematic naming. This designation combines outdated terminology with an incorrect anion identification.
B) Argent oxide
Oxide ions carry a 2- charge (Os²?) and form compounds such as Ag?O (silver oxide). AgNO? contains no oxygen atoms in oxide form; its oxygen atoms exist within the polyatomic nitrate group. The nitrate ion (NO??) comprises one nitrogen atom bonded to three oxygen atoms in a trigonal planar arrangement. Silver oxide and silver nitrate represent chemically distinct compounds with different solubilities, reactivities, and applications. This name misidentifies both the anion type and elemental composition.
C) Silver nitrite
Nitrite ions (NO??) contain one nitrogen atom bonded to two oxygen atoms and carry a single negative charge. Silver nitrite has the formula AgNO? and appears as a pale yellow crystalline solid. Nitrate ions (NO??) contain three oxygen atoms per nitrogen and exhibit different chemical behavior, including greater stability and distinct reactivity patterns. Confusing nitrite with nitrate represents a common nomenclature error with practical consequences in laboratory safety and chemical handling.
D) Silver nitrate
Silver commonly exhibits a +1 oxidation state in ionic compounds, forming Ag? cations. The nitrate ion (NO??) carries a -1 charge with nitrogen in the +5 oxidation state. Charge balance requires a 1:1 ratio, yielding AgNO? as the empirical formula. This compound appears as colorless crystals highly soluble in water and serves as a fundamental reagent in analytical chemistry, photography, and silver plating. IUPAC nomenclature rules specify combining the cation name "silver" with the anion name "nitrate" without modification.
Conclusion
AgNO? comprises Ag? cations and NO?? anions in a 1:1 stoichiometric ratio. The systematic name follows IUPAC conventions for binary ionic compounds containing polyatomic ions. "Silver nitrate" accurately reflects both constituent ions without archaic terminology or anion confusion. Precise nomenclature prevents dangerous misunderstandings between similarly named compounds like nitrate and nitrite, which exhibit markedly different toxicological and chemical properties.
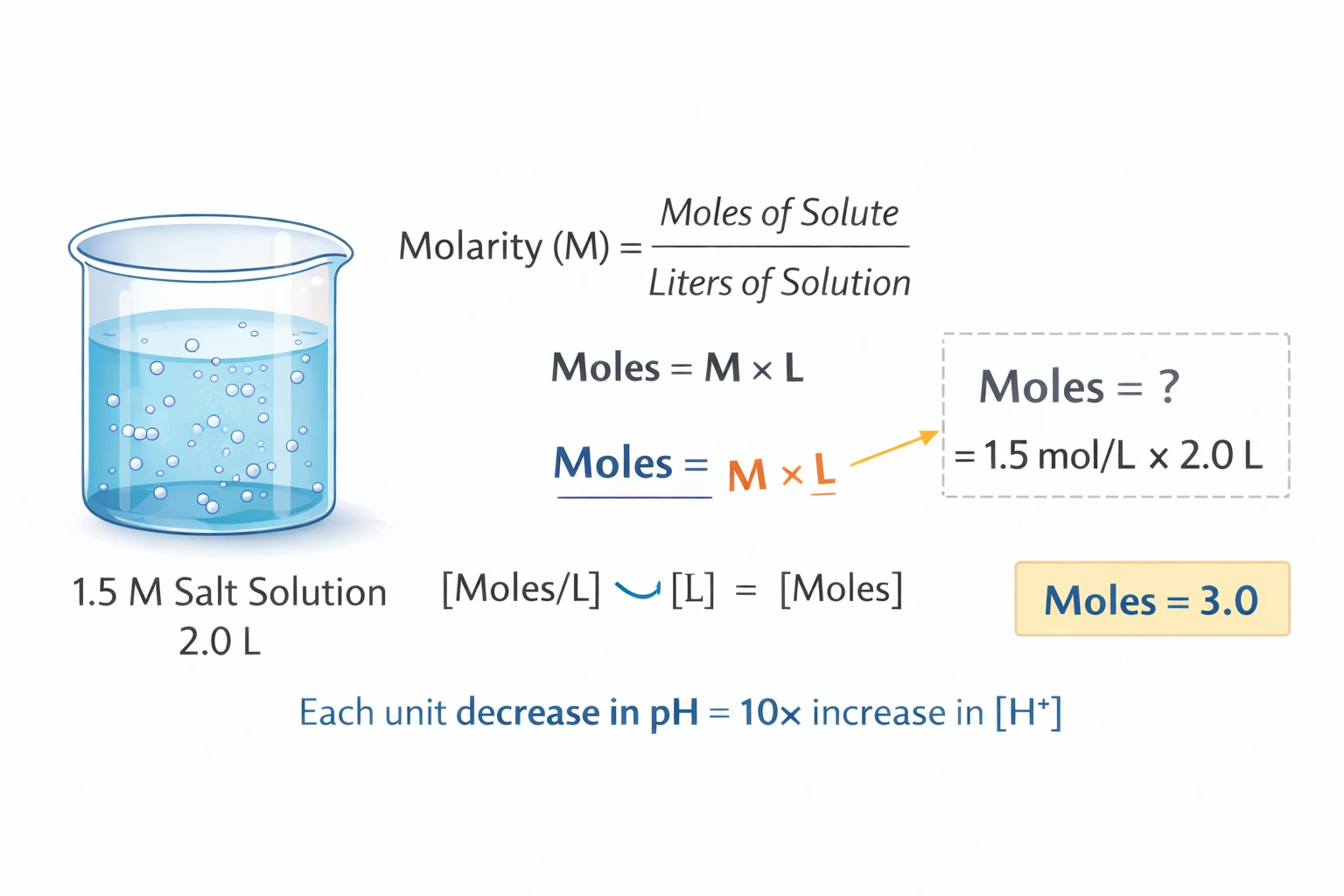
A salt solution has a molarity of 1.5 M. How many moles of this salt are present in 2.0 L of this solution?
A.
0.75
B. 1.5
C. 2
D. 3
Rationale
A 1.5 M salt solution contains 3.0 moles of solute in 2.0 liters of solution. Molarity defines concentration as moles of solute per liter of solution, requiring multiplication of molarity by volume to determine total moles. The calculation 1.5 mol/L x 2.0 L yields 3.0 moles with proper unit cancellation.
A) 0.75
This value results from incorrectly dividing molarity by volume rather than multiplying. The relationship moles = molarity x volume requires multiplication to scale concentration across solution volume. Division would produce moles per liter squared, a dimensionally invalid quantity. This calculation reverses the fundamental molarity equation and yields an unphysical result for the given parameters.
B) 1.5
This value represents the concentration alone without accounting for solution volume. Molarity describes intensive concentration independent of sample size, whereas moles represent extensive quantity dependent on volume. A 1.5 M solution contains 1.5 moles only in exactly 1.0 liter of solution. Doubling the volume to 2.0 liters proportionally doubles the mole quantity to 3.0 moles.
C) 2.0
This value corresponds solely to the solution volume in liters, ignoring solute concentration entirely. Volume alone cannot determine mole quantity without concentration information. Pure water at 2.0 L contains negligible dissolved ions, whereas a 1.5 M salt solution at identical volume contains substantial solute. Mole determination requires both concentration and volume parameters simultaneously.
D) 3.0
Molarity (M) equals moles of solute divided by liters of solution, rearranged as moles = M x L. Substituting 1.5 mol/L for molarity and 2.0 L for volume gives 1.5 x 2.0 = 3.0 moles. Unit analysis confirms liters cancel properly, leaving moles as the resulting dimension. This calculation aligns with laboratory practice for preparing solutions of specified concentration and volume.
Conclusion
Mole quantity scales linearly with both concentration and volume in homogeneous solutions. The 1.5 M concentration specifies 1.5 moles per liter, so two liters necessarily contain twice that amount. Dimensional analysis validates the multiplication operation as physically meaningful. This straightforward relationship underpins solution preparation, dilution calculations, and stoichiometric conversions in aqueous chemistry.
What is the expected pH of orange juice?
A.
Between 3.0 and 4.0
B. Between 6.0 and 7.0
C. Between 8.0 and 9.0
D. Between 11.0 and 12.0
Rationale
Orange juice exhibits a pH between 3.0 and 4.0 due to its organic acid composition. Citric acid constitutes the primary acidic component, supplemented by smaller amounts of ascorbic acid (vitamin C) and malic acid. These weak acids dissociate partially in aqueous solution, releasing hydrogen ions that establish the characteristic tart flavor and acidic pH range typical of citrus beverages.
A) Between 3.0 and 4.0
Citric acid concentration in orange juice ranges from 0.8% to 1.2% by mass, sufficient to depress pH into the moderately acidic range. Measured pH values for commercial orange juices consistently fall between 3.3 and 4.2 depending on variety and ripeness. This acidity level inhibits microbial growth, contributing to natural preservation while activating taste receptors responsible for sour perception. The pH remains well below neutral but avoids the extreme acidity of gastric fluids or strong mineral acids.
B) Between 6.0 and 7.0
This near-neutral range characterizes milk, saliva, and slightly acidic rainwater. Pure water maintains pH 7.0 at 25°C under standard conditions. Beverages in this range lack perceptible sourness and do not trigger significant acid-responsive taste sensations. Orange juice's pronounced tartness and chemical reactivity with bases contradict near-neutral pH behavior. Carbonated water without added acids approaches this range but lacks the organic acid profile of citrus juices.
C) Between 8.0 and 9.0
Basic solutions in this range include seawater, baking soda solutions, and egg whites. These substances feel slippery on skin due to saponification of surface oils. Basic environments promote hydrolysis reactions rather than the acid-catalyzed processes occurring in orange juice. Citrus fruits never achieve basic pH naturally; their metabolic pathways produce organic acids rather than base-generating compounds. Alkaline conditions would neutralize citric acid and eliminate characteristic citrus flavor.
D) Between 11.0 and 12.0
Strongly basic solutions at this pH include ammonia cleaners, lime water, and concentrated sodium bicarbonate solutions. These substances cause skin irritation and readily hydrolyze proteins and lipids. No common food or beverage maintains this pH due to palatability and safety constraints. Orange juice's acid content would require substantial neutralization with strong base to reach this range, fundamentally altering its chemical identity and sensory properties beyond recognition as juice.
Conclusion
The organic acid profile of orange juice, dominated by citric acid, establishes a pH firmly within the 3.0-4.0 range through partial dissociation in aqueous solution. This moderate acidity defines sensory characteristics, microbial stability, and chemical reactivity distinctive to citrus beverages. Measurements across commercial and freshly squeezed samples consistently validate this pH interval. Values approaching neutrality or basicity would indicate dilution, neutralization, or spoilage rather than authentic orange juice composition.

Fifty-six kg of a radioactive substance has a half-life of 12 days. How many days will it take the substance to decay naturally to only 7 kg?
A.
8
B. 12
C. 36
D. 48
Rationale
Thirty-six days are required for the radioactive substance to decay from 56 kg to 7 kg. This duration equals three half-lives of the 12-day decay cycle, with mass halving sequentially: 56 kg ? 28 kg ? 14 kg ? 7 kg. Each 12-day interval reduces the remaining quantity by half through first-order radioactive decay kinetics.
A) 8
Eight days represents two-thirds of a single half-life period. After this duration, approximately 35 kg of the substance would remain based on exponential decay calculations. The mass reduction falls far short of the three halving steps needed to reach 7 kg. Insufficient time elapses for even one complete half-life transition, leaving the sample well above the target mass.
B) 12
Twelve days constitutes exactly one half-life for this substance. The initial 56 kg decays to 28 kg after this interval, representing only the first halving step. Two additional half-lives remain necessary to achieve the 7 kg target. The 28 kg residue exceeds the goal by a factor of four, confirming incomplete decay progression.
C) 36
Thirty-six days equals three consecutive half-lives (3 x 12 days). The decay sequence progresses precisely: 56 kg ? 28 kg after 12 days, 28 kg ? 14 kg after 24 days, and 14 kg ? 7 kg after 36 days. This three-step halving reduces the original mass by a factor of eight (2³), matching the 56:7 ratio exactly. The calculation aligns with first-order decay mathematics where remaining fraction equals (1/2)¿ for n half-lives.
D) 48
Forty-eight days represents four complete half-lives. The decay progression continues beyond the target: 56 kg ? 28 kg ? 14 kg ? 7 kg ? 3.5 kg. After 36 days the substance reaches exactly 7 kg, but an additional 12 days further reduces it to 3.5 kg. This duration overshoots the specified endpoint by one full half-life, yielding only half the desired remaining mass.
Conclusion
Radioactive decay follows predictable halving intervals defined by half-life duration. Reducing 56 kg to 7 kg requires exactly three halving steps, as 56 /2³ = 7. Multiplying three half-lives by the 12-day period yields 36 days as the precise decay timeframe. Shorter durations leave excess mass while longer periods decay past the target, confirming 36 days as the mathematically exact solution.

You contain two odorous gases in vials with porous plugs. Gas A has twice the mass of Gas B. Which observation is most likely?
A.
You will smell Gas A before you smell Gas B.
B. You will smell Gas B before you smell Gas A.
C. You will smell Gas A but not Gas B.
D. You will smell Gas B but not Gas A.
Rationale
Gas B will be detected before Gas A due to its lower molar mass. According to Graham's Law of diffusion, the rate at which a gas diffuses is inversely proportional to the square root of its molar mass. Since Gas A has twice the molar mass of Gas B, Gas B diffuses approximately ?2 (about 1.4) times faster, reaching the observer's nose more quickly.
A) You will smell Gas A before you smell Gas B
This would imply that the heavier gas diffuses faster, contradicting the kinetic molecular theory. Heavier molecules move more slowly at the same temperature because their average speed is inversely related to the square root of mass. Gas A's greater inertia results in slower motion and delayed arrival through the porous plug. No physical mechanism allows a heavier gas to outpace a lighter one under identical conditions.
B) You will smell Gas B before you smell Gas A
Graham's Law states: Rate ˆ 1/?M. If MA = 2MB, then RateB/RateA = ?(MA/MB) = ?2 ‰ˆ 1.41. Thus, Gas B effuses and diffuses about 41% faster than Gas A. In an experiment with porous plugs, this difference in molecular speed translates directly into earlier detection of the lighter gas. Both gases are odorous, so detectability is not an issue, only timing differs.
C) You will smell Gas A but not Gas B
The question explicitly states both gases are odorous, meaning each produces a detectable scent when it reaches the olfactory receptors. Diffusion occurs for all gases, regardless of mass; only the rate varies. Gas B, being lighter, diffuses readily and will certainly be smelled. This option falsely assumes one gas is undetectable or non-diffusing, which contradicts the premise.
D) You will smell Gas B but not Gas A
Although Gas A diffuses more slowly, it still moves through the porous plug over time. Given sufficient duration, both gases will be detected. The observation concerns which is smelled first, not whether one is imperceptible. Gas A's higher mass delays, but does not prevent, its diffusion. This choice incorrectly implies complete absence of diffusion for the heavier gas.
Conclusion
Molecular diffusion rates depend on molar mass, with lighter gases moving faster under identical conditions. Graham's Law quantitatively predicts that Gas B, with half the molar mass of Gas A, will diffuse significantly faster and be smelled first. Both gases are odorous and will eventually be detected, but timing favors the lighter species. This principle underlies real-world phenomena such as the rapid spread of ammonia compared to heavier vapors like chlorine.

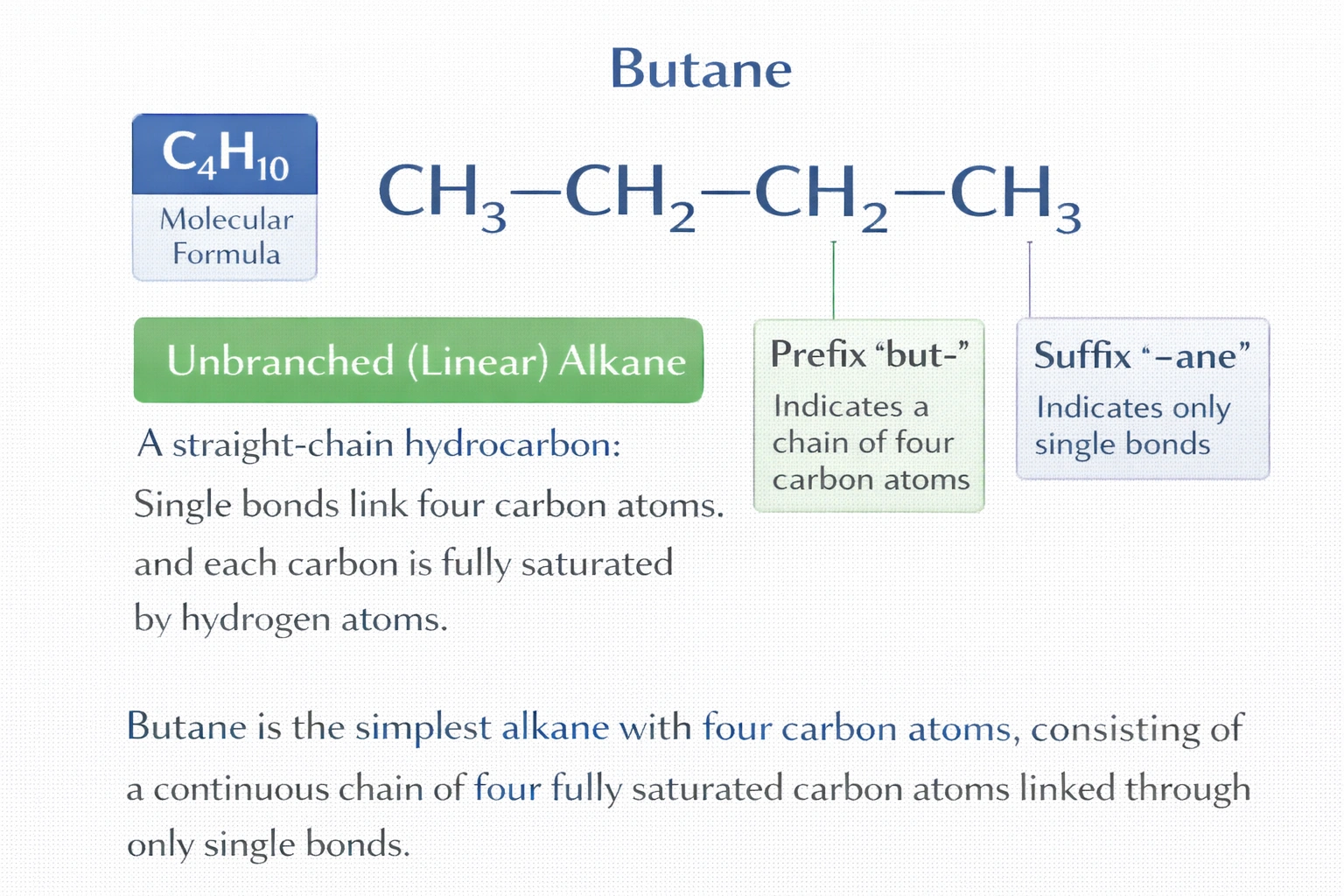
What is the name of the compound CH?-CH?-CH?-CH??
A.
Cyclobutane
B. Butane
C. Butene
D. Butyne
Rationale
The compound CH?-CH?-CH?-CH? is named butane. It is a straight-chain alkane containing four carbon atoms connected exclusively by single covalent bonds, with each carbon saturated by hydrogen atoms to fulfill the tetravalency rule. The prefix "but-" denotes four carbon atoms, and the suffix "-ane" indicates the absence of double or triple bonds, confirming its classification as a saturated hydrocarbon.
A) Cyclobutane
Cyclobutane has the molecular formula C?H‚ˆ and features a closed ring of four carbon atoms, introducing significant ring strain due to bond angles deviating from the ideal 109.5°. In contrast, the given structure is acyclic and linear, with the formula C?H‚‚€, two hydrogen atoms more than cyclobutane, confirming it is not cyclic. Ring formation requires loss of two hydrogens compared to the straight-chain alkane, which is absent here.
B) Butane
Butane (specifically n-butane) consists of an unbranched chain of four sp³-hybridized carbon atoms, each bonded to neighboring carbons and hydrogens via sigma bonds. Its molecular formula is C?H‚‚€, consistent with the general alkane formula C‚™H?‚™‚Š?. All bond angles approximate 109.5°, and free rotation occurs around C-C bonds. This structure matches the IUPAC naming convention for saturated, straight-chain hydrocarbons: "but-" for four carbons and "-ane" for single bonds only.
C) Butene
Butene refers to any C?H‚ˆ isomer containing at least one carbon-carbon double bond (e.g., 1-butene or 2-butene). The presence of a double bond reduces the hydrogen count by two compared to butane. The given structure shows only single bonds and has the formula C?H‚‚€, ruling out unsaturation. Alkenes like butene undergo addition reactions (e.g., with Br?), whereas butane does not, further distinguishing the two classes.
D) Butyne
Butyne describes C?H‚† isomers featuring a carbon-carbon triple bond (e.g., 1-butyne). Triple bonds reduce the hydrogen count by four relative to butane. The provided structure lacks any multiple bonds and contains no sp-hybridized carbons. Alkynes are more reactive than alkanes and exhibit distinct spectroscopic signatures (e.g., IR absorption near 2100 cm?²), none of which apply to the saturated molecule shown.
Conclusion
The compound is a saturated, straight-chain hydrocarbon with four carbon atoms and only single bonds. According to IUPAC nomenclature, this corresponds unambiguously to butane. Cyclobutane, butene, and butyne differ in structure, bonding, and molecular formula, making them incorrect. Recognition of the "-ane" suffix as indicative of single bonds is essential for accurate hydrocarbon identification.
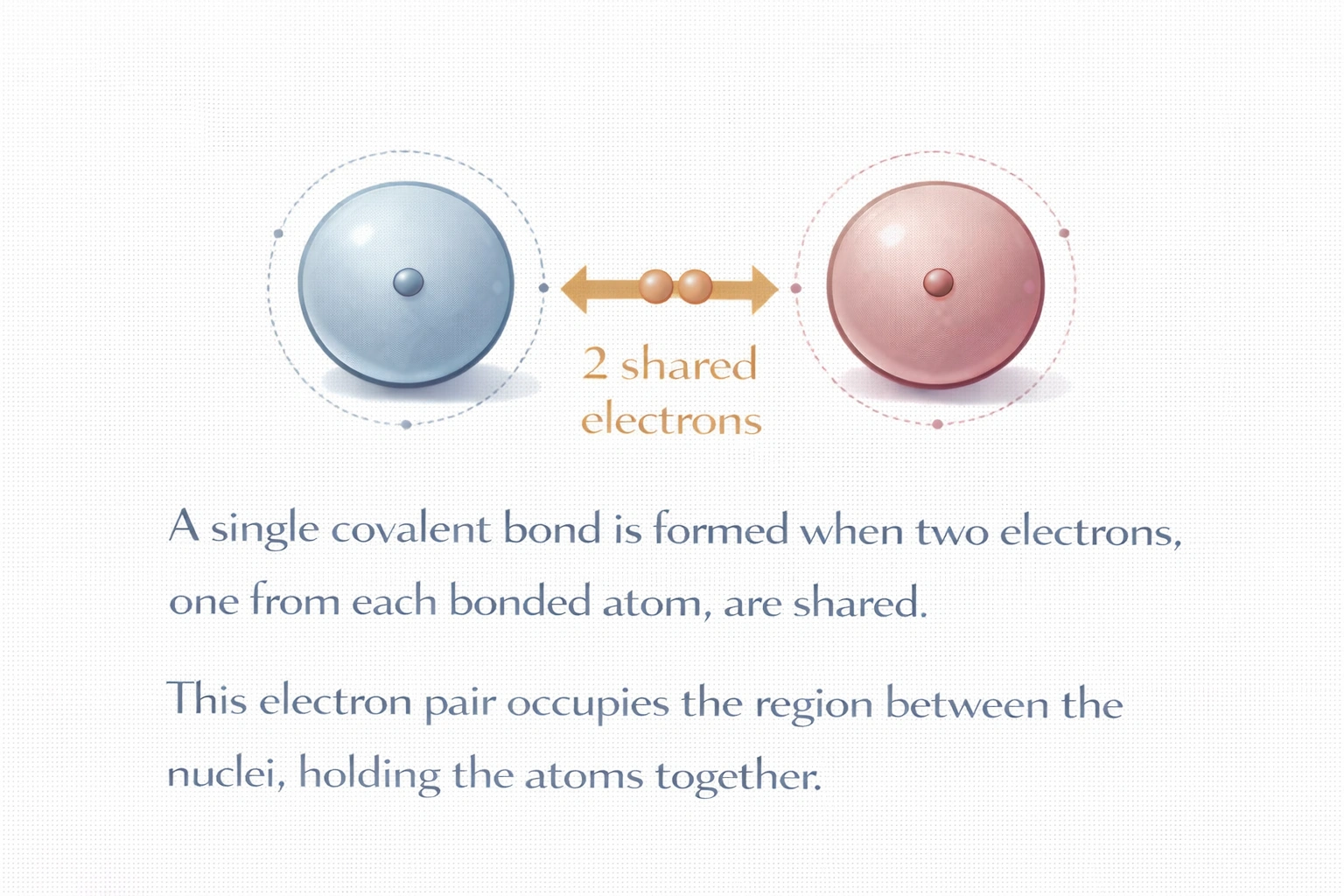
How many electrons are shared in a single covalent bond?
A.
1
B. 2
C. 3
D. 4
Rationale
A single covalent bond involves the sharing of two electrons. This electron pair consists of one electron contributed by each bonded atom, forming a stable sigma (σ) bond that satisfies the octet rule for many elements. The shared pair occupies the region between the two nuclei, creating electrostatic attraction that holds the atoms together in a molecule.
A) 1
A single electron cannot form a conventional covalent bond. Species with unpaired electrons, such as free radicals, are highly reactive and unstable due to incomplete valence shells. Stable covalent bonding requires paired electrons to achieve lower energy states and fulfill bonding requirements under the octet or duet rule. One-electron "bonds" exist only in exotic contexts like molecular hydrogen cation (H??), not in standard chemical compounds.
B) 2
A single covalent bond is defined as the mutual sharing of two electrons, one from each atom, forming an electron pair localized between the nuclei. This arrangement appears in diatomic molecules like H?, Cl?, and in organic frameworks such as C-H or C-C bonds. The bond order is 1, and the interaction is typically a sigma bond formed by head-on orbital overlap. This two-electron sharing is the foundation of Lewis theory and valence bond theory.
C) 3
Three shared electrons would imply an odd-electron system, resulting in a radical species rather than a stable bond. No common neutral molecule features a three-electron covalent bond as its primary linkage. While three-electron interactions can occur in certain excited states or transient ions (e.g., He??), they are weak and unstable compared to two-electron bonds. Standard covalent bonding does not involve odd numbers of shared electrons.
D) 4
Four shared electrons constitute a double bond, not a single bond. Double bonds consist of one sigma and one pi (Ï€) bond, as seen in O? (O=O) or ethene (H?C=CH?). The question specifically asks about a single covalent bond, which by definition involves only one bonding pair. Confusing double and single bonds misrepresents fundamental bonding concepts and bond-order relationships.
Conclusion
The single covalent bond is universally defined as a two-electron shared interaction between two atoms. This principle underpins molecular structure prediction, Lewis dot diagrams, and reactivity patterns across chemistry. Whether in elemental gases or complex biomolecules, every single bond, be it H-H, C-C, or N-H, involves exactly two shared electrons. Thus, B) 2 is the correct and definitive answer.
What is the molarity of a solution containing 0.45 moles of NaCl in 4 liters?
A.
0.11 M NaCl
B. 0.45 M NaCl
C. 1.8 M NaCl
D. 8.9 M NaCl
Rationale
The molarity of the solution is 0.11 M NaCl. Molarity is defined as moles of solute divided by liters of solution. With 0.45 moles of NaCl dissolved in 4.0 liters of solution, the calculation yields 0.45 mol /4.0 L = 0.1125 M, which rounds to 0.11 M when expressed with two significant figures, consistent with the precision of the given volume (4 L, interpreted as having one significant figure, though commonly treated as exact in such contexts).
A) 0.11 M NaCl
This result follows directly from the molarity formula:
Molarity = moles of solute / liters of solution
Molarity = 0.45 / 4.0 = 0.1125 ‰ˆ 0.11 M
The value reflects appropriate rounding and correct application of concentration principles. It represents a dilute saline solution, typical in laboratory or physiological contexts.
B) 0.45 M NaCl
This option incorrectly uses the mole quantity as the molarity without accounting for solution volume. Molarity is an intensive property that depends on both amount and volume; reporting 0.45 M would only be valid if the volume were exactly 1 liter. In 4 liters, the same number of moles produces a lower concentration.
C) 1.8 M NaCl
This arises from mistakenly multiplying moles by volume (0.45 x 4 = 1.8) instead of dividing. Such an error inverts the fundamental relationship between concentration, amount, and volume. A 1.8 M NaCl solution would require 7.2 moles in 4 liters, far more than provided.
D) 8.9 M NaCl
This value lacks any plausible arithmetic connection to the given numbers. It may stem from misreading decimals or applying unrelated formulas (e.g., involving molar mass). The solubility limit of NaCl in water is approximately 6.1 M at 20°C, making 8.9 M physically impossible under standard conditions.
Conclusion
Molarity calculations require strict adherence to the formula M = n/V. Here, 0.45 moles distributed over 4 liters yields a low-concentration solution of 0.11 M. The other options result from conceptual errors, ignoring volume, inverting operations, or introducing non-physical values. Thus, A) 0.11 M NaCl is the only chemically and mathematically valid answer.

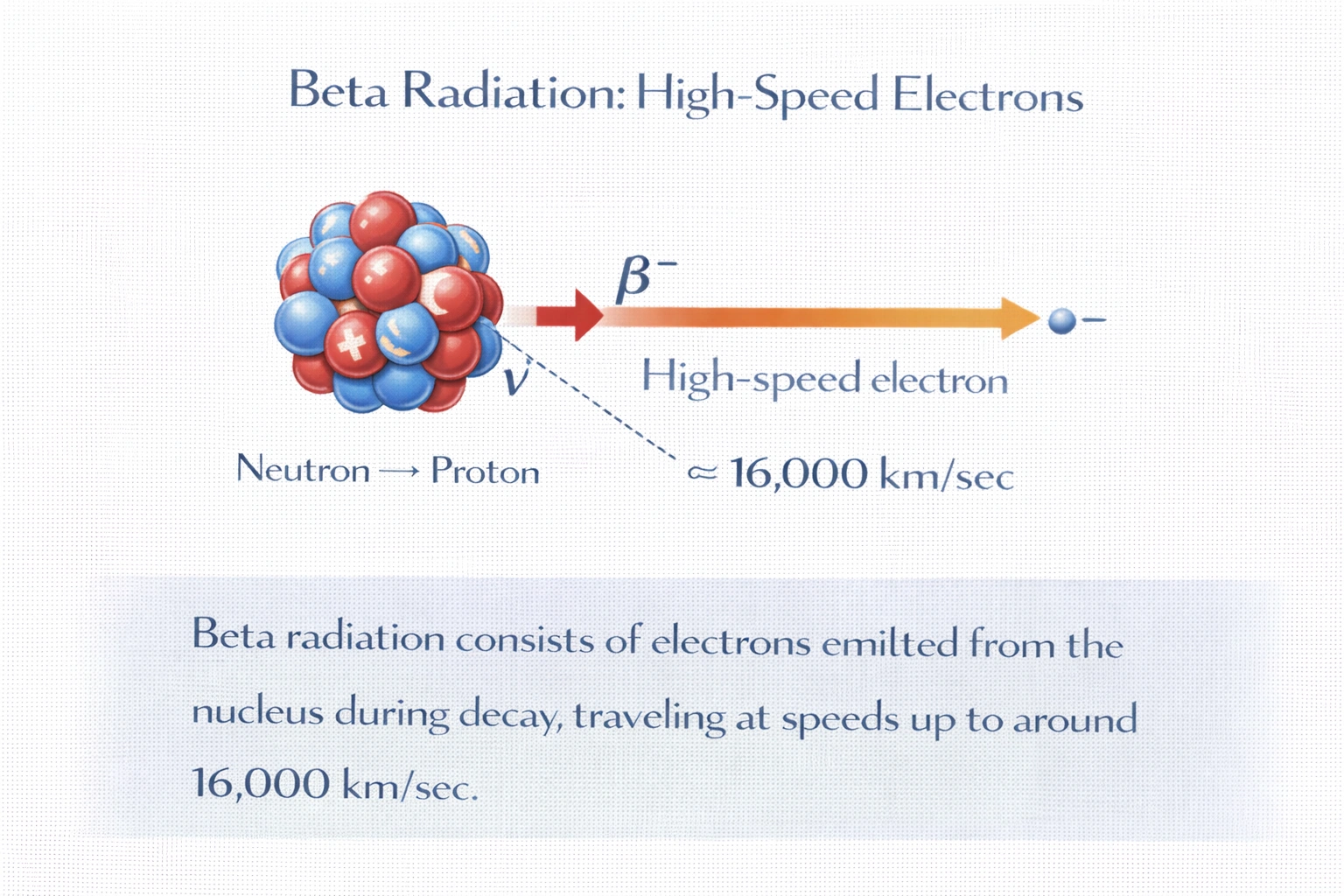
What form of radiation is composed of electrons traveling at around 16,000 km/sec?
A.
Alpha radiation
B. Beta radiation
C. Gamma radiation
D. Delta radiation
Rationale
Beta radiation consists of high-speed electrons emitted from the nucleus during radioactive decay. These beta particles (β?) are ejected when a neutron transforms into a proton within an unstable nucleus, releasing an electron and an antineutrino. Typical beta particle velocities range up to a significant fraction of the speed of light; 16,000 km/sec corresponds to approximately 5.3% of light speed (c ‰ˆ 300,000 km/sec), which aligns with lower-energy beta emissions observed in many isotopes.
A) Alpha radiation
Alpha radiation comprises helium-4 nuclei, each containing two protons and two neutrons, carrying a +2 charge. These particles are massive (~7,300 times the electron mass) and typically travel at 5-10% of light speed (15,000-30,000 km/sec), but they are not electrons. Their large mass and double positive charge result in high ionization but low penetration. The description of "electrons" definitively excludes alpha radiation.
B) Beta radiation
Beta radiation involves high-energy electrons (β?) or positrons (β?) emitted directly from the atomic nucleus. Despite electrons normally residing outside the nucleus, beta decay creates them via weak nuclear force interactions (n ? p + e? + ν̄). Speeds vary by isotope: for example, tritium β? particles average ~5,700 km/sec, while phosphorus-32 can emit electrons exceeding 90,000 km/sec. A velocity of 16,000 km/sec falls well within the typical beta energy spectrum, confirming this as the correct match.
C) Gamma radiation
Gamma rays are high-energy photons, electromagnetic waves with no mass or charge, that travel precisely at the speed of light (299,792 km/sec). They originate from nuclear de-excitation, not particle emission. Since gamma radiation involves no electrons, it cannot match a description specifying "electrons traveling at 16,000 km/sec." This option confuses wave-like radiation with particulate matter.
D) Delta radiation
"Delta radiation" is not a recognized form of nuclear radiation. The standard categories are alpha (helium nuclei), beta (electrons/positrons), and gamma (photons). Delta rays refer in specialized contexts to secondary electrons produced by ionizing radiation, but they are not a primary decay product. This term is likely included as a distractor and has no basis as a fundamental radiation type.
Conclusion
The question specifies electrons moving at high speed, a defining trait of beta radiation. Alpha particles are heavy and positively charged, gamma rays are massless photons, and delta radiation is not a standard classification. Only beta decay emits electrons from the nucleus at velocities matching the given value. Thus, B) Beta radiation is the correct and scientifically precise answer.
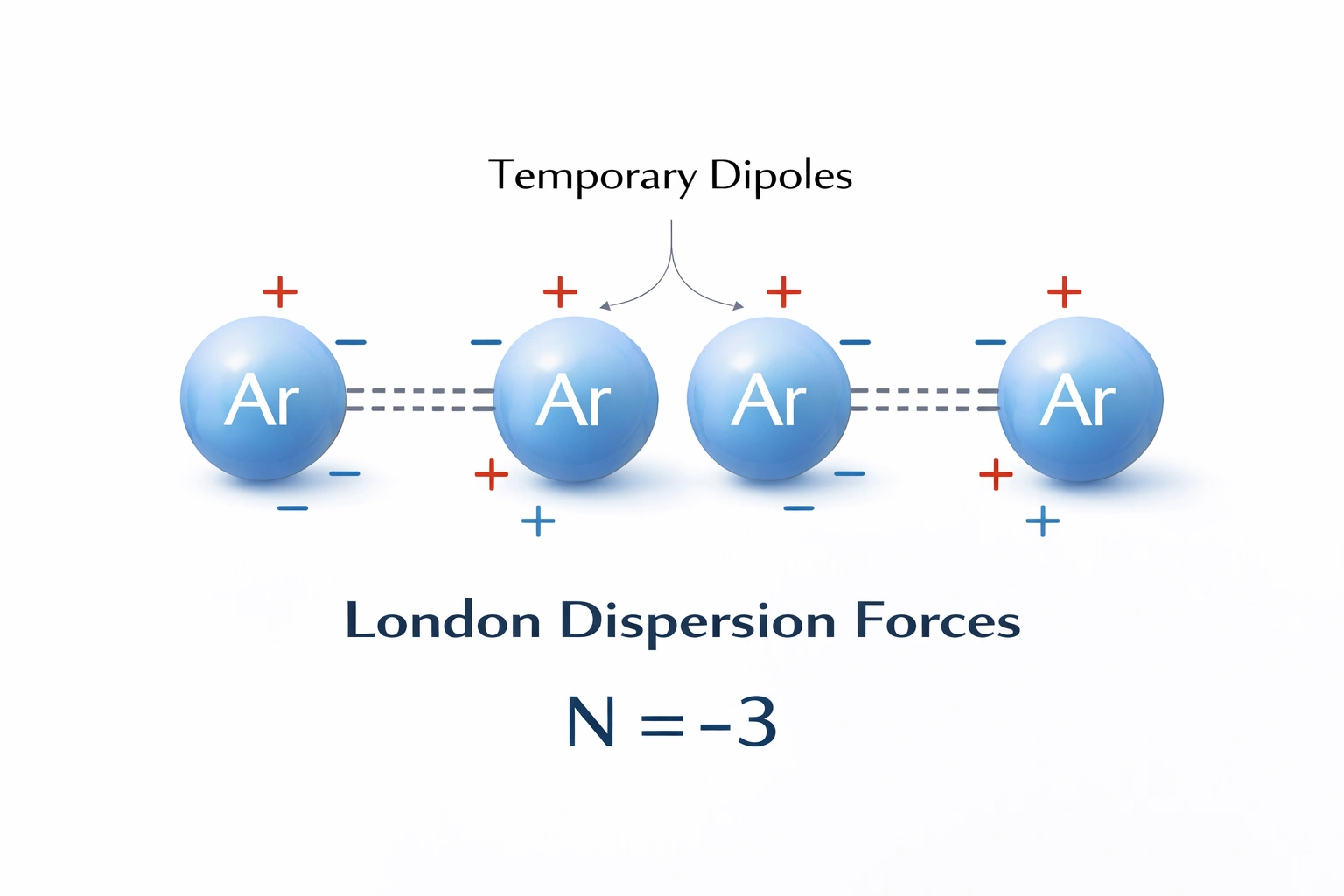
Which of these intermolecular forces would have the lowest boiling point?
A.
Dipole-dipole interaction
B. London dispersion force
C. Keesom interaction
D. Hydrogen bonding
Rationale
Substances dominated by London dispersion forces exhibit the lowest boiling points among the listed intermolecular interactions. These forces arise from temporary, instantaneous dipoles induced by electron motion and are the weakest type of van der Waals attraction. Because they require minimal energy to overcome during phase transitions, nonpolar molecules relying solely on dispersion forces, such as noble gases or hydrocarbons like methane, boil at very low temperatures.
A) Dipole-dipole interaction
Dipole-dipole forces occur between polar molecules with permanent charge separation, such as acetone or chloroform. These electrostatic attractions are stronger than London dispersion forces for molecules of comparable size, leading to higher boiling points. For example, acetone (58 g/mol, dipole-dipole) boils at 56°C, while butane (58 g/mol, dispersion only) boils at -1°C. This increased intermolecular cohesion rules out dipole-dipole as the weakest force.
B) London dispersion force
London dispersion forces are universal but dominant only in nonpolar substances like helium, neon, methane, or octane. Their strength depends on molecular surface area and polarizability, but even for larger molecules, they remain weaker than permanent dipole interactions or hydrogen bonds. Helium (He), governed solely by dispersion forces, has the lowest boiling point of any element (-269°C). This weakness directly correlates with minimal energy required to transition from liquid to gas.
C) Keesom interaction
Keesom forces describe the orientation-dependent attraction between permanent dipoles, essentially a subset of dipole-dipole interactions. Named after physicist Willem Keesom, they contribute to the overall van der Waals forces in polar molecules but are still significantly stronger than dispersion forces alone. Molecules exhibiting Keesom interactions, such as HCl, have higher boiling points (-85°C) than nonpolar analogs of similar mass (e.g., F?, bp -188°C).
D) Hydrogen bonding
Hydrogen bonding is a strong dipole-dipole interaction occurring when hydrogen is bonded to N, O, or F. It dramatically elevates boiling points: water (18 g/mol) boils at 100°C due to extensive hydrogen bonding, whereas neon (20 g/mol, dispersion only) boils at -246°C. This strength makes hydrogen-bonded substances the least volatile among the options, definitively excluding this choice.
Conclusion
Boiling point elevation directly reflects the strength of intermolecular forces. London dispersion forces, being the weakest and present in all molecules but dominant only in nonpolar species, result in the lowest energy requirement for
school,course,exam_set,exam_name,extract,question,choiceA,choiceB,choiceC,choiceD,choiceE,choiceF,choiceG,correct_answer,rationale,question_type,image_url,alt_text
HESI A2,HESI A2,Chemistry,HESI A2 Chemistry Practice,48. Concentrated HCl has a molarity of 12.0. What volume of concentrated HCl should be used to prepare 500 mL of a 3.00 M HCl solution?
Sodium's ground state electron configuration may be represented as 1ss?2ss?2p?3s?. How many energy levels does sodium have?
A.
1
B. 2
C. 3
D. 6
Rationale
Sodium has three principal energy levels. Its ground state electron configuration, 1s²2s²2p?3s², shows electrons occupying orbitals with principal quantum numbers n = 1 (1s²), n = 2 (2s²2p?), and n = 3 (3s²). The highest occupied energy level is n = 3, confirming that sodium utilizes three distinct electron shells.
A) 1
This would imply all electrons reside in the first shell (n = 1), which can hold only 2 electrons (1ss²). Sodium has 11 electrons, so this is impossible. Even helium (2 electrons) fills only n = 1; sodium, with far more electrons, must occupy higher shells.
B) 2
The first two energy levels (n = 1 and n = 2) can hold up to 2 + 8 = 10 electrons. Sodium has 11 electrons, so the 11th electron must occupy the next available orbital, 3s, in the third energy level (n = 3). Thus, two levels are insufficient.
C) 3
Electrons are distributed as follows:
1. n = 1: 1ss² ? 2 electrons
2. n = 2: 2ss²2p? ? 8 electrons
3. n = 3: 3s² ? 1 electron
This matches sodium's atomic number (11) and confirms occupation of three principal energy levels. The period number of sodium (Period 3) also directly corresponds to its highest principal quantum number.
D) 6
This likely results from miscounting orbitals (e.g., 1s, 2s, three 2p orbitals, and 3s = 6 orbitals), but the question asks for energy levels (shells defined by n), not the number of orbitals or subshells. Sodium has only three such levels, regardless of orbital count.
Conclusion
The number of principal energy levels in an atom equals the highest principal quantum number (n) containing electrons in its ground state. For sodium (1ss²2ss²2p?3s²), n = 3 is the highest, so it has three energy levels. This aligns with its position in Period 3 of the periodic table. Thus, C) 3 is correct.

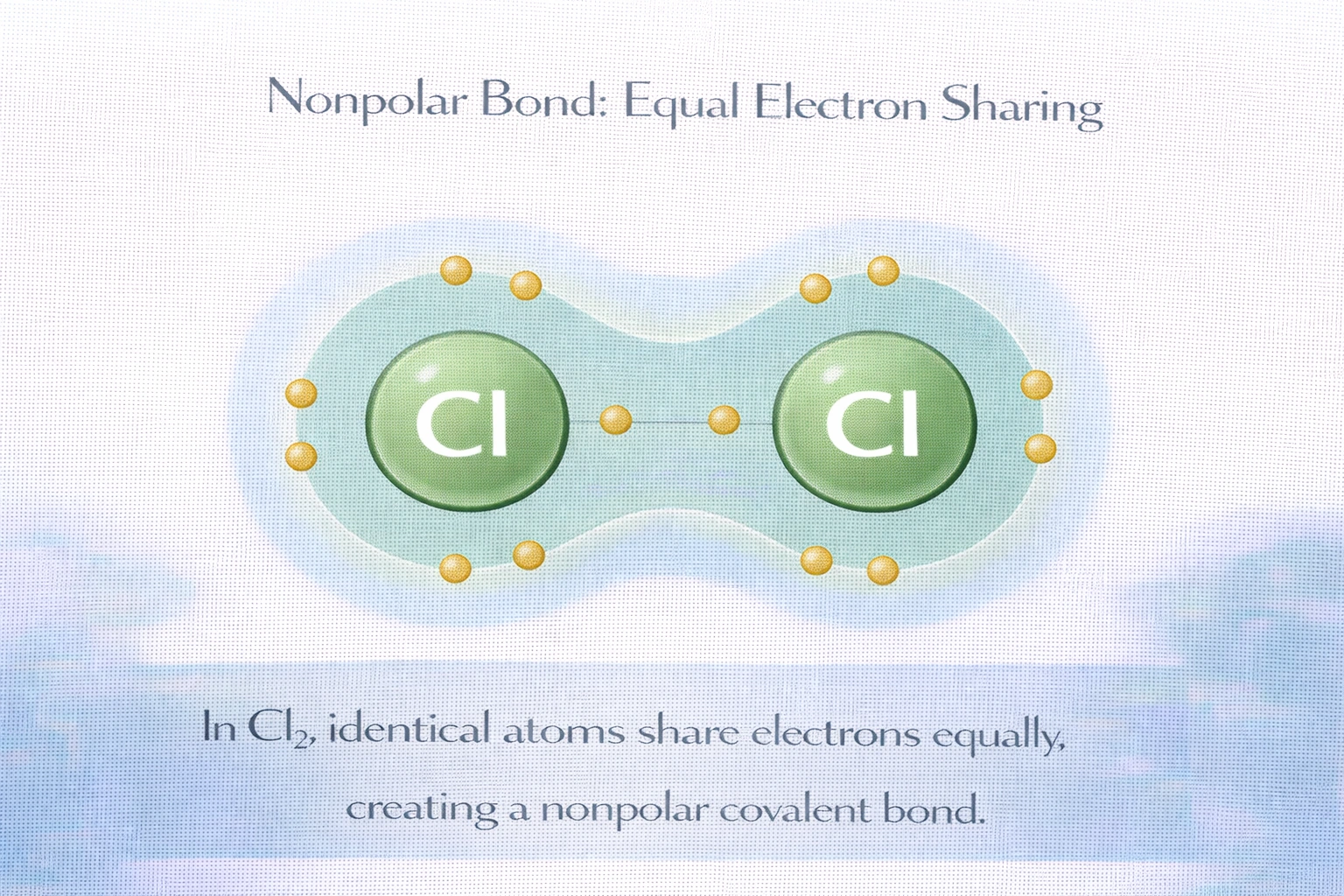
Which compound has a nonpolar bond in which the electrons are shared equally?
A.
H?O
B. NH?
C. Cl?
D. CH?
Rationale
Chlorine gas (Cl?) contains a nonpolar covalent bond where electrons are shared equally between two identical atoms. Since both chlorine atoms have identical electronegativity values (3.16 on the Pauling scale), the bonding electron pair distributes symmetrically with no charge separation. This equal sharing produces a nonpolar covalent bond characteristic of homonuclear diatomic molecules.
A) H?O
Water molecules contain polar covalent O-H bonds due to the significant electronegativity difference between oxygen (3.44) and hydrogen (2.20). The bonding electrons concentrate closer to oxygen, creating partial charges (δ? on O, δ? on H). The bent molecular geometry prevents dipole cancellation, resulting in an overall polar molecule.
B) NH?
Ammonia features polar covalent N-H bonds. Nitrogen (electronegativity 3.04) attracts bonding electrons more strongly than hydrogen (2.20), creating partial charges. The trigonal pyramidal geometry results in a net molecular dipole moment, making ammonia polar.
C) Cl?
Correct. Chlorine molecules consist of two identical chlorine atoms bonded together. Both atoms possess identical electronegativity values of 3.16. The shared electron pair occupies a symmetrical bonding orbital centered between the nuclei with no charge separation. Equal electron distribution prevents formation of partial charges or bond polarity. Cl? exhibits zero dipole moment and is the only option with truly nonpolar bonding.
D) CH?
Methane contains polar C-H bonds due to the electronegativity difference between carbon (2.55) and hydrogen (2.20). However, methane's symmetrical tetrahedral geometry causes individual bond dipoles to cancel, resulting in a nonpolar molecule overall. Despite the molecular nonpolarity, the individual C-H bonds themselves are slightly polar, not truly nonpolar bonds with equal electron sharing.
Conclusion
True nonpolar covalent bonds with perfectly equal electron sharing occur only between identical atoms. Among the options, only Cl? consists of two identical atoms with zero electronegativity difference, resulting in completely equal electron sharing. Thus, C) Cl? is the correct answer.
The molar mass of some gases is as follows: carbon monoxide, 28.00 g/mol; helium, 4.00 g/mol; nitrogen, 14.01 g/mol; and oxygen, 16.00 g/mol. Which would you expect to diffuse most rapidly?
A.
Carbon monoxide
B. Helium
C. Nitrogen
D. Oxygen
Rationale
Helium diffuses most rapidly among the listed gases because it has the lowest molar mass (4.00 g/mol). According to Graham's Law of Diffusion, the rate of diffusion of a gas is inversely proportional to the square root of its molar mass. Thus, lighter gases diffuse faster than heavier ones at the same temperature and pressure.
A) Carbon monoxide
CO has a molar mass of 28.00 g/mol. According to Graham's Law, heavier gases diffuse more slowly. This is the heaviest gas listed, so it would diffuse slowest, not fastest. This is incorrect.
B) Helium
Helium has a molar mass of only 4.00 g/mol, making it the lightest gas listed. According to Graham's Law of Diffusion, rate ˆ 1/?M. The lighter the gas, the faster it diffuses. Helium is about 7 times lighter than CO, so it diffuses about ?7 ‰ˆ 2.6 times faster. This is the correct answer.
C) Nitrogen
While nitrogen (14.01 g/mol) is lighter than CO and O?, it's still much heavier than helium. It would diffuse faster than CO or O?, but not as fast as helium. This is incorrect.
D) Oxygen
Oxygen (16.00 g/mol) is lighter than CO but heavier than helium and nitrogen. It would diffuse faster than CO but slower than helium and nitrogen. This is incorrect.
Conclusion
Diffusion speed is governed by molecular mass: lower mass ? higher average speed ? faster diffusion. Among CO (28), He (4), N? (14), and O? (16), helium is the lightest and therefore diffuses most rapidly. This principle explains why helium-filled balloons deflate faster than air-filled ones, helium escapes more quickly through tiny pores. Thus, B) Helium is the correct answer.

What is the product of combustion of a hydrocarbon in excess oxygen?
A.
Carbon dioxide and water
B. Naphthalene
C. Chlorine and bromine
D. Carbonium ions
Rationale
The combustion of a hydrocarbon in excess oxygen produces carbon dioxide and water. Hydrocarbons consist solely of carbon and hydrogen atoms, and when sufficient oxygen is present, complete oxidation occurs: carbon is fully converted to CO? and hydrogen to H?O. This reaction is highly exothermic and represents the ideal outcome for clean fuel combustion, as seen in natural gas (methane) or gasoline burning.
A) Carbon dioxide and water
Carbon dioxide and water are the exclusive products of complete hydrocarbon combustion in excess oxygen. The reaction follows the general form C‚“Hᵧ + (x + y/4) O? ? x CO? + (y/2) H?O. For example, methane burns as CH? + 2O? ? CO? + 2H?O. These stable, fully oxidized compounds result from the thermodynamically favored conversion of all carbon and hydrogen atoms under oxygen-rich conditions. This is the correct answer.
B) Naphthalene
Naphthalene (C‚‚€H‚ˆ) is a polycyclic aromatic hydrocarbon used as a fuel or chemical feedstock, not a combustion product. It would itself undergo combustion to produce CO? and H?O. No combustion process yields a more complex hydrocarbon as a final product; instead, molecular breakdown occurs. This option confuses reactant with product and is incorrect.
C) Chlorine and bromine
Chlorine and bromine are halogen elements not present in pure hydrocarbons, which contain only carbon and hydrogen. Their appearance in combustion products would require halogenated compounds (e.g., PVC), which are outside the scope of standard hydrocarbon combustion. In the absence of such elements, these substances cannot form and are irrelevant to the reaction. This is incorrect.
D) Carbonium ions
Carbonium ions (carbocations) are high-energy, transient intermediates in organic reaction mechanisms such as electrophilic substitution or elimination. They are unstable and do not persist under the high-temperature, oxidative conditions of combustion. Complete combustion drives reactions to thermodynamic endpoints, CO? and H?O, not reactive intermediates. This is incorrect.
Conclusion
Complete combustion of any hydrocarbon in excess oxygen yields only carbon dioxide and water, as all carbon and hydrogen atoms are fully oxidized. This principle underlies energy production, environmental science, and safety protocols for fuel use. Among the options, only A) Carbon dioxide and water accurately describes the products of this fundamental chemical process.
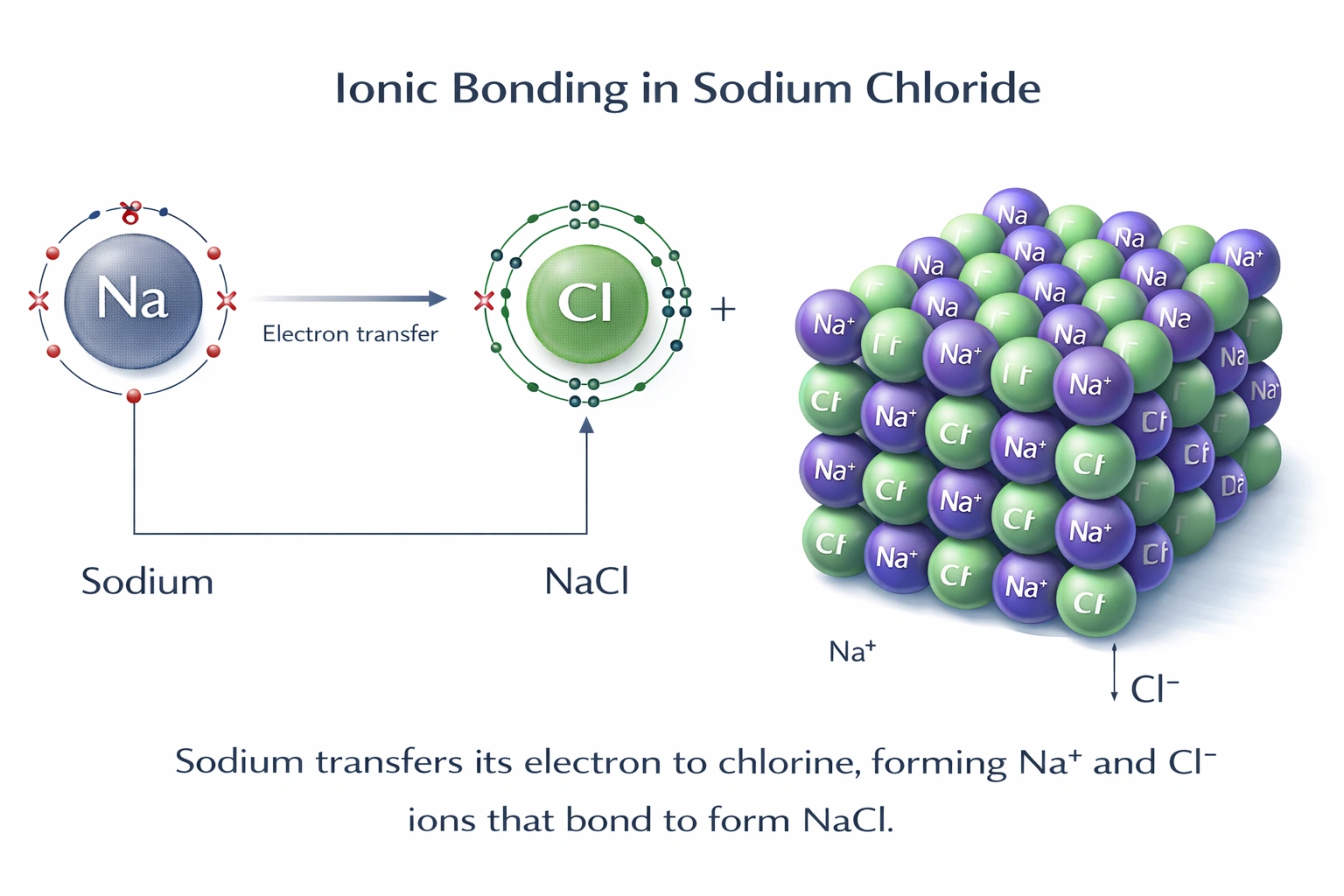
Which of the following compounds is ionic?
A.
NaCl
B. H?O
C. HCl
D. NH?
Rationale
Ionic compounds form through the complete transfer of electrons from a metal to a nonmetal, resulting in oppositely charged ions held together by strong electrostatic forces. Among the given options, only NaCl fits this definition, as it consists of sodium (a Group 1 metal) and chlorine (a halogen nonmetal), with an electronegativity difference of approximately 2.1, well above the typical threshold for ionic character.
A) NaCl
Sodium (Na) readily loses one valence electron to form Na?, while chlorine (Cl) gains that electron to form Cl?. The resulting ions arrange into a crystalline lattice stabilized by ionic bonds. This compound exhibits classic ionic properties: high melting point (801°C), solubility in water, and electrical conductivity in aqueous or molten states. It is unambiguously ionic.
B) H?O
Water is composed of hydrogen and oxygen, both nonmetals. The O-H bonds involve shared electrons, forming polar covalent bonds. Although water molecules are polar and capable of hydrogen bonding, no electron transfer occurs, and no discrete ions exist in pure water. Thus, it is a covalent molecular compound, not ionic.
C) HCl
Hydrogen chloride consists of two nonmetals. In the gaseous state, it exists as discrete HCl molecules with a polar covalent bond. While it ionizes in water to form H?O? and Cl?, the pure compound is covalent. Ionic classification applies to the substance itself, not its behavior in solution. Therefore, HCl is not an ionic compound.
D) NH?
This notation is chemically incomplete. "NH?" likely refers to the ammonium ion (NH??), which is a polyatomic cation, not a neutral compound. A stable ionic compound would be something like NH?Cl, but as written, "NH?" lacks a counterion and does not represent a valid chemical compound. Even if interpreted as NH??, it is an ion, not an ionic compound per se.
Conclusion
Ionic compounds require a metal and a nonmetal bonded via electron transfer. Only NaCl meets this criterion among the choices. Water and HCl are covalent molecules, and "NH?" is an incomplete or incorrect formula. Thus, A) NaCl is the correct answer.
A chemist takes 100 mL of a 0.40 M NaCl solution. She then dilutes it to 1 L. What is the concentration (molarity) of the new solution?
A.
0.04 M NaCl
B. 0.25 M NaCl
C. 0.40 M NaCl
D. 2.5 M NaCl
Rationale
When a solution is diluted, the number of moles of solute remains constant while the volume increases, resulting in a lower concentration. The chemist starts with 100 mL of a 0.40 M NaCl solution and dilutes it to a final volume of 1 L (1000 mL). Using the dilution equation M‚V‚ = M?V?, the new concentration is calculated as follows: (0.40 M)(100 mL) = M?(1000 mL), yielding M? = 0.04 M.
A) 0.04 M NaCl
This result correctly applies the dilution principle. The solution volume increases by a factor of 10 (from 100 mL to 1000 mL), so the concentration decreases by the same factor: 0.40 M /10 = 0.04 M. This maintains the same number of moles of NaCl (0.04 mol) before and after dilution, satisfying conservation of mass. This is the correct answer.
B) 0.25 M NaCl
This value does not correspond to any standard dilution calculation. It may arise from misapplying ratios or confusing molarity with other concentration units. The correct 10-fold dilution cannot produce 0.25 M from 0.40 M. This is incorrect.
C) 0.40 M NaCl
This implies no change in concentration despite a tenfold increase in volume, violating the fundamental principle of dilution. If concentration remained constant, the amount of solute would have to increase tenfold, which did not occur. This is incorrect.
D) 2.5 M NaCl
This concentration is higher than the original, which would require evaporation or addition of solute, not dilution. Diluting a solution can never increase its molarity. This contradicts basic solution chemistry and is incorrect.
Conclusion
Dilution reduces concentration proportionally to the volume increase. A 10-fold volume increase (100 mL ? 1000 mL) results in a 10-fold concentration decrease (0.40 M ? 0.04 M). The calculation adheres to the conservation of moles and the dilution formula M‚V‚ = M?V?. Therefore, A) 0.04 M NaCl is the correct answer.

What is the charge of a gamma ray?
A.
-1
B. 1
C. 2
D. No charge
Rationale
Gamma rays are high-energy photons with no charge and a form of electromagnetic radiation, not particles with mass or electric charge. They originate from nuclear transitions when an excited nucleus releases excess energy. Unlike alpha or beta radiation, gamma rays do not consist of matter and therefore carry no electric charge.
A) -1
This charge corresponds to beta-minus (β?) particles, which are high-speed electrons emitted during radioactive decay. Gamma rays are not electrons and possess no charge. This option confuses beta radiation with gamma radiation and is incorrect.
B) +1
A +1 charge describes positrons (β?), the antimatter counterparts of electrons, emitted in certain types of radioactive decay. Gamma rays are neutral photons and do not carry positive charge. This misattributes particle properties to electromagnetic radiation and is incorrect.
C) +2
This charge identifies alpha particles, which are helium nuclei composed of two protons and two neutrons. Alpha radiation involves massive, charged particles, whereas gamma rays are massless and uncharged. This option incorrectly applies nuclear particle characteristics to gamma radiation and is incorrect.
D) No charge
Correct. Gamma rays are photons, packets of pure energy within the electromagnetic spectrum. Like visible light, X-rays, and radio waves, they have zero rest mass and zero electric charge. Their neutrality allows them to penetrate matter deeply without deflection by electric or magnetic fields.
Conclusion
Gamma radiation consists of uncharged, massless photons emitted from atomic nuclei. Unlike alpha (+2) and beta (±1) particles, gamma rays carry no electric charge. Therefore, D) No charge is the correct answer.

Which of these intermolecular forces might represent attraction between atoms of a noble gas?
A.
Dipole-dipole interaction
B. London dispersion force
C. Keesom interaction
D. Hydrogen bonding
Rationale
Noble gases consist of individual atoms with completely filled electron shells, resulting in symmetrical charge distributions and no permanent dipole moments. As monatomic, nonpolar species, they lack the structural features required for dipole-dipole interactions, hydrogen bonding, or any permanent electrostatic attraction. The only intermolecular forces present between noble gas atoms are London dispersion forces, which arise from temporary fluctuations in electron density that induce instantaneous dipoles, leading to weak, short-lived attractions.
A) Dipole-dipole interaction
Dipole-dipole forces occur between molecules with permanent dipoles due to differences in electronegativity and asymmetric geometry (e.g., HCl or H?O). Noble gases are monatomic and nonpolar, with no bond polarity or molecular asymmetry. Thus, they cannot exhibit permanent dipoles or dipole-dipole interactions. This is incorrect.
B) London dispersion force
Correct. London dispersion forces are universal and operate in all atoms and molecules, but they are the only intermolecular force for noble gases. These forces result from momentary imbalances in electron distribution, creating transient dipoles that induce complementary dipoles in neighboring atoms. Although weak, these attractions allow noble gases to liquefy at very low temperatures (e.g., argon boils at -186°C). The strength increases with atomic size due to greater polarizability (He < Ne < Ar < Kr < Xe).
C) Keesom interaction
Keesom forces are a subset of dipole-dipole interactions involving the orientation-dependent attraction between permanent dipoles. Since noble gas atoms have no permanent dipole moment, Keesom interactions do not apply. This term is relevant only for polar molecules and is incorrect here.
D) Hydrogen bonding
Hydrogen bonding requires a hydrogen atom covalently bonded to nitrogen, oxygen, or fluorine, elements with high electronegativity and lone pairs. Noble gases are inert, monatomic, and form no covalent bonds under standard conditions. They possess no hydrogen and no N/O/F atoms, making hydrogen bonding impossible. This is incorrect.
Conclusion
Among all intermolecular forces, only London dispersion forces operate between noble gas atoms due to their nonpolar, monatomic nature. These weak attractions explain the physical behavior of noble gases, including their low boiling points and ability to condense at cryogenic temperatures. Therefore, B) London dispersion force is the correct answer.
Why does fluorine have a higher ionization energy than oxygen?
A.
Fluorine has a smaller number of neutrons.
B. Fluorine has a larger number of neutrons.
C. Fluorine has a smaller nuclear charge.
D. Fluorine has a larger nuclear charge.
Rationale
Fluorine has a higher ionization energy than oxygen because it possesses a larger nuclear charge. Both elements are in Period 2, but fluorine (atomic number 9) has 9 protons, while oxygen (atomic number 8) has 8 protons. The increased positive charge in fluorine's nucleus exerts a stronger electrostatic pull on its electrons, particularly the valence electrons, making them more difficult to remove. Although both atoms have similar electron shielding (same principal quantum level and inner-shell configuration), fluorine's greater nuclear charge results in a higher effective nuclear charge, leading to a higher ionization energy.
A) Fluorine has a smaller number of neutrons
Neutrons contribute to atomic mass and nuclear stability but do not influence electron-nucleus attraction, which governs ionization energy. Ionization energy depends on proton count and electron configuration, not neutron number. This is irrelevant and incorrect.
B) Fluorine has a larger number of neutrons
While fluorine-19 (the only stable isotope) has 10 neutrons compared to oxygen-16's 8, this difference affects mass, not electromagnetic interactions. Ionization energy is determined by the attraction between protons and electrons, not neutrons. This option misattributes the cause and is incorrect.
C) Fluorine has a smaller nuclear charge
This is factually false. Nuclear charge equals the number of protons. Fluorine (Z = 9) has a greater nuclear charge than oxygen (Z = 8). A smaller nuclear charge would imply weaker electron binding and lower ionization energy, opposite to the observed trend. This is incorrect.
D) Fluorine has a larger nuclear charge
Correct. The additional proton in fluorine increases the effective nuclear charge felt by valence electrons. Although oxygen has a slightly lower electron-electron repulsion in its 2p´ configuration (with two unpaired electrons), fluorine's stronger nuclear pull dominates, resulting in a higher first ionization energy (1681 kJ/mol for F vs. 1314 kJ/mol for O).
Conclusion
Ionization energy generally increases across a period due to increasing nuclear charge with relatively constant shielding. Fluorine's extra proton enhances electron-nucleus attraction, making electron removal more energy-intensive than in oxygen. Therefore, D) Fluorine has a larger nuclear charge is the correct answer.

HESI A2 Exams
Biology Quizzes
3 Practice Tests
Biology Quizzes
3 Practice Tests
Anatomy Quizzes
3 Practice Tests
Anatomy Quizzes
3 Practice Tests
Reading Quizzes
4 Practice Tests
Reading Quizzes
4 Practice Tests
Grammar Quizzes
3 Practice Tests
Grammar Quizzes
3 Practice Tests
Vocabulary Quizzes
3 Practice Tests
Vocabulary Quizzes
3 Practice Tests
Physics Quizzes
3 Practice Tests
Physics Quizzes
3 Practice Tests
HESI Quizzes
3 Practice Tests
HESI Quizzes
3 Practice Tests