CHEMISTRY HESI A2 PRACTICE TEST
This Chemistry HESI A2 Practice Test evaluates your understanding of essential chemistry concepts tested on the exam. The questions are structured to match the cognitive level and wording of actual HESI A2 questions, helping you build confidence in calculations, chemical reactions, and atomic theory.
Topics Covered
Atomic Structure
Periodic Table
Chemical Reactions
Acids and Bases
Solutions and Measurements
00:00
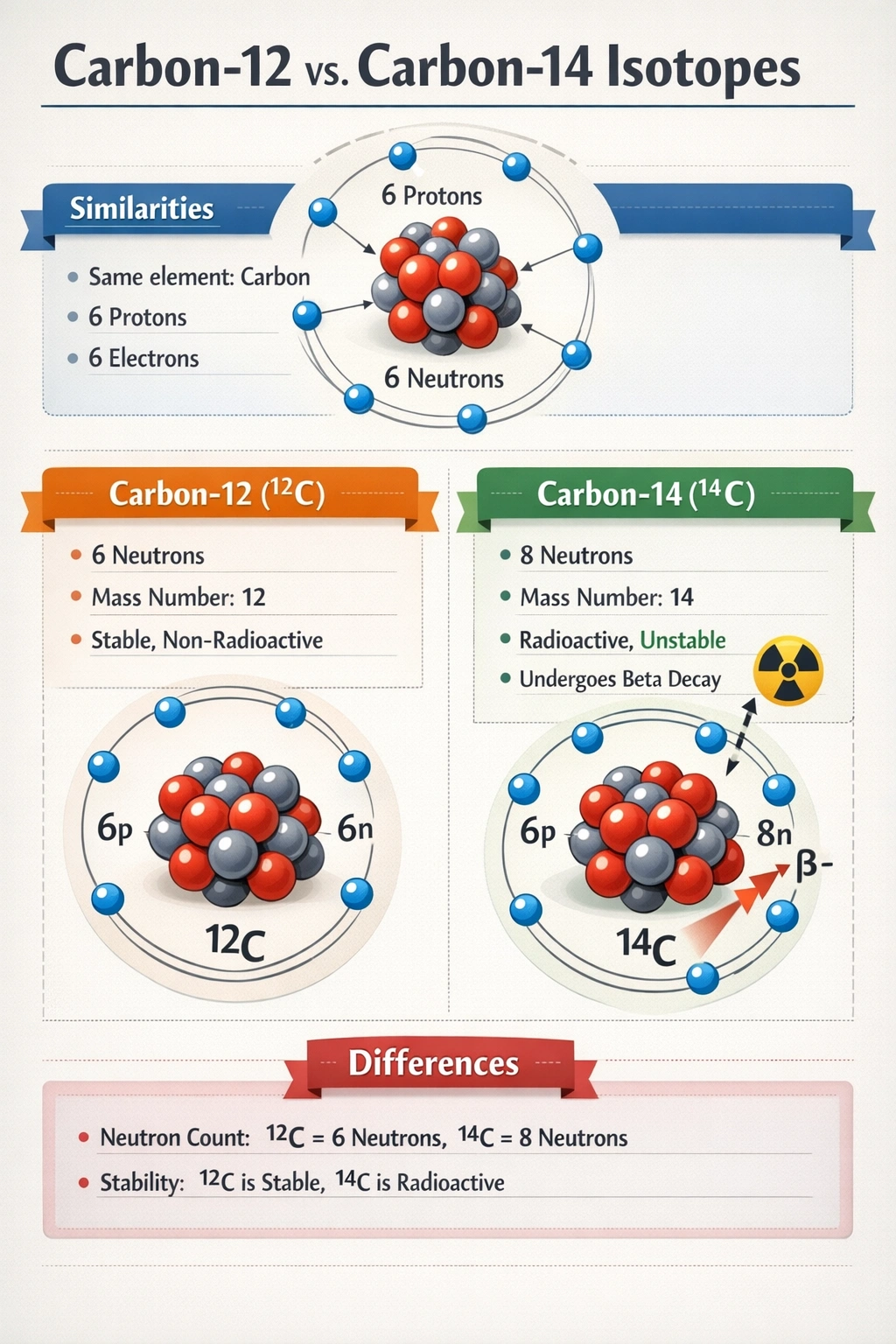
Carbon-12 and carbon-14 are isotopes. What do they have in common?
A.
Number of nuclear particles
B. Number of protons
C. Number of neutrons
D. Mass number
Rationale
Carbon-12 and carbon-14 share the same number of protons.
Both contain exactly six protons in their nuclei, defining their atomic number as 6 and establishing their identity as carbon atoms. This identical proton count governs electron configuration in neutral atoms and ensures identical chemical behavior across all carbon isotopes, regardless of nuclear mass differences.
A) Number of nuclear particles
Nuclear particles, or nucleons, consist of protons and neutrons combined within the atomic nucleus. Carbon-12 contains 12 nucleons total, 6 protons paired with 6 neutrons, while carbon-14 contains 14 nucleons, 6 protons paired with 8 neutrons. Since isotopes are defined by variations in neutron count, their total nucleon numbers necessarily differ. The term "nuclear particles" therefore describes a variable property rather than a common feature between isotopic forms.
B) Number of protons
Both carbon-12 and carbon-14 contain exactly six protons embedded in the nucleus, a value that defines atomic number and elemental identity on the periodic table. This proton count remains invariant across every carbon isotope, from carbon-11 to carbon-14 and beyond. The fixed number of protons determines the electron configuration in neutral atoms, which in turn governs all chemical bonding behavior and reactivity. Consequently, isotopes exhibit identical chemical properties precisely because their proton numbers, and thus their electronic structures, remain unchanged.
C) Number of neutrons
Carbon-12 contains 6 neutrons, calculated by subtracting its 6 protons from its mass number of 12. Carbon-14 contains 8 neutrons, derived from its mass number of 14 minus 6 protons. This difference in neutron count is the defining characteristic that distinguishes one isotope from another within the same element. The additional neutrons in carbon-14 contribute to its greater atomic mass and render the nucleus unstable, resulting in radioactive decay with a half-life of approximately 5,730 years, unlike the stable configuration of carbon-12.
D) Mass number
Mass number represents the sum of protons and neutrons present in an atom's nucleus and serves as the numerical identifier in isotopic notation. Carbon-12 possesses a mass number of 12, reflecting its 6 protons and 6 neutrons, while carbon-14 possesses a mass number of 14, reflecting its 6 protons and 8 neutrons. These distinct values explicitly differentiate the isotopes and correlate directly with measurable physical properties such as atomic mass and nuclear stability. Mass number is inherently variable among isotopes and cannot represent a shared characteristic.
Conclusion
Isotopes are defined as atoms of the same element possessing identical proton counts but differing neutron numbers. Between carbon-12 and carbon-14, the proton count of six stands as the sole nuclear property held in common, preserving their chemical identity as carbon. All other nuclear characteristics, neutron count, total nucleons, and mass number, vary by definition. This fundamental principle enables applications like radiocarbon dating, where carbon-14's nuclear instability provides chronological information while its chemical equivalence to carbon-12 allows seamless integration into biological molecules.
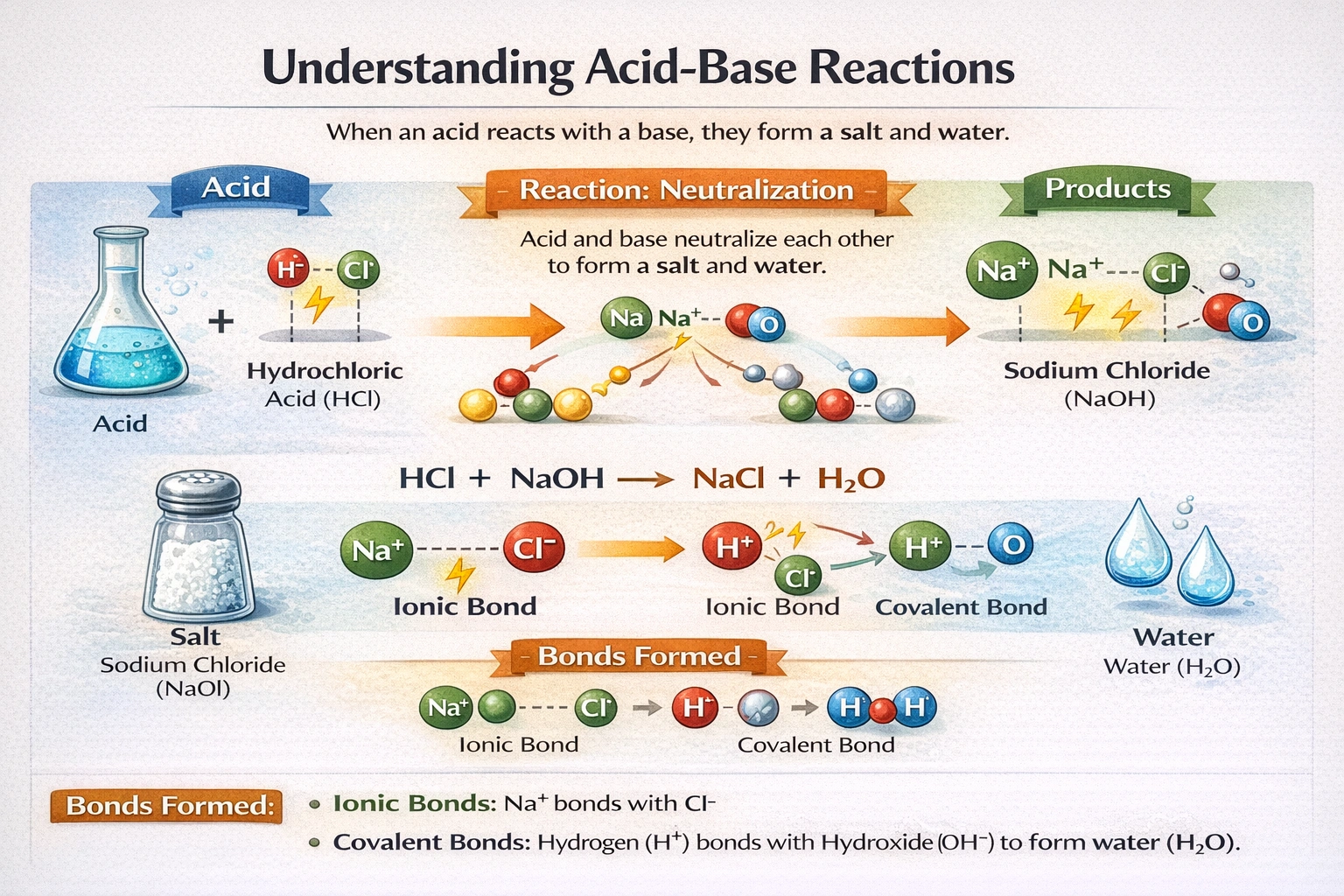
When an acid is added to a base, water and a salt form. What kinds of bonds form in these two compounds?
A.
Liquid and metallic
B. Polar and nonpolar covalent
C. Polar covalent and ionic
D. Ionic only
Rationale
Polar covalent and ionic bonds form during acid-base neutralization.
Water molecules contain polar covalent bonds between hydrogen and oxygen atoms due to unequal electron sharing driven by oxygen's higher electronegativity. Salts form through ionic bonding, where complete electron transfer creates cations and anions that assemble into crystalline lattices held together by electrostatic forces.
A) Liquid and metallic
Liquid describes a physical state of matter, not a chemical bond type. Metallic bonding occurs exclusively between metal atoms in solid conductive materials. Water exists as a liquid at room temperature but its intermolecular forces differ fundamentally from metallic bonds. Salts form crystalline solids with ionic lattices, not metallic structures. This pairing misrepresents both bond classification and material properties.
B) Polar and nonpolar covalent
Polar covalent bonds characterize water's O-H linkages, with significant electronegativity differences creating partial charges. Nonpolar covalent bonds require minimal electronegativity differences between identical or similar atoms, as seen in diatomic gases like O? or N?. Salts such as sodium chloride consist of Na? and Cl? ions arranged in extended ionic lattices, not discrete covalent molecules. Nonpolar covalent bonding does not describe ionic compound formation.
C) Polar covalent and ionic
Water's molecular structure features two polar covalent O-H bonds with bond dipoles oriented toward oxygen. The electronegativity difference of 1.24 between oxygen and hydrogen creates permanent molecular polarity. Salts form when metal cations from bases combine with nonmetal anions from acids through complete electron transfer. Sodium chloride exemplifies this with Na? and Cl? ions held in a cubic lattice by strong electrostatic attractions. These two bond types emerge predictably from proton transfer during neutralization reactions.
D) Ionic only
Ionic bonding accurately describes salt formation between cations and anions. Water molecules, however, consist of covalently bonded atoms sharing electrons within discrete H?O units. Hydrogen and oxygen achieve stable electron configurations through shared pairs, not electron transfer. Pure ionic bonding cannot account for water's molecular structure, low boiling point relative to ionic compounds, or dipole moment. This description omits water's fundamental covalent character.
Conclusion
Neutralization reactions produce two chemically distinct products governed by different bonding principles. Water forms through polar covalent bonding within individual molecules, while salts crystallize via ionic bonding between oppositely charged ions. This dual bonding outcome reflects the proton transfer mechanism inherent to acid-base chemistry. Recognizing both bond types is essential for understanding the physical and chemical properties of neutralization products.
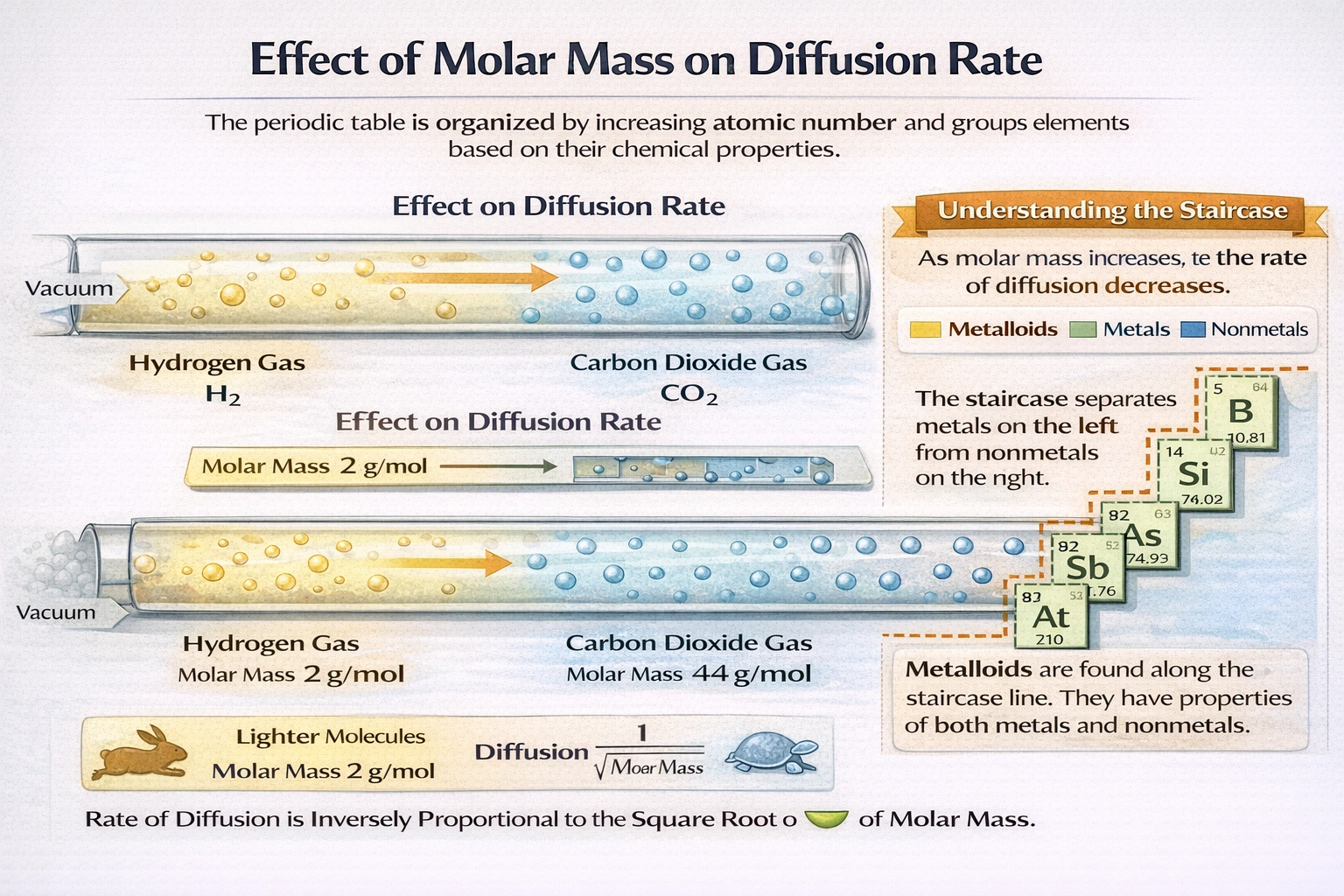
If gas A has four times the molar mass of gas B, you would expect it to diffuse through a plug ___________.
A.
at half the rate of gas B
B. at twice the rate of gas B
C. at a quarter the rate of gas B
D. at four times the rate of gas B
Rationale
Gas A diffuses at half the rate of gas B. Graham's Law of diffusion states that diffusion rate is inversely proportional to the square root of molar mass (Rate ˆ 1/?M). When gas A has four times the molar mass of gas B, the ratio ?(MB/MA) equals ?(1/4), or 1/2, establishing the half-rate relationship.
A) at half the rate of gas B
Graham's Law defines diffusion rate as inversely proportional to the square root of molar mass. For gas A with molar mass 4M and gas B with molar mass M, the rate ratio equals ?(M/4M) = ?(1/4) = 1/2. This square root dependence arises from kinetic theory, where average molecular speed varies with 1/?M at constant temperature. Experimental measurements of gas effusion through porous barriers consistently validate this inverse square root relationship. The half-rate prediction holds for ideal gases under identical temperature and pressure conditions.
B) at twice the rate of gas B
Twice the diffusion rate would require gas A to possess one-fourth the molar mass of gas B. Heavier gases inherently move slower at equivalent temperatures due to lower average molecular velocities. Kinetic energy equality (½mvs²) dictates that increased mass necessitates decreased velocity. A heavier gas cannot diffuse faster than a lighter counterpart under identical conditions. This option contradicts the fundamental inverse relationship between mass and diffusion speed.
C) at a quarter the rate of gas B
A quarter-rate relationship would imply direct inverse proportionality to molar mass rather than its square root. Graham's Law specifically involves the square root function derived from molecular kinetic energy distributions. Direct proportionality would predict rates of 1/4 for a 4x mass difference, but experimental data consistently show 1/2. Effusion experiments with gases of known molar masses confirm the square root dependence across diverse molecular weights. The quarter-rate value misapplies the mathematical relationship governing gas motion.
D) at four times the rate of gas B
Four times faster diffusion would require gas A to have one-sixteenth the molar mass of gas B. Increased molar mass always reduces molecular speed and diffusion rate under constant thermal energy. The premise that heavier gases diffuse faster violates kinetic molecular theory principles. No physical mechanism permits a fourfold rate increase with quadrupled mass. This option inverts the established inverse relationship between mass and diffusion velocity.
Conclusion
Graham's Law precisely quantifies diffusion behavior through the inverse square root relationship with molar mass. A fourfold increase in molar mass produces a twofold decrease in diffusion rate, not a fourfold or quarter-fold change. This square root dependence reflects fundamental kinetic theory principles governing molecular motion. The half-rate prediction remains consistently validated across experimental effusion and diffusion measurements.
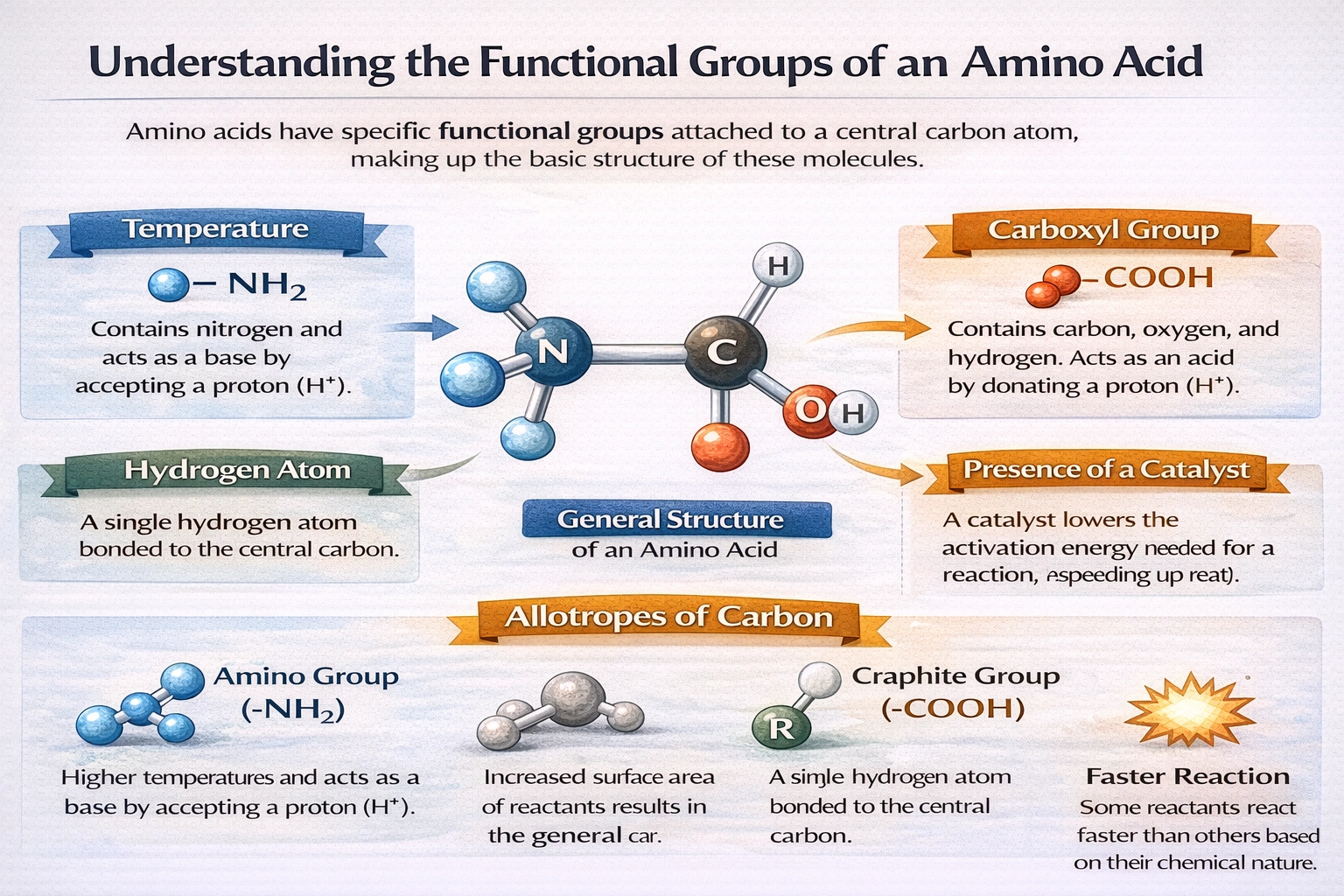
You would expect an amino acid to contain which two functional groups?
A.
R-NH? and R-COOH
B. R-CHO and R-CO-NH?
C. R-OH and R-COOR
D. R-O-R and R-COOH
Rationale
Amino acids contain an amino group (R-NH?) and a carboxylic acid group (R-COOH). These two functional groups attach to the same central carbon atom, known as the α-carbon, forming the structural foundation of all proteinogenic amino acids. The amino group provides basic character while the carboxylic acid group contributes acidity, enabling amino acids to exist as zwitterions and participate in peptide bond formation.
A) R-NH? and R-COOH
The amino group (-NH?) acts as a proton acceptor under physiological conditions, typically existing as -NH?? in aqueous solution. The carboxylic acid group (-COOH) functions as a proton donor, commonly ionized as -COO? at neutral pH. Both groups bond directly to the α-carbon along with a hydrogen atom and a variable side chain (R group). This arrangement creates amphoteric molecules capable of forming dipolar zwitterions. The complementary reactivity of these groups enables condensation reactions that link amino acids into polypeptide chains through peptide bonds.
B) R-CHO and R-CO-NH?
The aldehyde group (-CHO) characterizes reducing sugars and appears at the terminal position of open-chain carbohydrate forms. Amide groups (-CO-NH?) represent the backbone linkage in proteins after amino acids polymerize, but they do not define monomeric amino acid structure. Free amino acids contain amino and carboxyl groups, not pre-formed amide bonds. Aldehydes lack the basic nitrogen functionality essential to amino acid identity. These groups appear in metabolic intermediates but not as the defining features of standard amino acids.
C) R-OH and R-COOR
Hydroxyl groups (-OH) define alcohols and appear in serine, threonine, and carbohydrate structures. Ester groups (-COOR) characterize lipids, triglycerides, and metabolic derivatives like acetylcholine. Neither group provides the dual acid-base functionality required for amino acid classification. Compounds containing only these groups cannot form zwitterions or participate directly in ribosomal protein synthesis. These functional groups dominate lipid and carbohydrate chemistry rather than amino acid architecture.
D) R-O-R and R-COOH
Ether linkages (-O-) connect monosaccharide units in disaccharides and polysaccharides like cellulose. Carboxylic acid groups appear in fatty acids and some amino acid side chains but require pairing with an amino group for amino acid classification. Ether bonds lack nitrogen entirely and cannot confer basic properties to the molecule. Compounds with this combination represent ester derivatives or modified metabolites, not fundamental amino acid structures.
Conclusion
The amino and carboxylic acid groups constitute the invariant structural core distinguishing amino acids from other organic compounds. Their attachment to a common α-carbon enables zwitterion formation, amphoteric behavior, and polymerization into proteins. Alternative functional group combinations characterize carbohydrates, lipids, or metabolic derivatives but fail to satisfy amino acid definition. This dual functionality remains essential for biological protein synthesis and enzymatic catalysis across all living systems.
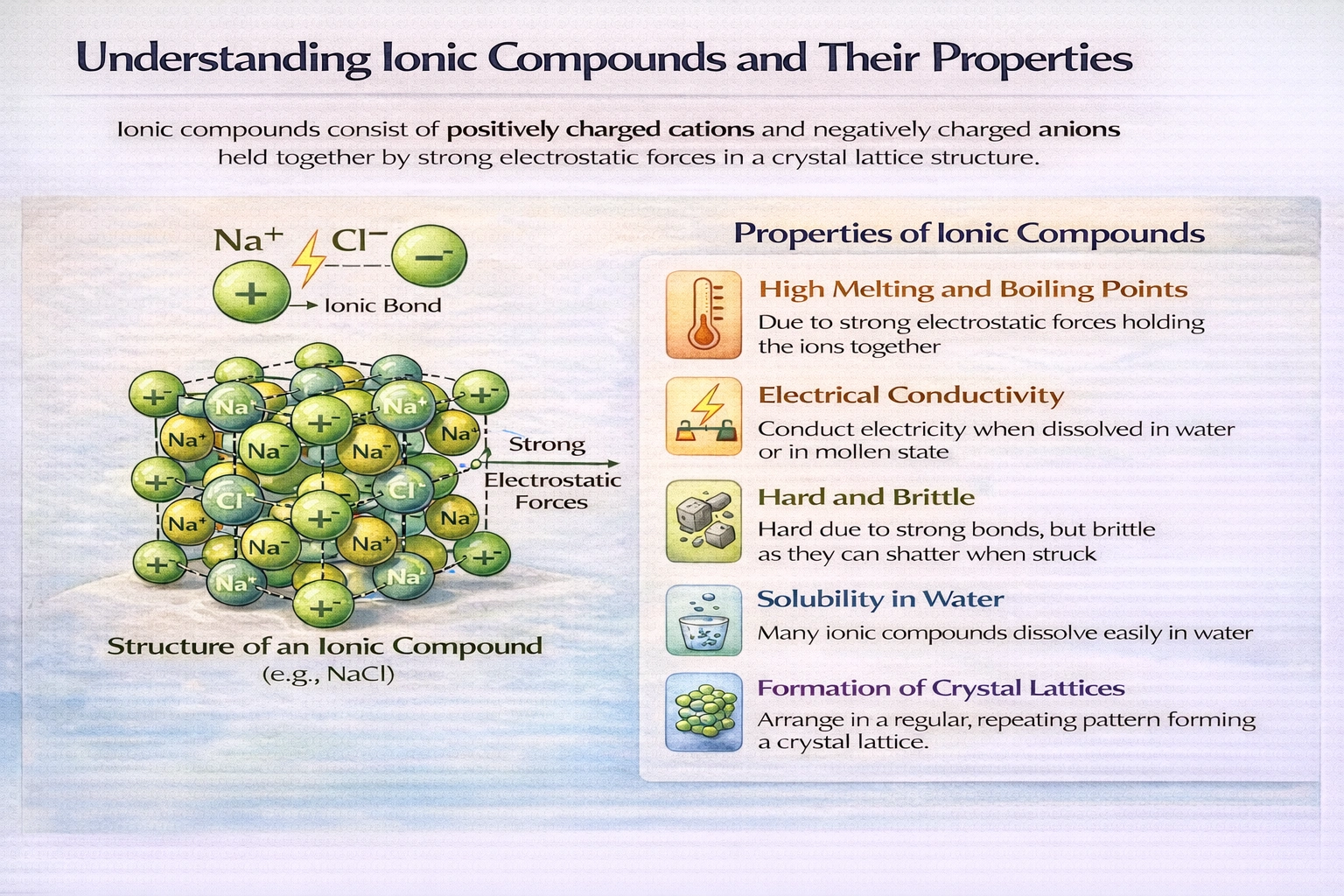
Which is a property of an ionic compound?
A.
Low melting point
B. Poor conductivity
C. Shared electrons
D. Crystalline shape
Rationale
Ionic compounds exhibit crystalline shape as a defining physical property. Their constituent cations and anions arrange in highly ordered, repeating three-dimensional lattice structures governed by electrostatic attraction. This long-range periodic organization produces characteristic geometric external forms and distinct cleavage planes when fractured.
A) Low melting point
Ionic compounds possess strong electrostatic forces between oppositely charged ions throughout the crystal lattice. These Coulombic attractions require substantial thermal energy to overcome during phase transitions. Sodium chloride melts at 801°C and magnesium oxide at 2,852°C, reflecting high thermal stability. Low melting points characterize molecular solids held by weaker intermolecular forces such as van der Waals interactions or hydrogen bonding. The energy required to disrupt ionic lattices consistently exceeds that needed for covalent molecular substances.
B) Poor conductivity
Solid ionic compounds immobilize ions within fixed lattice positions, preventing charge transport and yielding electrical insulation. Upon melting or dissolution in polar solvents, ions gain translational freedom and conduct electricity through directional migration toward oppositely charged electrodes. Aqueous sodium chloride solutions readily complete electrical circuits, demonstrating ionic conduction mechanisms distinct from electronic conduction in metals. Conductivity behavior depends critically on physical state rather than representing an inherent poor-conducting nature.
C) Shared electrons
Shared electron pairs define covalent bonding where atoms achieve stable configurations through orbital overlap and electron density sharing. Ionic bonding involves complete electron transfer from electropositive metals to electronegative nonmetals, generating discrete cations and anions. Sodium chloride forms when sodium donates its 3s electron to chlorine, creating Na? and Cl? ions without electron sharing. X-ray diffraction reveals separated ion centers in ionic crystals, contrasting with defined bond lengths between shared-electron atoms in covalent molecules.
D) Crystalline shape
Ionic compounds spontaneously organize into crystalline lattices minimizing electrostatic potential energy through alternating cation-anion arrangements. X-ray crystallography reveals precise geometric patterns such as face-centered cubic in NaCl or hexagonal in ZnS. These internal symmetries manifest externally as polyhedral crystal habits with flat faces intersecting at characteristic angles. Cleavage along crystallographic planes produces smooth fracture surfaces reflecting underlying lattice periodicity. Crystallinity persists across diverse ionic substances regardless of specific ion identities or charges.
Conclusion
Crystalline structure emerges directly from the nondirectional, long-range electrostatic forces governing ionic bonding. The requirement for charge balance and maximum ion packing efficiency produces predictable lattice geometries observable at macroscopic scales. While melting points and conductivity vary with ion charge and size, crystallinity remains universal across ionic compounds. This structural regularity distinguishes ionic solids from amorphous materials and molecular crystals with different bonding motifs.
The molar mass of glucose is 180.0 g/mol. If an IV solution contains 5 g glucose in 100 g water, what is the molarity of the solution?
A.
0.28M
B. 1.8M
C. 2.8M
D. 18M
Rationale
The molarity of the glucose solution is 0.28 M. Dissolving 5 grams of glucose (molar mass 180.0 g/mol) yields 0.0278 moles of solute. In this dilute aqueous solution, 100 grams of water approximates 0.100 liters of solution volume, giving a concentration of 0.0278 mol /0.100 L = 0.278 M, which rounds to 0.28 M.
A) 0.28M
Molarity requires moles of solute divided by liters of solution. Five grams of glucose converts to 0.0278 moles using the molar mass ratio 5 g /180.0 g/mol. The solution contains 100 g water, which for dilute aqueous systems approximates 100 mL or 0.100 L volume. Dividing moles by volume yields 0.0278 mol /0.100 L = 0.278 M. Rounding to two significant figures produces 0.28 M, consistent with the mass measurement precision.
B) 1.8M
This value exceeds the plausible concentration range for a 5% glucose solution by more than sixfold. Achieving 1.8 M would require approximately 324 grams of glucose dissolved in 1 liter of solution. The calculation 5 g /180 g/mol = 0.0278 mol cannot produce 1.8 mol without a tenfold mass error. Volume misinterpretation, such as treating 100 g as 1 L while simultaneously inflating moles, would still not yield this value mathematically.
C) 2.8M
A 2.8 M glucose solution would contain 504 grams of solute per liter, representing a highly concentrated syrup rather than a dilute IV solution. This value approximates ten times the correct answer, suggesting a decimal place error in volume conversion, possibly using 0.01 L instead of 0.100 L. Such an error contradicts the density relationship where 100 g water occupies approximately 100 mL, not 10 mL.
D) 18M
Eighteen molar represents an impossibly concentrated glucose solution exceeding solubility limits. Pure glucose solubility in water reaches approximately 5 M at room temperature before saturation occurs. This value might arise from inverting the molar mass ratio (180 /10) or misplacing decimal points in both mass and volume calculations. No valid arithmetic pathway from 5 g solute and 100 g solvent produces this concentration.
Conclusion
Molarity calculations require accurate mole determination and volume estimation for the final solution. The 5 g glucose sample yields 0.0278 moles, and the aqueous solvent provides approximately 0.100 L volume in this dilute system. The resulting 0.28 M concentration aligns with standard IV dextrose solutions used clinically. Alternative values stem from fundamental arithmetic errors or misapplication of concentration formulas that violate mass-balance principles.

What is the charge of an alpha particle?
A.
-1
B. 1
C. 2
D. No charge
Rationale
An alpha particle carries a +2 charge. It consists of two protons and two neutrons bound together, identical to a helium-4 nucleus stripped of its electrons. The absence of electrons combined with two protons yields a net charge of +2 elementary charge units.
A) -1
A negative unit charge characterizes beta-minus particles, which are high-energy electrons emitted during neutron-rich radioactive decay. Electrons possess a single negative elementary charge and negligible mass compared to nucleons. Alpha particles contain no electrons and consist entirely of nucleons, making negative charge impossible. Beta radiation and alpha radiation represent fundamentally distinct decay products with opposite charge signs.
B) +1
A +1 charge appears in particles such as protons or positrons, or in hydrogen nuclei. Alpha particles contain two protons rather than one, doubling the positive charge contribution. Single-proton systems cannot account for the mass or penetration characteristics of alpha radiation. The helium nucleus structure inherently requires +2 charge to balance its two-proton composition without compensating electrons.
C) +2
Alpha particles comprise two protons each contributing +1 charge and two neutrons contributing zero charge. With no orbital electrons to neutralize the nuclear charge, the net charge equals +2. This charge state matches helium-4 nuclei (´Hes²?) observed in mass spectrometry and nuclear reactions. The +2 charge governs alpha particle behavior in electric and magnetic fields, producing characteristic deflection patterns distinct from other radiation types.
D) No charge
Neutral particles include neutrons, photons (gamma rays), and neutrinos. Gamma radiation consists of high-energy electromagnetic waves without charge or mass. Neutrons contain one up quark and two down quarks yielding net zero charge. Alpha particles interact strongly with matter due to their charge, unlike neutral radiation that penetrates deeply without Coulombic interactions. Charge neutrality contradicts the proton content defining alpha particle identity.
Conclusion
Alpha decay emits helium nuclei containing two protons and two neutrons without accompanying electrons. The two-proton composition mandates a +2 charge state under all circumstances. This charge determines alpha particle interactions with matter, including limited penetration depth and strong ionization capacity. Recognizing the +2 charge remains essential for radiation detection, shielding design, and understanding nuclear decay processes.

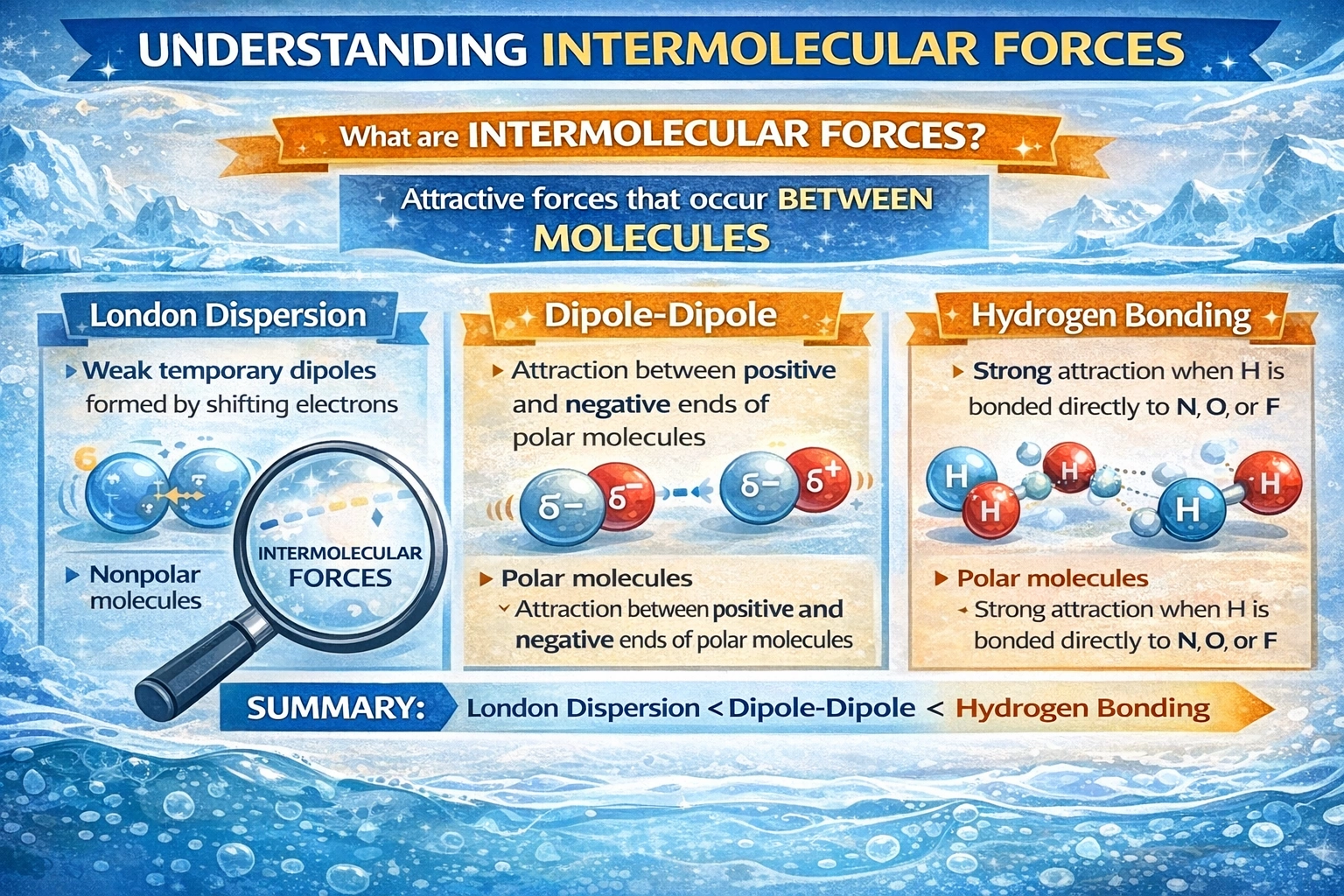
Which of these types of intermolecular force is strongest?
A.
Dipole-dipole interaction
B. London dispersion force
C. Keesom interaction
D. Hydrogen bonding
Rationale
Hydrogen bonding represents the strongest intermolecular force among the listed options. This specialized attraction occurs when hydrogen bonds to highly electronegative nitrogen, oxygen, or fluorine atoms, creating intense partial charges. The small atomic radius of hydrogen enables exceptionally close approach between molecules, amplifying electrostatic attraction beyond ordinary dipole interactions.
A) Dipole-dipole interaction
Dipole-dipole forces arise between polar molecules possessing permanent charge separation due to electronegativity differences. The positive pole of one molecule attracts the negative pole of a neighboring molecule through electrostatic forces. These interactions typically range from 0.5 to 2 kJ/mol in strength. Molecular orientation influences interaction magnitude, with antiparallel alignment maximizing attraction. While stronger than dispersion forces in small molecules, dipole-dipole interactions lack the intensity of hydrogen bonding due to larger atomic sizes and reduced charge density.
B) London dispersion force
London dispersion forces originate from instantaneous electron distribution asymmetries creating temporary dipoles. These fleeting dipoles induce complementary dipoles in adjacent molecules, producing weak attraction. Present in all molecules regardless of polarity, dispersion forces dominate only in nonpolar substances. Strength increases with molecular size and surface area due to greater polarizability. Typical magnitudes range from 0.1 to 10 kJ/mol, making them the weakest intermolecular force category. Even large hydrocarbons exhibit weaker intermolecular cohesion than small hydrogen-bonded molecules.
C) Keesom interaction
Keesom interactions describe the electrostatic attraction between permanent molecular dipoles that rotate to achieve favorable alignment. Named after physicist Willem Keesom, this force represents the orientational component of van der Waals interactions. Thermal motion disrupts optimal dipole alignment, reducing effective interaction strength at higher temperatures. Keesom forces contribute to the broader dipole-dipole interaction category rather than constituting a distinct force type. Their energy range overlaps with standard dipole-dipole interactions and remains substantially weaker than hydrogen bonding.
D) Hydrogen bonding
Hydrogen bonding requires hydrogen covalently bound to nitrogen, oxygen, or fluorine, elements with high electronegativity and small atomic radii. The resulting large partial positive charge on hydrogen interacts strongly with lone pairs on adjacent electronegative atoms. Bond energies typically range from 10 to 40 kJ/mol, an order of magnitude stronger than ordinary dipole-dipole forces. Directionality characterizes hydrogen bonds, with optimal geometry occurring when donor-hydrogen-acceptor atoms align linearly. This force governs water's anomalous properties, DNA base pairing, and protein secondary structure stabilization.
Conclusion
Hydrogen bonding exceeds other intermolecular forces in strength due to the unique combination of high polarity and minimal atomic size at the interaction site. While dipole-dipole, Keesom, and dispersion forces operate through less concentrated charge distributions, hydrogen bonding concentrates electrostatic attraction into a highly localized region. This strength differential manifests physically in elevated boiling points, surface tension, and structural stability of hydrogen-bonded substances. Among common intermolecular forces, only ion-dipole interactions surpass hydrogen bonding in magnitude.
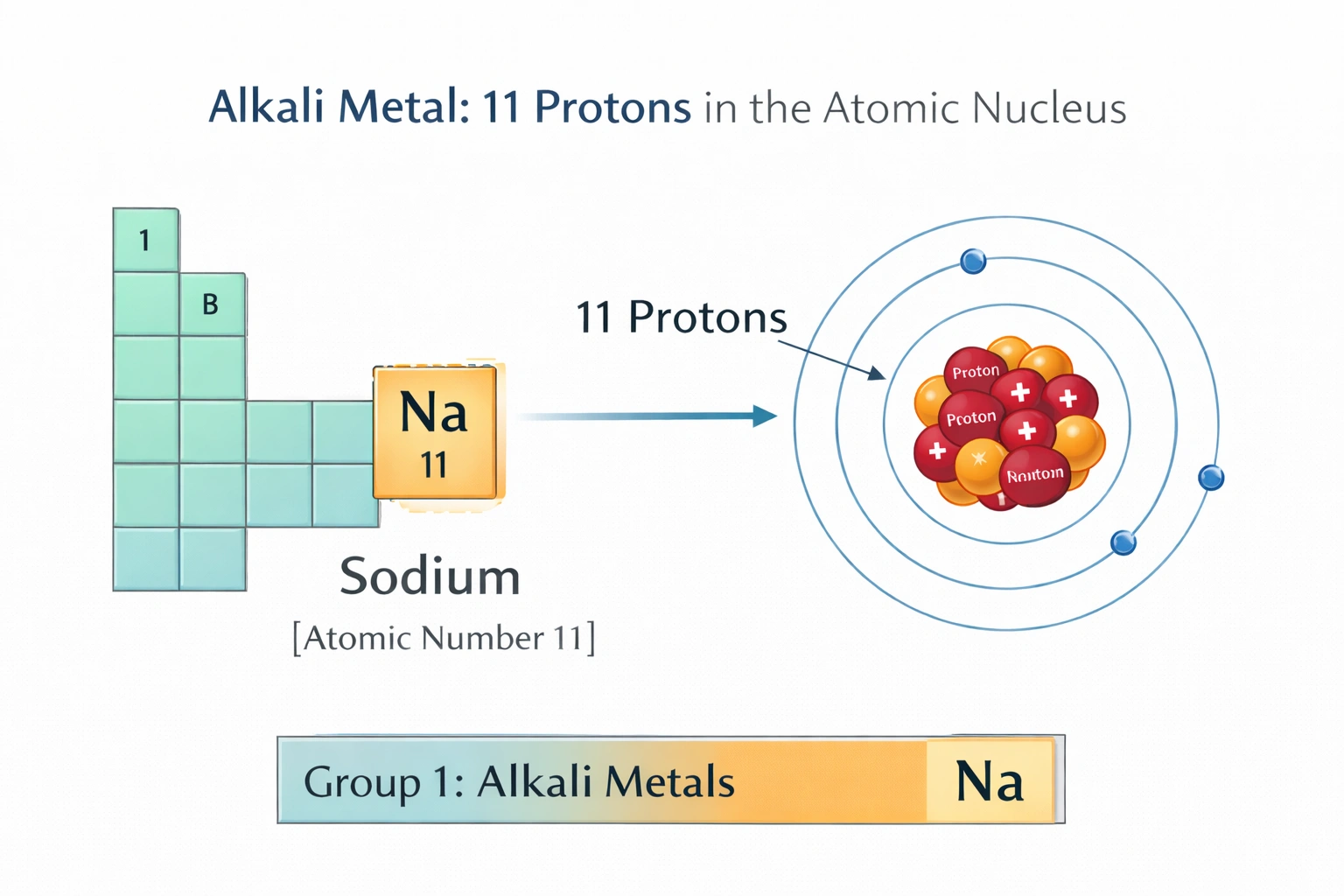
Which gives the number of protons in the atomic nucleus of an alkali metal?
A.
9
B. 10
C. 11
D. 12
Rationale
Alkali metals possess 11 protons in the case of sodium, the lightest stable alkali metal after lithium. Sodium resides in Group 1 of the periodic table with an atomic number of 11, defining its nuclear proton count. This proton number establishes sodium's electron configuration and characteristic +1 oxidation state, hallmarks of alkali metal chemistry.
A) 9
Atomic number 9 identifies fluorine, a halogen in Group 17 with high electronegativity. Fluorine exists as a pale yellow diatomic gas under standard conditions. Its seven valence electrons drive strong electron affinity rather than electron donation. Halogens gain electrons to form anions, contrasting sharply with alkali metal behavior. This proton count defines nonmetallic reactivity incompatible with alkali metal classification.
B) 10
Atomic number 10 corresponds to neon, a noble gas occupying Group 18 with a complete octet configuration. Neon exists as a monatomic gas with negligible chemical reactivity under ordinary conditions. Its filled valence shell prevents electron loss or gain, rendering it inert. Noble gases lack metallic character entirely and do not form cations or conduct electricity. This proton count represents maximum stability rather than alkali metal reactivity.
C) 11
Atomic number 11 defines sodium, the prototypical alkali metal following lithium in Group 1. Sodium's nucleus contains exactly 11 protons, balanced by 11 electrons in the neutral atom with configuration [Ne]3s². The single valence electron readily ionizes to form Na?, driving vigorous reactions with water and oxygen. Sodium exhibits metallic luster, softness, and high electrical conductivity characteristic of alkali metals. Its position initiates the period 3 alkali metal series.
D) 12
Atomic number 12 identifies magnesium, an alkaline earth metal in Group 2 with two valence electrons. Magnesium forms +2 cations rather than +1 and reacts less vigorously with water than sodium. Its higher ionization energy and melting point distinguish it from alkali metals. Alkaline earth metals occupy the column immediately right of alkali metals but exhibit distinctly different chemical behavior and physical properties. This proton count defines Group 2 characteristics, not Group 1.
Conclusion
Alkali metals occupy Group 1 of the periodic table and share the characteristic of possessing a single valence electron. Among the given proton counts, only atomic number 11 corresponds to an alkali metal, sodium. Fluorine (9), neon (10), and magnesium (12) represent halogen, noble gas, and alkaline earth metal classifications respectively. Proton count definitively determines elemental identity and group placement, making 11 the sole alkali metal option presented.
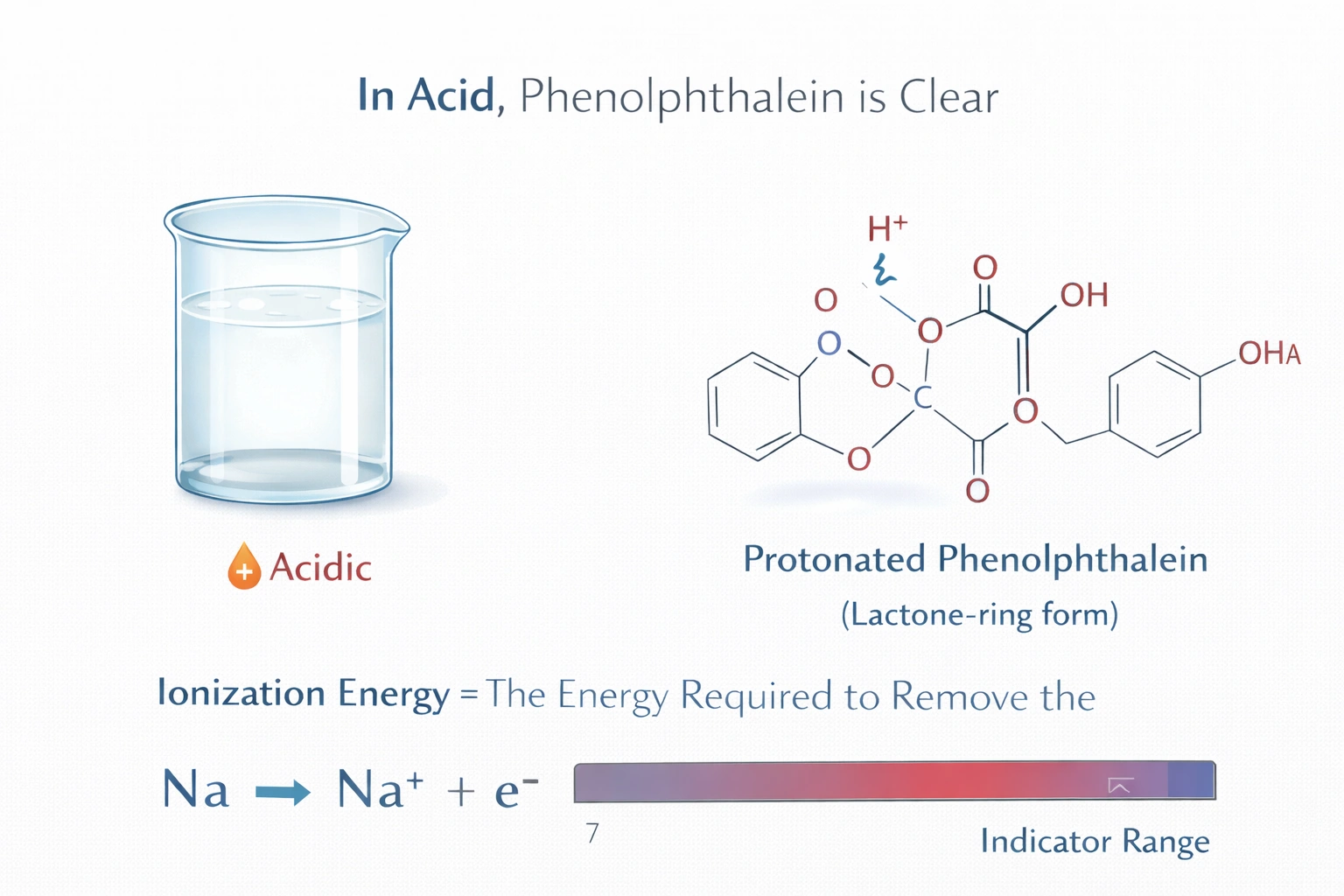
In the presence of an acid, what color is phenolphthalein?
A.
Clear
B. Blue
C. Pink
D. Red
Rationale
Phenolphthalein appears clear and colorless in acidic solutions. Below pH 8.2, the indicator molecule remains in its protonated form, lacking the extended conjugated electron system required to absorb visible light. This transparency persists through neutral conditions until sufficient hydroxide ions trigger deprotonation and structural rearrangement.
A) Clear
Phenolphthalein exists as a lactone-ring structure in acidic environments, preventing delocalization of π-electrons across the molecule. Without an extended conjugated system, the compound cannot absorb photons in the visible spectrum, resulting in optical transparency. This colorless state remains stable from strongly acidic conditions up to approximately pH 8.2. The transition to colored form requires base-catalyzed ring opening that only occurs beyond this threshold.
B) Blue
Blue coloration characterizes indicators such as bromothymol blue in basic solutions or copper(II) complexes in aqueous media. Phenolphthalein's molecular structure lacks the chromophore arrangement necessary to produce blue absorption bands under any pH conditions. Its conjugated system, when formed in basic solution, absorbs in the green-yellow region (~550 nm), transmitting magenta rather than blue wavelengths. No documented pH range or chemical modification yields blue phenolphthalein.
C) Pink
Pink or fuchsia color emerges when phenolphthalein deprotonates in basic solutions above pH 8.2. Hydroxide ions open the lactone ring, creating a quinoid structure with extended conjugation spanning three aromatic rings. This rearrangement shifts absorption maxima into the visible spectrum, transmitting complementary magenta-pink light. The intensity deepens with increasing pH until approximately pH 10, beyond which further deprotonation may cause fading. Acidic conditions prevent this structural transformation entirely.
D) Red
Red coloration appears in indicators like methyl orange below pH 3.1 or litmus in acidic environments. Phenolphthalein's protonated form remains transparent rather than red because its molecular orbitals do not support absorption in the blue-green region required to transmit red light. Even in strongly acidic media, no protonation state generates a chromophore absorbing at wavelengths that would yield red appearance. Reddish hues only manifest in phenolphthalein under highly alkaline conditions (pH > 12) where secondary deprotonation causes fading toward colorless, not red.
Conclusion
Phenolphthalein's color behavior depends entirely on solution pH and the resulting molecular structure. Acidic conditions maintain the closed lactone form with no visible light absorption, producing a clear solution. The characteristic pink color requires basic conditions to initiate ring opening and conjugation extension. This sharp transition near pH 8.2 makes phenolphthalein valuable for acid-base titrations with basic endpoints. Recognition of its colorless acidic state prevents misinterpretation during laboratory analysis.
Among the following elements, which is a nonmetal?
A.
Mercury
B. Magnesium
C. Sulfur
D. Potassium
Rationale
Sulfur is a nonmetal located in Group 16 of the periodic table. It exhibits classic nonmetallic properties: it is a brittle, yellow solid at room temperature, lacks electrical conductivity, and tends to gain electrons to form anions such as Ss²?. Its position in the upper-right region of the periodic table, away from the metallic elements on the left and center, confirms its classification as a nonmetal.
A) Mercury
Mercury is a transition metal in Group 12, notable for being liquid at room temperature. Despite its unusual physical state, it displays definitive metallic characteristics: high luster, excellent electrical and thermal conductivity, and the ability to form cations (e.g., Hgs²? or Hg?s²?). Its electron configuration ([Xe]4f²´5d²°6ss²) supports metallic bonding behavior. Mercury's placement in the d-block further confirms its metallic identity.
B) Magnesium
Magnesium is an alkaline earth metal in Group 2 with a shiny, silvery appearance and good electrical conductivity. It readily loses two valence electrons to form Mgs²? ions in ionic compounds. Magnesium participates in metallic bonding in its solid state and reacts vigorously with acids to produce hydrogen gas, hallmarks of metallic behavior. Its low ionization energy and electropositive nature align with typical metal characteristics.
C) Sulfur
Sulfur resides in Group 16 (the chalcogens) and occupies the nonmetal region of the periodic table. It exists as S‚ˆ molecules in its most stable allotrope, forming a brittle crystalline solid with no free electrons. Sulfur acts as an oxidizing agent, accepting electrons to form sulfide ions (Ss²?) or covalent bonds in compounds like H?S or SO?. It lacks malleability, ductility, and conductivity, all consistent with nonmetallic classification.
D) Potassium
Potassium is a highly reactive alkali metal in Group 1 with a soft, silvery surface that tarnishes rapidly in air. It has a single valence electron that it readily donates to form K? ions, exhibiting strong reducing behavior. Potassium conducts electricity efficiently in both solid and molten states and reacts violently with water to produce hydrogen gas and KOH. These traits define it as a quintessential metal.
Conclusion
Nonmetals are concentrated in the upper-right portion of the periodic table and are characterized by poor conductivity, brittleness, and a tendency to gain or share electrons. Among the listed elements, only sulfur meets all these criteria. Mercury, magnesium, and potassium are all metals, despite mercury's liquid state, and exhibit electron-donating behavior, luster, and conductivity. Thus, sulfur stands alone as the correct nonmetal in this set.

Here are the solubilities of four substances at 0?C, in grams of solute per 100 mL of water. | Substance | Solubility (g/100 mL) | | Solid citric acid | 49 | | Solid potassium phosphate | 44 | | Gaseous nitrogen | 0.0030 | | Gaseous oxygen | 0.0070 | If the temperature increases to 20?C, what would you expect to happen to the solubility figures?
A.
Citric acid and potassium phosphate will decrease; nitrogen and oxygen will increase.
B. Citric acid and potassium phosphate will increase; nitrogen and oxygen will decrease.
C. All four figures will increase.
D. All four figures will decrease.
Rationale
As temperature rises from 0°C to 20°C, the solubility of solid solutes (citric acid and potassium phosphate) increases while that of gaseous solutes (nitrogen and oxygen) decreases. Citric acid and potassium phosphate, both ionic or polar solids, dissolve more readily at higher temperatures due to enhanced solvent-solute interactions and greater kinetic energy overcoming lattice forces. In contrast, nitrogen and oxygen, nonpolar gases, become less soluble because increased thermal energy enables gas molecules to escape the solvent phase more easily.
A) Citric acid and potassium phosphate will decrease; nitrogen and oxygen will increase
This option inverts the established temperature dependence for both classes of solutes. Solid solubility typically increases with temperature as endothermic dissolution processes are favored by added thermal energy. Gas solubility universally decreases with warming due to exothermic dissolution and increased molecular motion favoring the gas phase. Carbonated beverages exemplify this: they lose CO? faster at room temperature than when refrigerated. This choice contradicts fundamental solubility principles.
B) Citric acid and potassium phosphate will increase; nitrogen and oxygen will decrease
Citric acid, a molecular solid with extensive hydrogen bonding, exhibits increased solubility with temperature as water molecules more effectively disrupt intermolecular forces in the crystal. Potassium phosphate, an ionic compound, benefits from higher temperatures that enhance hydration of K? and PO?³? ions, overcoming stronger lattice energy. Conversely, nitrogen and oxygen follow Henry's law: their solubility is inversely proportional to temperature. Warmer water holds less dissolved atmospheric gas, consistent with observations like reduced oxygen levels in heated aquatic environments. This accurately reflects the dual behavior of solids and gases.
C) All four figures will increase
While true for most solids, this ignores the well-documented inverse relationship between gas solubility and temperature. Gases dissolve exothermically; Le Chatelier's principle dictates that heating shifts equilibrium toward the undissolved gas phase. Oxygen solubility in water drops from ~14.6 mg/L at 0°C to ~9.1 mg/L at 20°C, a nearly 40% decrease. Assuming uniform solubility trends across all solute types disregards critical differences in dissolution thermodynamics between condensed and gaseous phases.
D) All four figures will decrease
This misapplies gas behavior to solids. Most ionic and polar solids, including citric acid and potassium phosphate, show positive temperature-solubility correlations. Their dissolution is often endothermic (ΔH > 0), so increased temperature favors solubility per Le Chatelier's principle. Only a few solids (e.g., cerium(III) sulfate) exhibit decreased solubility with warming. Applying gas-phase logic universally ignores the dominant role of lattice energy disruption in solid dissolution.
Conclusion
Solubility responses to temperature depend fundamentally on solute phase and dissolution energetics. Solids generally become more soluble as thermal energy aids in breaking intermolecular or ionic bonds, while gases become less soluble as kinetic energy overcomes solvent-gas attractions. The contrasting behaviors of citric acid/potassium phosphate versus nitrogen/oxygen illustrate this dichotomy precisely. Thus, only option B correctly predicts the direction of solubility change for all four substances.

Which two elements are most alike in reactivity?
A.
He and H
B. K and Ar
C. Cl and P
D. Ba and Mg
Rationale
Barium (Ba) and magnesium (Mg) exhibit the most similar reactivity among the listed pairs because both belong to Group 2 of the periodic table, the alkaline earth metals. They each possess two valence electrons in an nss² configuration, which they readily lose to form +2 cations. This shared electron configuration results in comparable chemical behaviors, including reactions with water, oxygen, and acids, though barium is more reactive due to its larger atomic size and lower ionization energy.
A) He and H
Helium is a noble gas with a complete 1ss² electron shell, rendering it chemically inert under nearly all conditions. Hydrogen, though also possessing a filled first shell when neutral (1s²), is highly reactive, it readily forms covalent bonds or loses its electron to become H?. Their positions in different groups (Group 18 vs. Group 1) and fundamentally distinct electronic structures lead to opposite reactivity profiles: one is nonreactive, the other highly reactive.
B) K and Ar
Potassium is a soft, highly reactive alkali metal (Group 1) that violently reacts with water to produce hydrogen gas and KOH. Argon is a noble gas (Group 18) with a stable octet, exhibiting no tendency to gain, lose, or share electrons. Despite being adjacent in Period 4, their reactivities are diametrically opposed, potassium is among the most reactive elements, while argon is essentially inert.
C) Cl and P
Chlorine (Group 17) is a halogen that strongly attracts electrons to form Cl? ions or polar covalent bonds. Phosphorus (Group 15) typically forms covalent compounds and can exhibit multiple oxidation states (-3 to +5). While both are nonmetals, their valence electron counts differ (7 vs. 5), leading to distinct bonding preferences and reactivity patterns, chlorine acts as an oxidizing agent, whereas phosphorus often serves as a reducing agent.
D) Ba and Mg
Both barium and magnesium have the nss² valence configuration (Mg: [Ne]3ss²; Ba: [Xe]6ss²) and form Ms²? ions in compounds. They react with water, magnesium slowly with hot water, barium more vigorously even with cold water, and both form oxides (MO) and react with acids to release H? gas. Although reactivity increases down Group 2 due to decreasing ionization energy, their fundamental chemical behavior remains consistent, reflecting periodic trends within the same group.
Conclusion
Chemical reactivity is primarily governed by valence electron configuration, which is identical for elements in the same group. Barium and magnesium, as Group 2 alkaline earth metals, share core chemical properties despite differences in reaction vigor. In contrast, the other pairs span different groups and exhibit incompatible or opposing reactivities. Thus, D) Ba and Mg correctly identifies the most chemically similar pair.

What is the correct electron configuration for magnesium?
A.
1s?2s?
B. 1s?2s?2p?
C. 1s?2s?2p?3s?
D. 1s?2s?2p?3s?3p?
Rationale
The correct electron configuration for magnesium is 1s²2s²2p?3s². Magnesium has an atomic number of 12, meaning it contains 12 electrons distributed across available orbitals in order of increasing energy. Following the Aufbau principle, electrons fill the 1s, 2s, 2p, and finally the 3s orbitals, with the 3s subshell holding the two valence electrons that define magnesium's chemical behavior as a Group 2 alkaline earth metal.
A) 1ss²2ss²
This configuration accounts for only 4 electrons (2 in 1s + 2 in 2s), corresponding to beryllium (atomic number 4), not magnesium. Magnesium requires 12 electrons to achieve neutrality, so this arrangement is both incomplete and chemically mismatched. It omits the entire second-period p-subshell and all third-period electrons.
B) 1ss²2ss²2p?
This represents the electron configuration of neon (atomic number 10), a noble gas with a complete second shell. While this core forms the inner electron structure of magnesium, it lacks the two additional valence electrons in the 3s orbital. Magnesium's chemistry is dominated by these outer electrons, which are absent in this configuration.
C) 1ss²2ss²2p?3ss²
This configuration correctly distributes all 12 electrons: 2 in 1s, 2 in 2s, 6 in 2p, and 2 in 3s. The filled 1s-2p core matches neon's configuration, while the 3ss² valence shell aligns with magnesium's position in Period 3, Group 2. This arrangement satisfies the Pauli exclusion principle, Hund's rule, and the Aufbau filling order, and explains magnesium's tendency to lose two electrons to form Mgs²? ions.
D) 1ss²2ss²2p?3ss²3p²
This configuration totals 13 electrons, matching aluminum (atomic number 13), not magnesium. It incorrectly places an electron in the 3p orbital before the 3s subshell is relevant, but more critically, magnesium's 3s orbital is fully occupied with 2 electrons, and no electrons occupy 3p in its ground state. This violates the Aufbau principle, which dictates that 3s must be filled before 3p receives electrons.
Conclusion
Magnesium's 12 electrons fill orbitals sequentially up to 3ss², yielding the configuration 1ss²2ss²2p?3ss². This reflects its position in the periodic table and underpins its +2 oxidation state and metallic reactivity. Only option C accurately represents the ground-state electron arrangement for neutral magnesium atoms.

To the nearest whole number, what is the mass of one mole of sodium chloride?
A.
36 g/mol
B. 43 g/mol
C. 58 g/mol
D. 72 g/mol
Rationale
The molar mass of sodium chloride is 58 g/mol to the nearest whole number. Sodium (Na) has an atomic mass of approximately 23.0 g/mol, and chlorine (Cl) has an atomic mass of about 35.5 g/mol. Adding these gives 23.0 + 35.5 = 58.5 g/mol, which rounds to 58 g/mol when expressed as a whole number.
A) 36 g/mol
This value is far too low and may result from confusing NaCl with a different compound or using incorrect atomic masses, such as substituting hydrogen (1 g/mol) for sodium. No plausible combination of standard atomic weights yields 36 g/mol for NaCl. This underestimates the actual mass by more than one-third and contradicts experimental data.
B) 43 g/mol
This number lacks a clear basis in common miscalculations. It does not correspond to any known salt (e.g., NaF is ~42 g/mol, but that's not NaCl). It may arise from arithmetic errors or misreading periodic table values, but it fails to reflect the correct sum of sodium and chlorine atomic masses. This value is inconsistent with both theory and measurement.
C) 58 g/mol
Sodium contributes 22.99 g/mol and chlorine 35.45 g/mol, totaling 58.44 g/mol. Rounded to the nearest whole number, this is 58 g/mol. This value aligns with laboratory measurements and is used universally in stoichiometric calculations involving table salt. The slight deviation from whole numbers arises from natural isotopic abundance, but standard practice accepts 58 g/mol for whole-number approximations.
D) 72 g/mol
This overestimates NaCl's molar mass and may confuse it with potassium chloride (KCl ‰ˆ 74.5 g/mol) or include extra atoms (e.g., NaCl?, which doesn't exist). Chlorine's atomic mass is ~35.5, not ~49, so even with correct sodium mass, 72 g/mol cannot be justified. Such a value would imply non-existent elemental compositions or severe calculation errors.
Conclusion
The molar mass of NaCl is derived directly from the sum of its constituent atomic masses: Na (~23 g/mol) + Cl (~35.5 g/mol) = 58.5 g/mol ‰ˆ 58 g/mol. This value is foundational in chemistry for preparing solutions, balancing equations, and analytical work. Only option C reflects accurate atomic data and proper rounding.

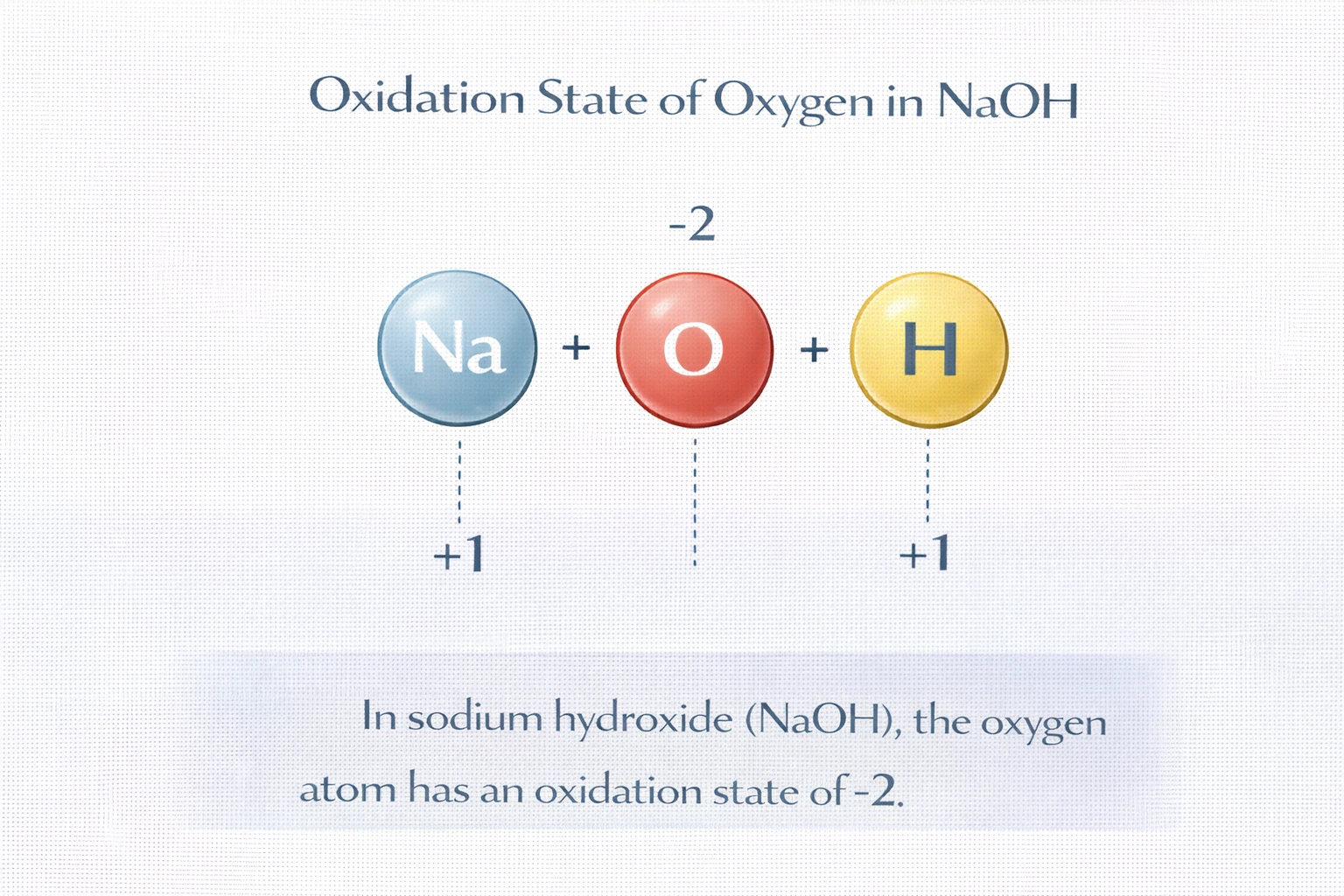
What is the oxidation state of the oxygen atom in the compound NaOH?
A.
-2
B. -1
C. 0
D. 2
Rationale
In NaOH, oxygen exhibits an oxidation state of -2. Sodium, as a Group 1 metal, always assumes a +1 oxidation state in compounds. Hydrogen, when bonded to nonmetals like oxygen, carries a +1 oxidation state. For the neutral compound NaOH, the sum of oxidation states must equal zero: (+1 for Na) + (+1 for H) + (oxidation state of O) = 0, yielding -2 for oxygen, its standard value in most oxides and hydroxides.
A) -2
This is the typical oxidation state of oxygen in ionic compounds and hydroxides. In NaOH, oxygen is part of the hydroxide ion (OH?), where it forms a polar covalent bond with hydrogen. Oxygen's high electronegativity (3.44) draws electron density away from hydrogen, resulting in a formal charge of -2 on oxygen within the OH? framework. This value aligns with oxygen's behavior in water (H?O), metal oxides (e.g., CaO), and nearly all non-peroxide compounds.
B) -1
An oxidation state of -1 occurs only in peroxides, such as hydrogen peroxide (H?O?) or sodium peroxide (Na?O?), where an O-O bond exists. In these compounds, each oxygen is assigned -1 due to shared bonding between identical atoms. NaOH contains no O-O bond; it consists of Na? and OH? ions, with oxygen bonded solely to hydrogen. Thus, -1 does not apply.
C) 0
A zero oxidation state indicates elemental oxygen (O? or O?), where atoms share electrons equally. In any chemical compound, oxygen adopts a negative oxidation state due to its high electronegativity, except in rare cases with fluorine. NaOH is a compound, not elemental oxygen, so this value is invalid.
D) +2
Oxygen achieves positive oxidation states only when bonded to fluorine, the sole element more electronegative than oxygen. In OF?, oxygen is +2. In NaOH, oxygen is bonded to hydrogen (less electronegative), so it cannot exhibit a positive oxidation state. Such a value contradicts fundamental electronegativity principles.
Conclusion
The hydroxide ion (OH?) in NaOH follows standard oxidation state rules: hydrogen is +1, so oxygen must be -2 to give the ion a net -1 charge. Combined with Na? (+1), the compound is neutral. This assignment is consistent across hydroxides and distinguishes them from peroxides or fluorine compounds. Therefore, A) -2 is the correct and chemically accurate answer.
Which of these elements has the greatest atomic mass?
A.
Au
B. Ba
C. I
D. W
Rationale
Gold (Au) has the greatest atomic mass among the listed elements. With an atomic mass of approximately 197.0 g/mol, gold exceeds the masses of tungsten (~183.8 g/mol), barium (~137.3 g/mol), and iodine (~126.9 g/mol). This high mass reflects its position as a dense, heavy transition metal in Period 6 of the periodic table.
A) Au
Gold (atomic number 79) has a standard atomic weight of 196.97 g/mol, typically rounded to 197.0 g/mol. It is one of the heaviest non-radioactive elements commonly encountered. Its high atomic mass contributes to its exceptional density (19.3 g/cm³) and historical use in coinage and jewelry. Among the given choices, it is unambiguously the heaviest.
B) Ba
Barium (atomic number 56) has an atomic mass of 137.33 g/mol. As an alkaline earth metal, it is significantly lighter than gold. Though considered a "heavy metal" in toxicology, its mass is over 59 g/mol less than gold's, nearly a 30% difference.
C) I
Iodine (atomic number 53) is a halogen with an atomic mass of 126.90 g/mol, making it the lightest option listed. Despite being a solid at room temperature and relatively dense for a nonmetal, it is far lighter than any of the metals in the other choices.
D) W
Tungsten (symbol W, from wolfram; atomic number 74) has an atomic mass of 183.84 g/mol. It is very close to gold in mass but still 13.1 g/mol lighter. Tungsten is known for its high melting point and density (19.25 g/cm³), nearly matching gold's density, but its atomic mass remains lower due to fewer protons and neutrons.
Conclusion
Atomic mass increases generally with atomic number, though nuclear binding effects cause minor deviations. Here, gold (Z = 79) surpasses tungsten (Z = 74), barium (Z = 56), and iodine (Z = 53) in both atomic number and atomic mass. The correct ranking by mass is: Au > W > Ba > I. Therefore, A) Au is the correct answer.

A radioactive isotope has a half-life of 20 years. How many grams of a 6-gram sample will remain after 40 years?
A.
8
B. 6
C. 3
D. 1.5
Rationale
After 40 years, 1.5 grams of the original 6-gram sample will remain. The isotope has a half-life of 20 years, meaning the quantity halves every 20 years. Over 40 years, exactly two half-lives, the sample undergoes two successive halvings:
1. After the first 20 years: 6 g /2 = 3 g
2. After the next 20 years (total 40 years): 3 g /2 = 1.5 g
A) 8
This exceeds the initial mass, violating the principle of radioactive decay, which only reduces the amount of the original isotope over time. Mass cannot increase spontaneously in a closed system undergoing decay.
B) 6
This implies no decay occurred, which would only be possible if the half-life were infinite. A 20-year half-life guarantees measurable decay within 40 years, making this option physically impossible.
C) 3
This reflects the amount remaining after one half-life (20 years), not two. Since 40 years equals two half-lives, the sample must halve again, reducing from 3 g to 1.5 g. This answer stops short of the full decay period.
D) 1.5
Correct. Mathematically, the remaining mass is calculated as:
Remaining mass = Initial mass x (1/2)^(t/T‚/?) = 6 x (1/2)^(40/20) = 6 x (1/2)s² = 6 x 1/4 = 1.5 g
This aligns with both stepwise reasoning and the exponential decay formula.
Conclusion
Radioactive decay follows a predictable exponential pattern based on half-life. After two half-lives, only one-fourth of the original sample remains. For a 6-gram sample, this equals 1.5 grams. Thus, D) 1.5 is the correct answer.

In the solid state, you would expect a nonmetal to be __________.
A.
brittle
B. lustrous
C. malleable
D. conductive
Rationale
Solid nonmetals are typically brittle, meaning they fracture or shatter when subjected to mechanical stress rather than deforming. This behavior arises from their bonding and structural characteristics: most solid nonmetals form either covalent network solids (e.g., diamond, sulfur) or molecular crystals (e.g., iodine, white phosphorus), where directional covalent bonds or weak intermolecular forces prevent layers of atoms from sliding past one another. When force is applied, these rigid structures crack instead of bending.
A) Brittle
This is correct. Examples include:
1. Sulfur: crumbles into powder when struck.
2. Phosphorus (white): soft but fractures easily.
3. Iodine crystals: cleave along planes but do not bend.
4. Diamond: extremely hard yet brittle, can be shattered with a sharp impact.
The absence of delocalized electrons and non-directional bonding (as in metals) eliminates ductility and malleability, making brittleness a hallmark of solid nonmetals.
B) Lustrous
Luster, the ability to reflect light uniformly, is a metallic property due to free electrons interacting with photons. Most nonmetals are dull (e.g., sulfur, red phosphorus). Exceptions like iodine (shiny crystals) or graphite (metallic sheen) are rare and do not represent typical nonmetal behavior. Luster is not a general characteristic of nonmetals.
C) Malleable
Malleability requires atoms to slide past each other without breaking bonds, a feature of metallic bonding, where a "sea" of delocalized electrons allows deformation. Nonmetals lack this mobility; their bonds are either rigidly fixed (covalent networks) or held by weak forces (molecular solids), causing fracture under pressure. Thus, nonmetals are not malleable.
D) Conductive
Electrical conductivity in solids relies on mobile charge carriers, free electrons (metals) or ions (electrolytes). Most nonmetals are insulators (e.g., sulfur, oxygen, diamond). Graphite is a notable exception due to delocalized π-electrons in its layered structure, but it is atypical. Conductivity is not a defining property of nonmetals.
Conclusion
Brittleness is the most consistent and distinguishing physical property of solid nonmetals, directly resulting from their bonding nature. In contrast, luster, malleability, and conductivity are characteristic of metals. Therefore, A) brittle is the correct answer.

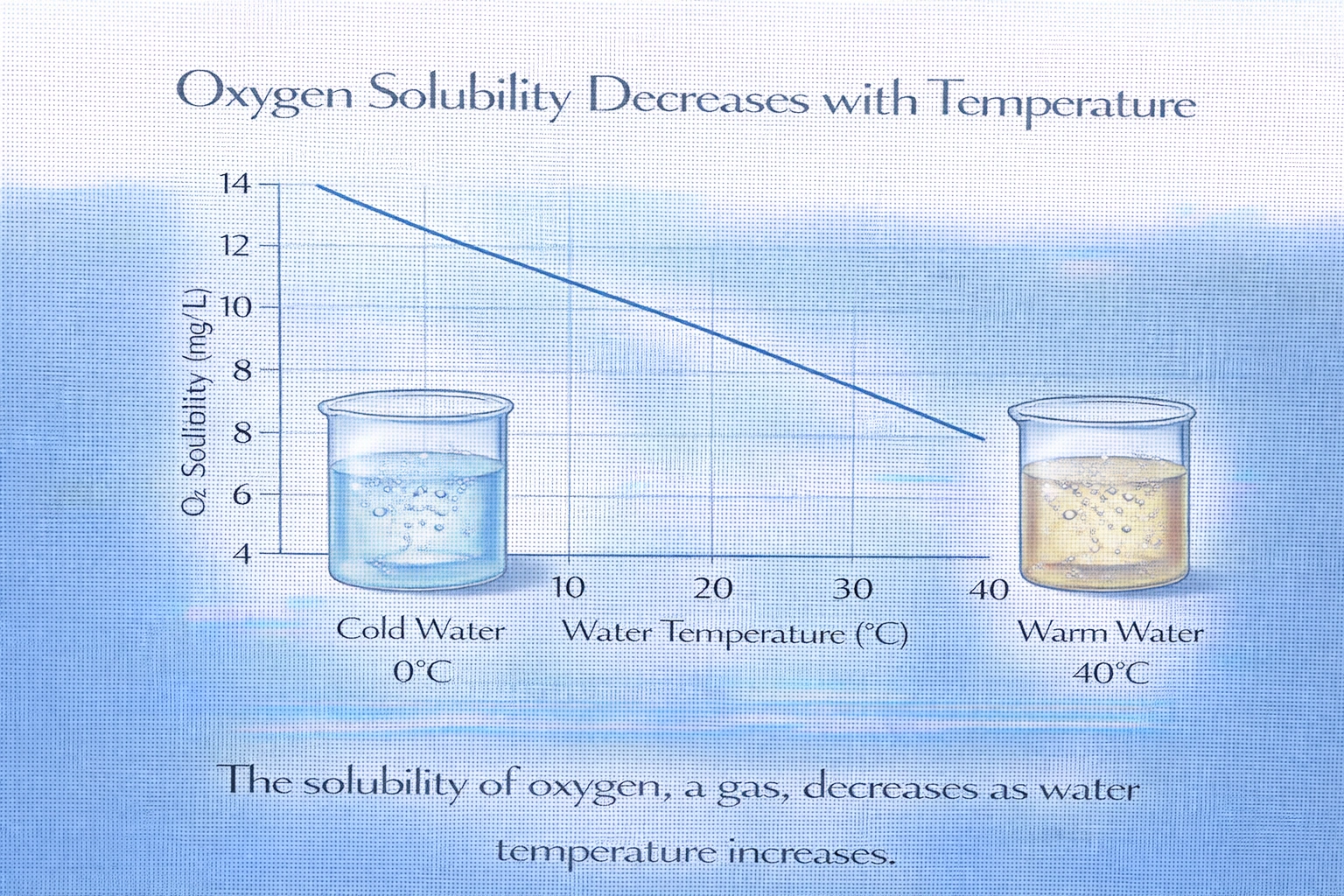
Which substance shows a decrease in solubility in water with an increase in temperature?
A.
NaCl
B. O?
C. KI
D. CaCl?
Rationale
Oxygen (O?) is the only substance among the options that decreases in solubility as temperature increases. This behavior is characteristic of gases, which dissolve exothermically in water. According to Le Chatelier's principle, increasing the temperature shifts the equilibrium toward the undissolved gas phase, reducing solubility. Additionally, higher thermal energy allows gas molecules to overcome intermolecular attractions with water and escape into the atmosphere, explaining why warm soda goes flat faster than cold soda.
A) NaCl
Sodium chloride is a solid ionic compound whose solubility increases slightly with temperature. The endothermic nature of its dissolution (energy absorbed to break the crystal lattice) means higher temperatures favor solubility. Thus, this option does not show decreased solubility.
B) O?
Correct. Oxygen is a nonpolar gas, and its solubility in water drops significantly as temperature rises. For example:
1. At 0°C: ~14.6 mg/L dissolved O?
2. At 20°C: ~9.1 mg/L
3. At 30°C: ~7.5 mg/L
This inverse relationship is universal for gases in aqueous solutions and is critical in environmental systems (e.g., lower oxygen levels in warm rivers affect aquatic life).
C) KI
Potassium iodide is a highly soluble ionic solid. Like most salts, its solubility increases with temperature due to favorable entropy and endothermic dissolution. It is commonly used in experiments demonstrating increased solubility at higher temperatures.
D) CaCl?
Calcium chloride is not only soluble but also hygroscopic and releases heat when dissolved (exothermic). Despite the exothermic process, its solubility still increases with temperature because the entropy gain dominates. It remains highly soluble even at low temperatures, but solubility does not decrease with warming.
Conclusion
The key distinction lies in the phase of the solute:
1. Solids (NaCl, KI, CaCl?) ? solubility generally increases with temperature.
2. Gases (O?) ? solubility decreases with temperature.
Since O? is the only gas listed, it is the only substance expected to become less soluble as temperature rises. Therefore, B) O? is the correct answer.
On the periodic table, where are atoms with the largest atomic radius located?
A.
At the top of their group
B. In the middle of their group
C. At the bottom of their group
D. Along the right-hand side
Rationale
Atoms with the largest atomic radius are located at the bottom of their group on the periodic table. As one moves down a group, each successive element gains an additional principal energy level (electron shell), increasing the distance between the nucleus and the outermost electrons. Although nuclear charge also increases, the shielding effect of inner electrons dominates, reducing effective nuclear attraction on valence electrons and resulting in larger atomic size.
A) At the top of their group
Elements at the top of a group have the fewest electron shells. For example, lithium (Period 2) has only two shells, while cesium (Period 6) has six. The minimal shielding and strong nuclear pull in top-group elements produce the smallest atomic radii in the group. This contradicts the trend and is incorrect.
B) In the middle of their group
Atomic radius increases steadily down a group without peaks or plateaus in the middle. No element in the middle, such as sodium in Group 1, exhibits a maximum radius; francium (bottom) is larger than cesium, which is larger than rubidium, and so on. The idea of a "middle" maximum lacks basis in periodic trends and is incorrect.
C) At the bottom of their group
Correct. Each step down a group adds a new electron shell (e.g., K: n=4, Rb: n=5, Cs: n=6), significantly increasing atomic radius. Bottom-group elements like francium (Group 1) or astatine (Group 17) possess the largest radii in their respective groups due to maximal electron shielding and greatest orbital distance from the nucleus.
D) Along the right-hand side
Moving left to right across a period, atomic radius decreases due to increasing nuclear charge without additional shielding. Noble gases (far right) are smaller than alkali metals (far left) in the same period. Thus, right-side elements have the smallest radii per period, not the largest. This is incorrect.
Conclusion
Atomic radius increases down a group due to the addition of electron shells, making atoms at the bottom of their group the largest. This trend is fundamental to understanding periodic properties such as ionization energy, electronegativity, and metallic character. Therefore, C) At the bottom of their group is the correct answer.

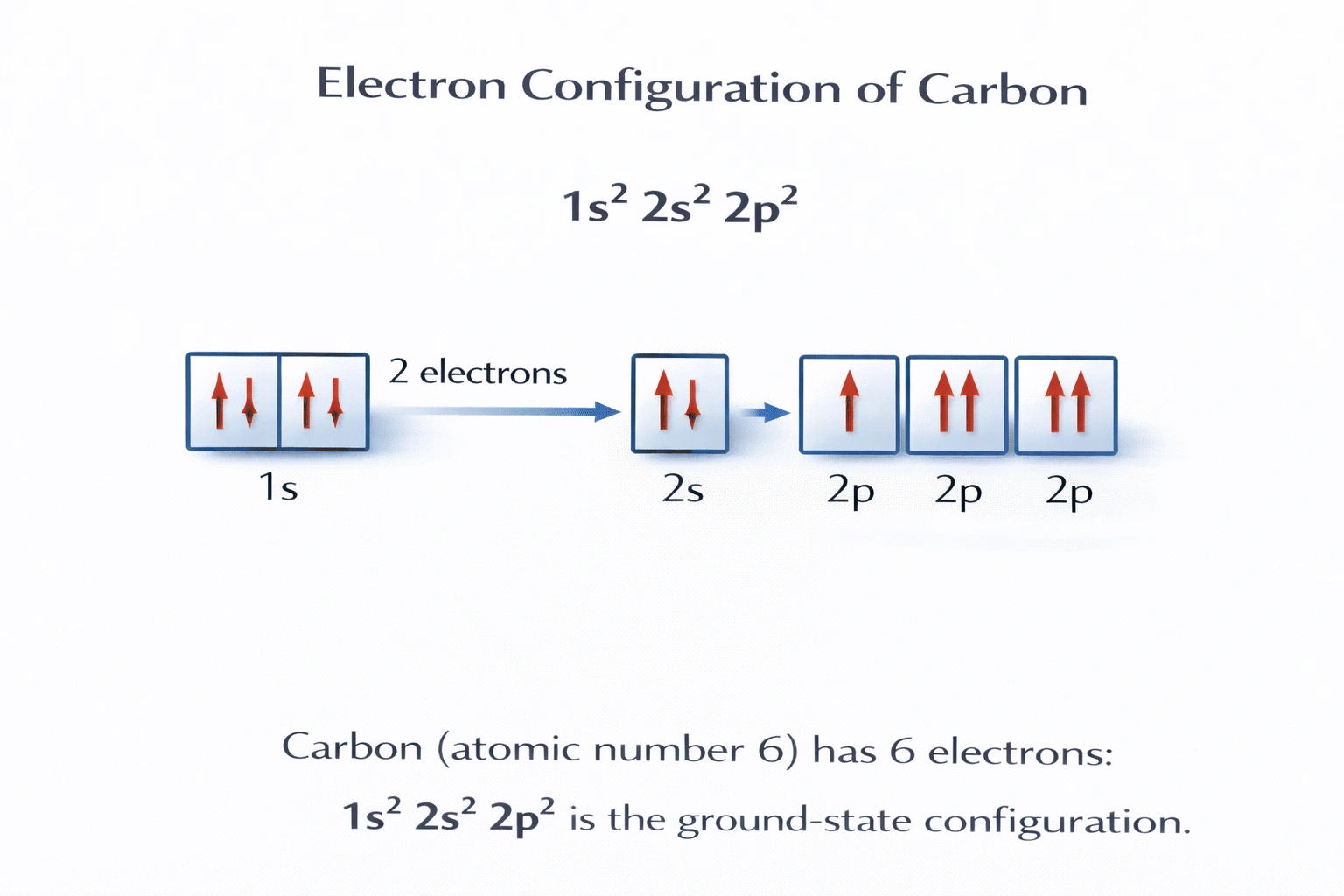
What is the correct electron configuration for carbon?
A.
1s?2s?2p?
B. 1s?2s?2p?
C. 1s?2s?2p?
D. 1s? 2s? 2p? 3s?
Rationale
Carbon has an atomic number of 6, meaning a neutral carbon atom contains six electrons. These electrons fill orbitals in order of increasing energy according to the Aufbau principle: first the 1s orbital, then 2s, and finally the 2p orbitals. The resulting ground-state electron configuration is 1ss²2ss²2ps², with two unpaired electrons in separate 2p orbitals as required by Hund's rule.
A) 1ss²2ss²2p²
This configuration totals five electrons (2 + 2 + 1 = 5), corresponding to boron (atomic number 5), not carbon. Carbon requires six electrons to match its atomic number. This option undercounts the electron total and misassigns the element. It is incorrect.
B) 1ss²2ss²2ps²
This configuration accounts for exactly six electrons: 2 in 1s, 2 in 2s, and 2 in 2p. It follows the correct filling order and satisfies Hund's rule by placing the two 2p electrons in separate orbitals with parallel spins. This matches carbon's position in Period 2, Group 14, and explains its tetravalency through promotion and hybridization. This is the correct answer.
C) 1ss²2ss²2p³
This sums to seven electrons (2 + 2 + 3 = 7), which describes nitrogen (atomic number 7). Carbon cannot have seven electrons in its neutral state. This overcounts and corresponds to the next element in the periodic table. It is incorrect.
D) 1ss²2ss²2p?3s²
This configuration contains eleven electrons (2 + 2 + 6 + 1 = 11), matching sodium (atomic number 11). It also violates the Aufbau principle for carbon by populating the 3s orbital before the 2p subshell is complete, unnecessary for an element with only six electrons. This is chemically implausible for carbon and is incorrect.
Conclusion
The electron configuration of an element is determined by its atomic number and governed by quantum mechanical rules: the Aufbau principle, Pauli exclusion principle, and Hund's rule. For carbon (Z = 6), the only valid ground-state configuration is 1ss²2ss²2ps². Thus, B) 1ss²2ss²2ps² is the correct answer.
To the nearest whole number, what is the mass of one mole of hydrogen chloride?
A.
36 g/mol
B. 38 g/mol
C. 71 g/mol
D. 74 g/mol
Rationale
Hydrogen chloride (HCl) has a molar mass of approximately 36.46 g/mol, calculated by summing the atomic masses of hydrogen (~1.01 g/mol) and chlorine (~35.45 g/mol). Rounded to the nearest whole number, this yields 36 g/mol.
A) 36 g/mol
This is correct. The precise molar mass of HCl is 1.008 + 35.45 = 36.458 g/mol, which rounds to 36 g/mol. This value is consistent with standard periodic table data and is used in stoichiometric calculations involving hydrochloric acid or gaseous HCl.
B) 38 g/mol
This overestimates the molar mass by nearly 2 g/mol. It may result from using an incorrect atomic mass for chlorine (e.g., 37 instead of 35.45), possibly confusing it with the mass number of a chlorine isotope like ³·Cl. However, natural chlorine is a mixture of isotopes (³µCl and ³·Cl), yielding an average atomic mass of ~35.45 g/mol. This option is incorrect.
C) 71 g/mol
This is approximately the molar mass of Cl? (35.45 x 2 ‰ˆ 70.9 g/mol), not HCl. It likely arises from mistakenly using two chlorine atoms instead of one. HCl contains only one Cl atom per molecule, so this value doubles the correct mass and is incorrect.
D) 74 g/mol
This has no clear basis in HCl's composition. It may stem from confusing HCl with another compound like CaCl? (~111 g/mol) or misadding atomic masses. It significantly exceeds the true molar mass and is incorrect.
Conclusion
The molar mass of HCl is the sum of one hydrogen atom and one chlorine atom: 1.01 + 35.45 = 36.46 g/mol ‰ˆ 36 g/mol. Only option A reflects accurate atomic masses and proper rounding. Therefore, A) 36 g/mol is the correct answer.

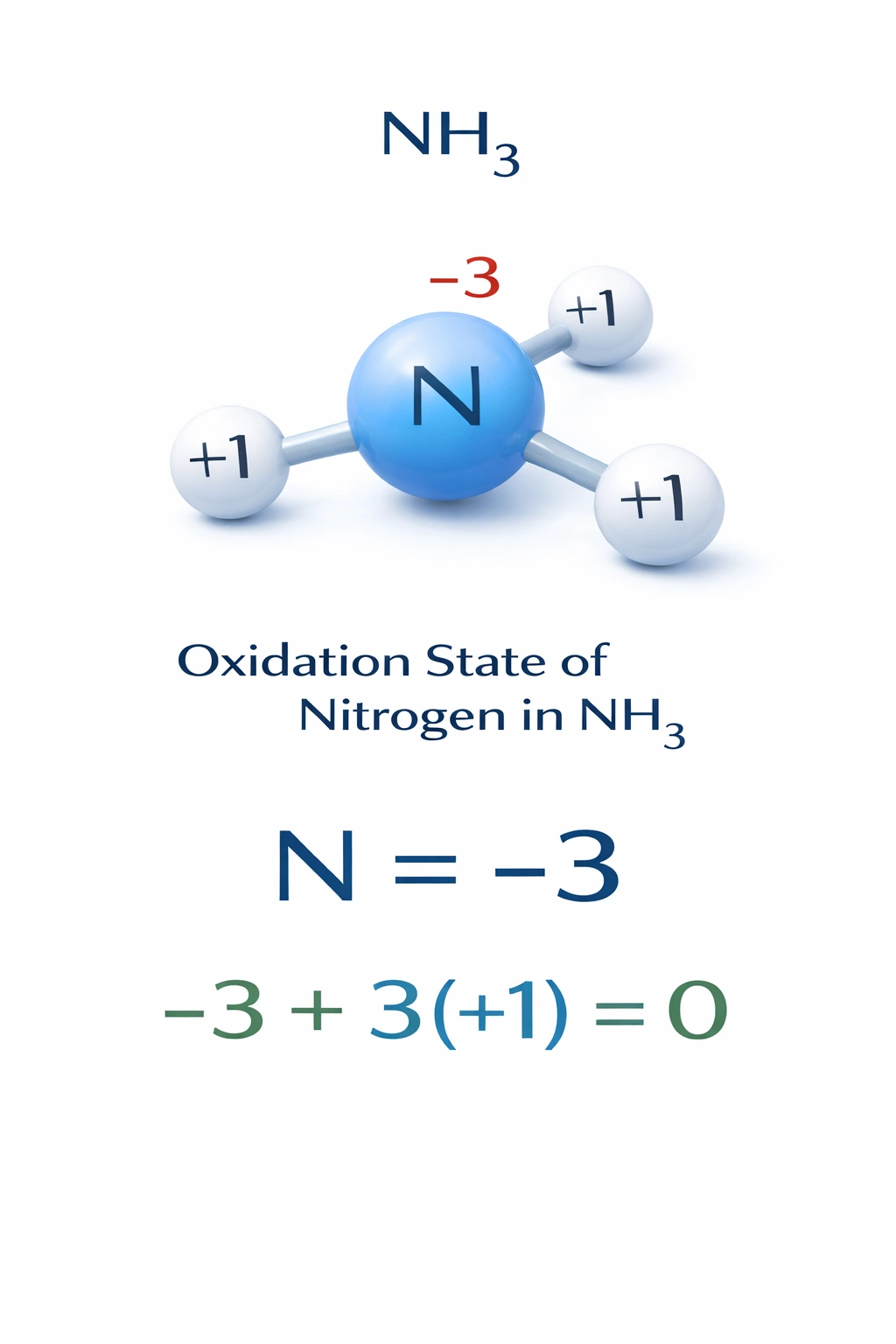
What is the oxidation state of the nitrogen atom in the compound NH??
A.
-3
B. -1
C. 1
D. 3
Rationale
In ammonia (NH?), nitrogen exhibits an oxidation state of -3. This results from nitrogen's higher electronegativity (3.04) compared to hydrogen (2.20), causing it to attract the shared electrons more strongly. In a neutral molecule, the sum of oxidation states must equal zero. With each hydrogen assigned +1, the equation is: N + 3(+1) = 0 ? N = -3.
A) -3
Correct. Nitrogen in NH? has an oxidation state of -3, consistent with its position in Group 15 and its tendency to gain three electrons to achieve a noble gas configuration. This value aligns with standard rules: hydrogen is +1 when bonded to nonmetals, and the more electronegative atom (nitrogen) assumes the negative oxidation state.
B) -1
This would imply an incorrect charge balance: -1 + 3(+1) = +2 ‰ 0. Such a value might apply in hydrazine (N?H?), where each nitrogen is -2, but not in NH?. The stoichiometry of NH? requires nitrogen to be -3 for neutrality. This is incorrect.
C) +1
A +1 oxidation state for nitrogen would mean it is less electronegative than hydrogen, contradicting known electronegativity values. It would also yield +1 + 3(+1) = +4, violating charge neutrality. Nitrogen does not exhibit positive oxidation states when bonded only to hydrogen. This is incorrect.
D) +3
This would give +3 + 3(+1) = +6, far from neutral. Positive oxidation states for nitrogen occur only when bonded to more electronegative elements like oxygen (e.g., +3 in HNO?). In NH?, nitrogen is the most electronegative atom and cannot have a positive oxidation state. This is incorrect.
Conclusion
The oxidation state of nitrogen in NH? is determined by electronegativity and charge balance. Hydrogen is assigned +1, so nitrogen must be -3 to yield a neutral molecule. This reflects nitrogen's electron-gaining behavior in covalent compounds with less electronegative elements. Therefore, A) -3 is the correct answer.
What is the correct formula for sodium nitrate?
A.
Na?NO
B. Na?NO
C. NaNO?
D. NaNO?
Rationale
Sodium nitrate is an ionic compound formed from the sodium cation (Na?) and the nitrate anion (NO??). Sodium, a Group 1 metal, loses one electron to form Na?, while the nitrate ion, a polyatomic ion composed of one nitrogen atom and three oxygen atoms, carries a -1 charge. The charges balance in a 1:1 ratio, yielding the formula NaNO?.
A) Na?NO
This formula is chemically invalid. It implies a nonexistent "NO" anion with a -2 charge, which does not correspond to any known polyatomic ion. Nitrogen and oxygen form stable anions such as nitrite (NO??) and nitrate (NO??), but not "NO." This stoichiometry violates charge balance and chemical reality. It is incorrect.
B) Na?NO
This suggests a -3 charge on a hypothetical "NO" ion, which is not recognized in chemistry. Even if such an ion existed, it would not represent nitrate. Sodium nitrate must contain the NO?? ion, not an undefined NO group. This formula is inconsistent with standard nomenclature and is incorrect.
C) NaNO?
This is the formula for sodium nitrite, not sodium nitrate. Nitrite (NO??) contains two oxygen atoms, whereas nitrate (NO??) contains three. Though both are nitrogen-oxygen anions, they differ in oxidation state (N is +3 in nitrite, +5 in nitrate) and chemical behavior. Confusing nitrite with nitrate is a common error, but the question specifically asks for nitrate. This is incorrect.
D) NaNO?
Correct. The Na? ion (+1) and NO?? ion (-1) combine in a 1:1 ratio to form a neutral compound. The nitrate ion has a trigonal planar structure with resonance stabilization, and its -1 charge is well-established. Sodium nitrate is widely used in fertilizers and food preservation, and its formula is universally accepted as NaNO?.
Conclusion
The correct formula for sodium nitrate is determined by pairing the Na? cation with the NO?? anion in a charge-balanced 1:1 ratio. Only NaNO? reflects this combination accurately. Therefore, D) NaNO? is the correct answer.

HESI A2 Exams
Biology Quizzes
3 Practice Tests
Biology Quizzes
3 Practice Tests
Anatomy Quizzes
3 Practice Tests
Anatomy Quizzes
3 Practice Tests
Reading Quizzes
4 Practice Tests
Reading Quizzes
4 Practice Tests
Grammar Quizzes
3 Practice Tests
Grammar Quizzes
3 Practice Tests
Vocabulary Quizzes
3 Practice Tests
Vocabulary Quizzes
3 Practice Tests
Physics Quizzes
3 Practice Tests
Physics Quizzes
3 Practice Tests
HESI Quizzes
3 Practice Tests
HESI Quizzes
3 Practice Tests