HESI A2 CHEMISTRY PRACTICE QUESTIONS
These HESI A2 Chemistry practice questions allow focused practice on specific chemistry topics tested on the exam. Each question comes with a rationale to improve understanding and retention of key chemical principles.
Topics Covered
Atoms and Elements
Chemical Compounds
Reactions
Solutions
pH and Acidity
00:00
Radioactive isotopes are frequently used in medicine. What kind of half-life would a medical isotope probably have?
A.
Seconds long
B. Days long
C. Year long
D. Many years long
Rationale
Medical isotopes typically possess half-lives on the order of days.
This duration provides sufficient time for production, transportation, administration, and localization within target tissues such as tumors or specific organs. The decay timeframe also ensures radioactivity diminishes to safe levels within days to weeks, balancing diagnostic or therapeutic utility with patient safety.
A) Seconds long
An isotope decaying within seconds would vanish before reaching the intended site of action. Production facilities could not ship such material to hospitals without complete decay en route. Administration protocols require minutes to hours for injection and biodistribution. Extremely rapid decay would also concentrate radiation dose delivery into an unsafe instantaneous burst. These constraints render second-scale half-lives impractical for clinical use.
B) Days long
A half-life spanning days aligns with clinical workflow timelines for radiopharmaceutical handling and imaging procedures. Technetium-99m, the most widely used diagnostic isotope, has a 6-hour half-life, within the day-scale range, allowing same-day imaging with minimal residual exposure. Therapeutic isotopes like iodine-131 (8-day half-life) deliver sustained radiation to thyroid tissue while clearing within weeks. This timeframe supports effective tissue targeting without imposing long-term radiation burden on patients or medical staff.
C) Year long
Isotopes with year-scale half-lives persist in biological systems far beyond clinical necessity. Prolonged internal exposure increases cumulative radiation dose to healthy tissues surrounding the target site. Waste disposal becomes problematic due to extended radioactivity in patient excreta and contaminated materials. Regulatory frameworks restrict such isotopes to specialized applications like permanent brachytherapy seeds, not routine diagnostic or systemic therapeutic use.
D) Many years long
Half-lives extending across decades or centuries create persistent environmental and biological hazards. These isotopes accumulate in ecosystems and human tissues with negligible natural clearance. Medical use would violate the ALARA principle (As Low As Reasonably Achievable) for radiation exposure. Such long-lived radionuclides appear primarily in nuclear waste streams or geological dating, not in patient-facing medical applications.
Conclusion
The optimal medical isotope half-life spans hours to days, long enough for practical logistics and therapeutic effect, yet short enough to limit unnecessary exposure. This balance enables precise diagnostic imaging and targeted radiation therapy while adhering to radiation safety standards. Day-scale decay kinetics represent the established norm across nuclear medicine practice worldwide. Isotopes outside this range introduce logistical barriers or unacceptable risk profiles for routine clinical deployment.

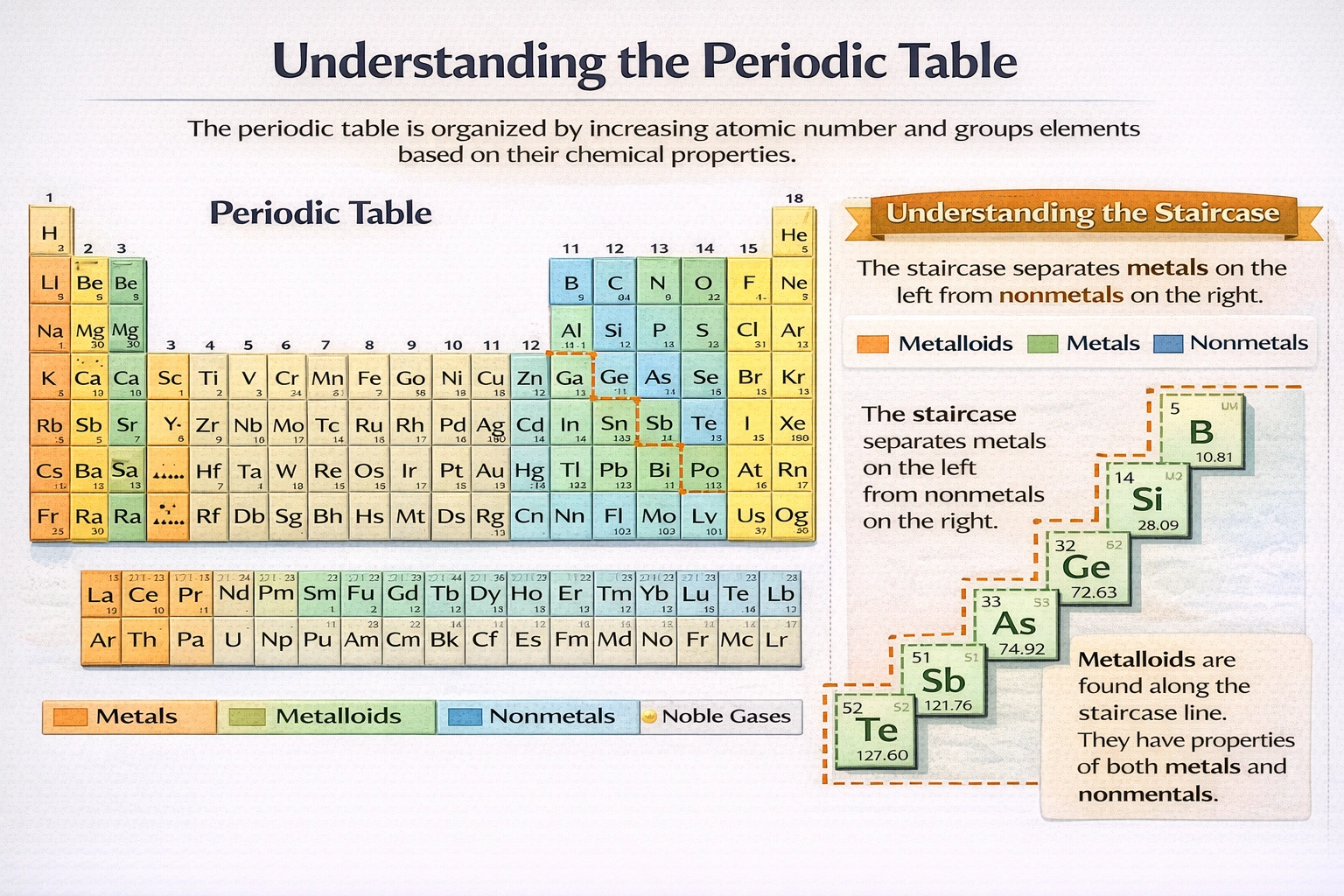
B, Si, As, Te, At, Ge, and Sb form a staircase pattern on the right side of the periodic table. How can these elements be classified?
A.
As metals
B. As semimetals
C. As nonmetals
D. As ultrametals
Rationale
Semimetals, also known as metalloids, form the distinctive staircase pattern separating metals from nonmetals on the periodic table.
The elements boron, silicon, germanium, arsenic, antimony, tellurium, and astatine occupy this diagonal boundary and exhibit intermediate physical and chemical properties. Their electrical conductivity falls between conductors and insulators, making them essential semiconducting materials in modern electronics.
A) As metals
Metals dominate the left side and center of the periodic table, characterized by luster, malleability, ductility, and high electrical conductivity. These staircase elements lack consistent metallic luster and malleability, boron is brittle, silicon shatters under stress. Their electrical conductivity is orders of magnitude lower than true metals and often increases with temperature, opposite to metallic behavior. These elements do not form cations readily in aqueous solution as typical metals do.
B) As semimetals
Semimetals occupy the diagonal boundary between metals and nonmetals, forming the characteristic staircase beginning at boron and extending to astatine. These elements display hybrid properties: silicon and germanium conduct electricity better than nonmetals but worse than metals, with conductivity tunable through doping. Arsenic and antimony show metallic appearance yet brittle mechanical behavior. Tellurium conducts electricity anisotropically, varying with crystal direction. This intermediate behavior defines the metalloid classification central to periodic table organization.
C) As nonmetals
Nonmetals cluster in the upper-right portion of the periodic table, including carbon, nitrogen, oxygen, phosphorus, sulfur, and the halogens. They typically exist as gases or brittle solids with poor thermal and electrical conductivity. Nonmetals gain electrons to form anions and create acidic oxides. The staircase elements contradict these traits, antimony and tellurium exhibit metallic luster, arsenic conducts electricity moderately, and several form amphoteric oxides rather than purely acidic ones. Their position and properties place them distinctly apart from true nonmetals.
D) As ultrametals
The term "ultrametal" has no recognition in chemical nomenclature or periodic table classification systems. Standard categories include metals, nonmetals, and semimetals (metalloids), with additional subgroups like alkali metals or halogens. No peer-reviewed chemistry literature or IUPAC documentation employs "ultrametal" as a classification. This term likely represents either a distractor option or confusion with unrelated concepts such as ultracold atomic physics.
Conclusion
The staircase elements represent the transitional zone where metallic character diminishes across a period and increases down a group. Their intermediate conductivity, mixed physical properties, and position on the periodic table define them unequivocally as semimetals. This classification underpins their technological importance in semiconductor devices and reflects fundamental periodic trends in elemental behavior. No other category accurately captures their hybrid chemical identity.
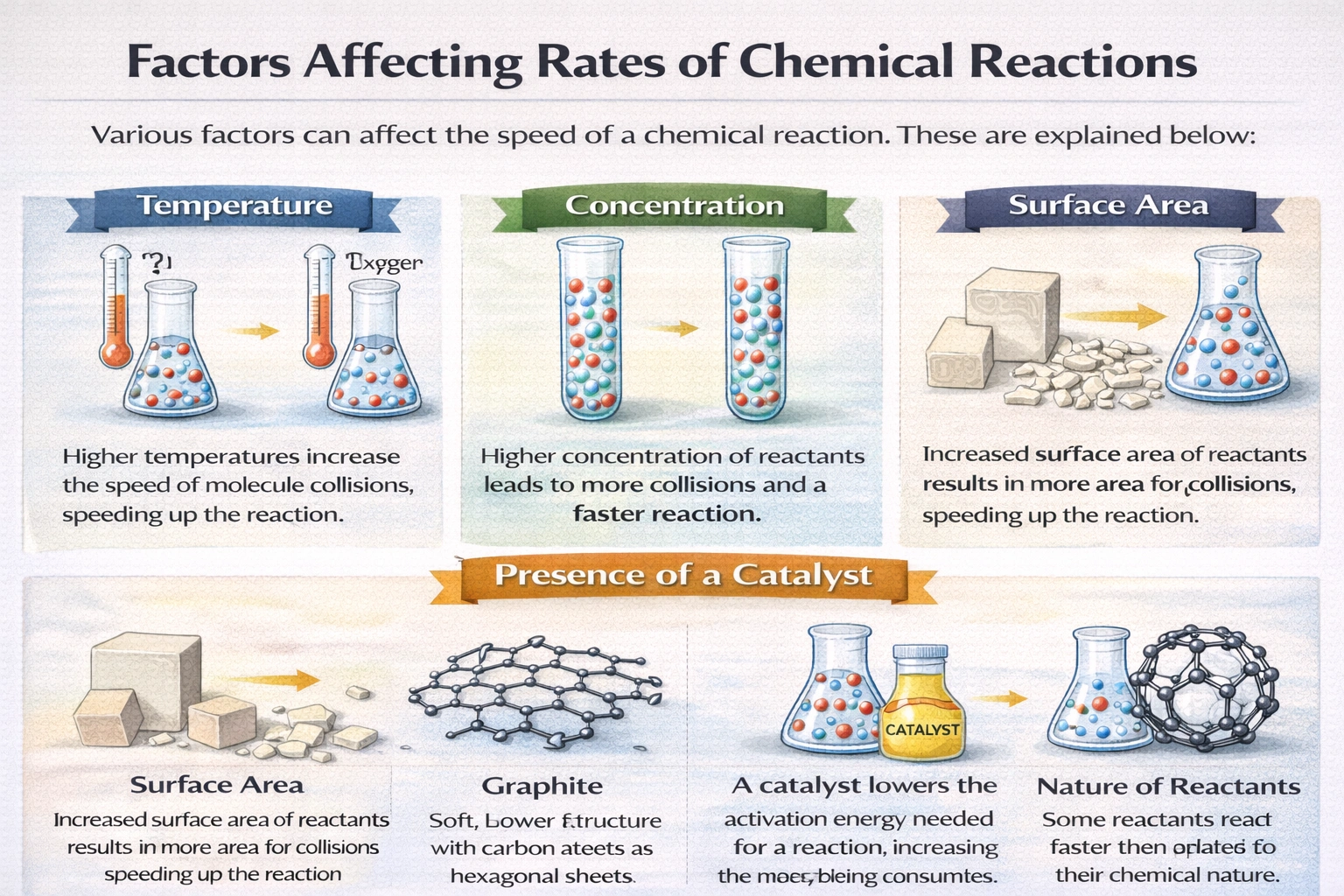
Which of these factors would not affect rates of reaction?
A.
Temperature
B. Surface area
C. Pressure
D. Time
Rationale
Time does not affect reaction rates. Reaction rate represents the speed at which reactants convert to products per unit time, making time a measurement dimension rather than an influencing variable. Factors such as temperature, concentration, and surface area alter molecular collision frequency or energy, whereas time merely quantifies reaction progress without causally modifying the rate itself.
A) Temperature
Temperature directly influences molecular kinetic energy and collision frequency. A ten-degree Celsius increase typically doubles reaction rates for many chemical processes. Higher temperatures shift the Maxwell-Boltzmann distribution, increasing the fraction of molecules exceeding the activation energy threshold. This relationship is quantified by the Arrhenius equation, confirming temperature as a primary kinetic control variable.
B) Surface area
Surface area governs reactant exposure in heterogeneous reactions involving solids. Grinding a solid into fine particles exponentially increases available reaction sites for collision with liquids or gases. Powdered zinc reacts visibly faster with hydrochloric acid than an equivalent-mass zinc pellet due to greater interfacial contact. This geometric factor directly scales with observable reaction velocity in multiphase systems.
C) Pressure
Pressure affects reaction rates exclusively in gaseous systems by altering effective concentration. Compressing a gas increases molecules per unit volume, elevating collision frequency proportionally. For reactions with unequal moles of gaseous reactants and products, pressure shifts also influence equilibrium position per Le Chatelier's principle. This concentration-mediated effect makes pressure a legitimate rate-modifying factor for gas-phase kinetics.
D) Time
Time serves as the independent variable against which concentration changes are measured in rate calculations. Reaction rate is defined mathematically as Δ[concentration]/Δt, positioning time in the denominator as a reference frame. While concentrations change over time as reactants deplete, time itself exerts no causal influence on molecular collision dynamics or activation barriers. No experimental manipulation of time alone alters instantaneous reaction velocity under constant conditions.
Conclusion
Reaction rates respond to variables that modify molecular collision frequency, energy distribution, or activation barriers. Temperature, surface area, and pressure each directly influence these microscopic parameters. Time functions solely as a metric for tracking progress rather than an active kinetic variable. Distinguishing between causal factors and measurement dimensions remains essential for accurate kinetic analysis and experimental design.
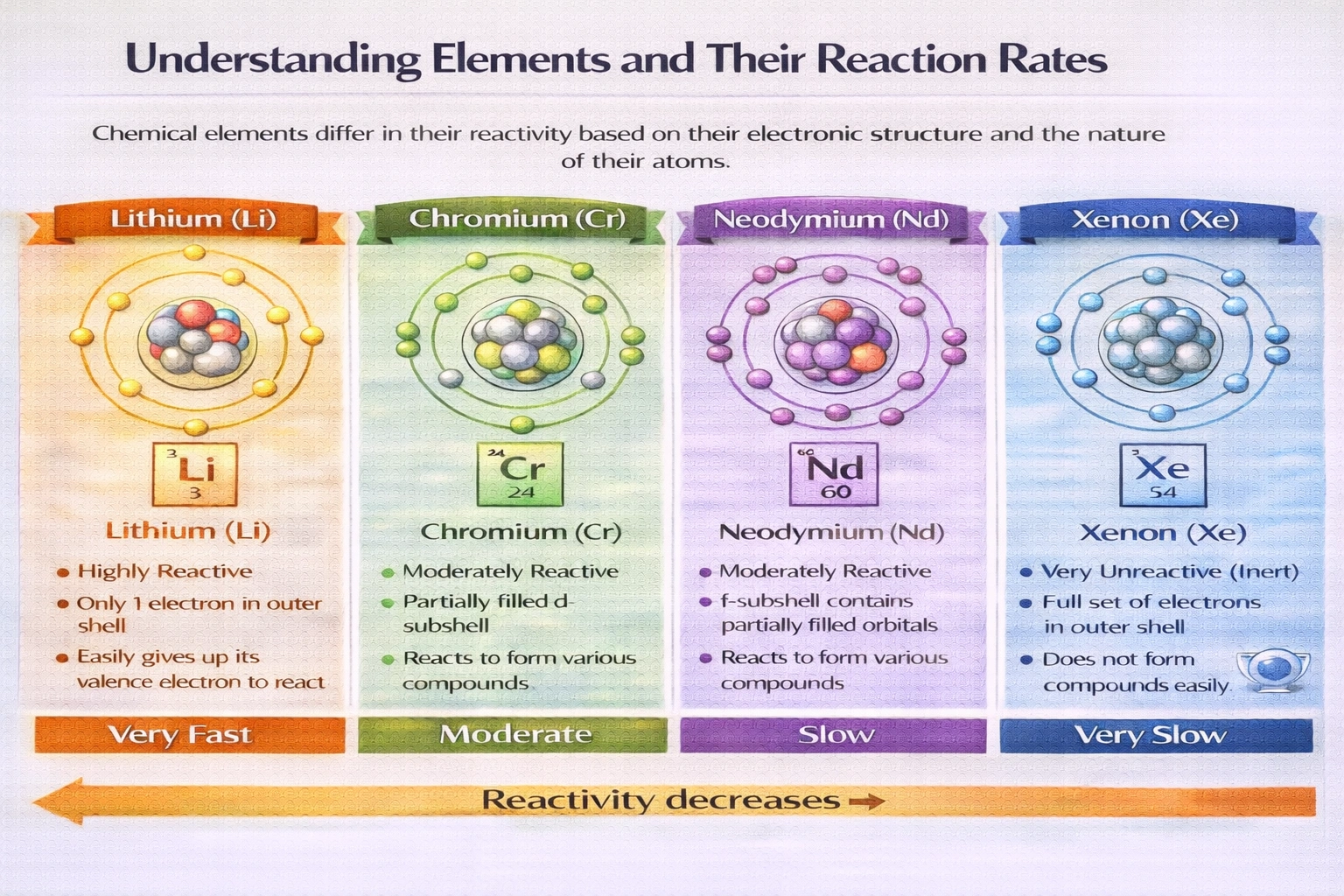
Which element would you expect to be least reactive?
A.
Li
B. Cr
C. Nd
D. Xe
Rationale
Xenon exhibits the lowest reactivity among these elements. As a noble gas in Group 18, it possesses a complete valence electron shell with eight electrons, satisfying the octet rule and conferring exceptional thermodynamic stability. This electronic configuration minimizes tendency to gain, lose, or share electrons under standard conditions.
A) Li
Lithium resides in Group 1 as an alkali metal with a single valence electron. It readily donates this electron to achieve noble gas configuration, forming Li? ions with high exothermicity. Lithium reacts vigorously with water to produce hydrogen gas and lithium hydroxide. It oxidizes rapidly in air, requiring storage under mineral oil to prevent reaction with atmospheric moisture and oxygen. Its low ionization energy (520 kJ/mol) reflects minimal resistance to electron loss.
B) Cr
Chromium is a transition metal exhibiting multiple stable oxidation states including +2, +3, and +6. It forms coordination complexes with ligands such as water, ammonia, and chloride ions. Chromium metal reacts slowly with oxygen to form a protective oxide layer but dissolves in strong acids to release hydrogen gas. Its partially filled d-orbitals enable variable bonding behavior and catalytic activity in redox reactions. Chromium compounds participate readily in electron transfer processes.
C) Nd
Neodymium belongs to the lanthanide series with electron configuration [Xe]4f´6ss². It tarnishes rapidly in air, forming an oxide coating that spalls off to expose fresh metal surface. Neodymium reacts slowly with cold water and more rapidly with steam to produce neodymium hydroxide and hydrogen gas. It ignites spontaneously when heated in air, burning to form Nd?O?. Its electropositive character drives ready participation in ionic compound formation.
D) Xe
Xenon contains eight valence electrons arranged in a stable octet configuration (5ss²5p?). Its first ionization energy measures 1,170 kJ/mol, significantly higher than lithium's 520 kJ/mol, reflecting strong electron retention. Xenon remains inert toward water, oxygen, acids, and bases under ambient conditions. Compound formation requires extreme measures: high pressure, electrical discharge, or highly electronegative partners like fluorine to produce XeF?, XeF?, or XeO?. These compounds decompose readily, underscoring xenon's inherent stability.
Conclusion
Noble gases occupy Group 18 with fully occupied valence shells that resist perturbation under ordinary conditions. Xenon's complete octet creates a substantial energy barrier to electron transfer or sharing, rendering it effectively inert compared to metals that readily oxidize. While forced compound formation is possible under extraordinary circumstances, xenon's baseline reactivity remains negligible relative to alkali metals, transition metals, and lanthanides. This electronic stability defines noble gas behavior across the periodic table.
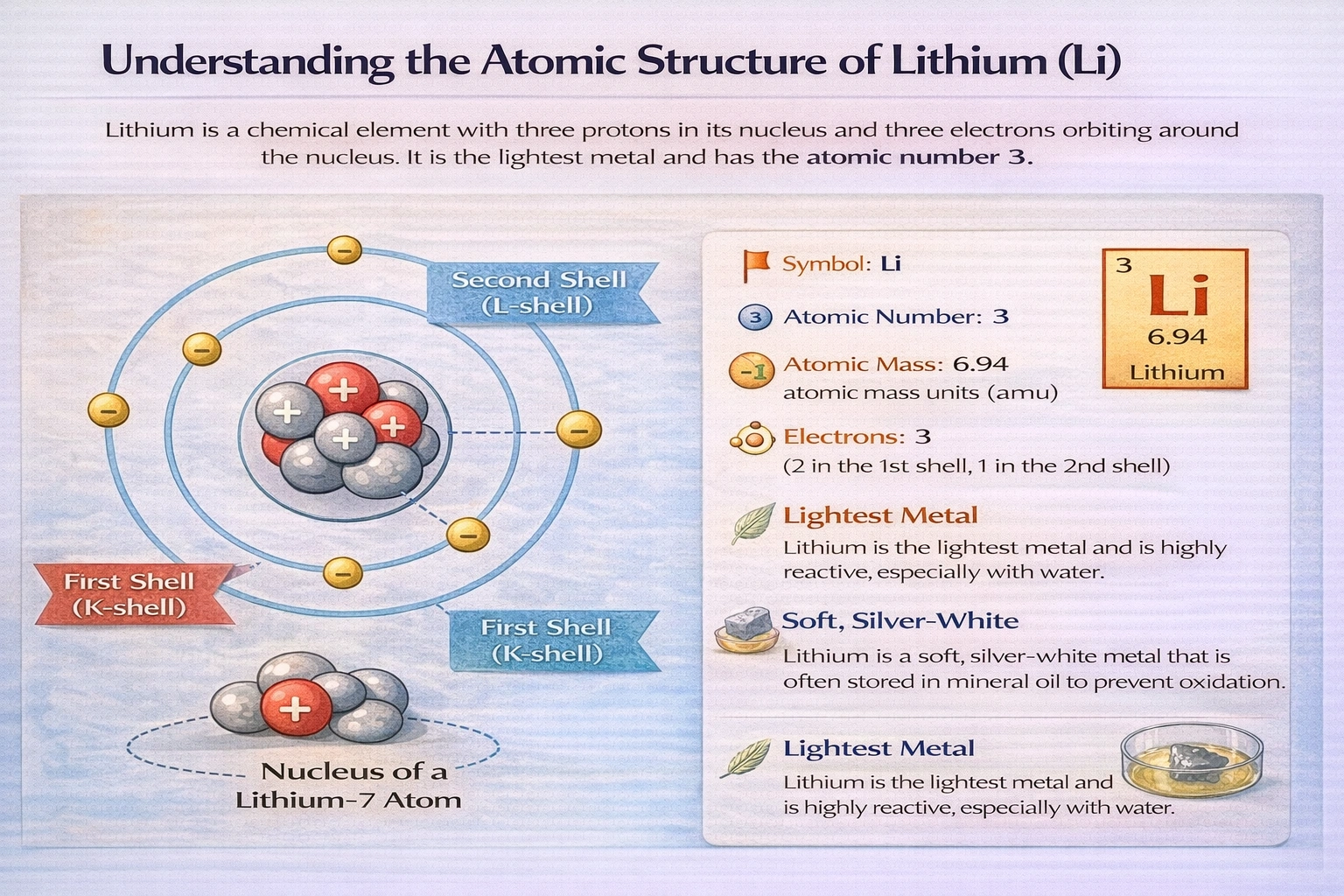
What is the correct electron configuration for lithium?
A.
1s?2s?
B. 1s?2s?
C. 1s?2s?2p?
D. 1s?2s?2p?
Rationale
Lithium's electron configuration is 1ss²2s². With an atomic number of 3, lithium contains three electrons that occupy the lowest available energy orbitals according to the Aufbau principle. The first two electrons fill the 1s orbital completely, and the third electron occupies the next lowest energy orbital, 2s, resulting in the ground state configuration 1ss²2s².
A) 1ss²2s²
Lithium possesses three electrons distributed across principal quantum levels n=1 and n=2. The 1s orbital accommodates two electrons with opposite spins, satisfying the Pauli exclusion principle. The remaining electron occupies the 2s orbital, which has higher energy than 1s but lower than 2p orbitals. This arrangement minimizes total energy and represents lithium's ground electronic state. Spectroscopic measurements confirm this configuration through characteristic emission lines. The single valence electron in the 2s orbital explains lithium's +1 oxidation state and position in Group 1 of the periodic table.
B) 1ss²2ss²
This configuration contains four electrons total, two in 1s and two in 2s. Lithium's atomic number of 3 limits its neutral atom to exactly three electrons. The 1ss²2ss² arrangement corresponds to beryllium (atomic number 4), not lithium. Adding a fourth electron to lithium would create the Li? anion, not the neutral atom. Neutral lithium cannot accommodate two electrons in the 2s orbital while maintaining charge neutrality.
C) 1ss²2s²2p²
This arrangement distributes four electrons across three orbitals, exceeding lithium's three-electron count. The configuration violates the Aufbau principle, which requires the 2s orbital to fill completely before electrons occupy higher-energy 2p orbitals. Even if lithium possessed four electrons, the fourth would enter 2ss² rather than 2p² due to energy ordering. The 2p orbital remains unoccupied in lithium's ground state and first excited states under normal conditions.
D) 1s²2s²2ps²
This configuration contains four electrons with improper orbital filling sequence. The 1s orbital must contain two electrons before any electrons occupy n=2 orbitals, as 1s represents the lowest energy state. Placing only one electron in 1s while populating higher orbitals contradicts quantum mechanical energy minimization. No neutral atom with atomic number 3 adopts this arrangement; it violates both electron count requirements and fundamental orbital filling rules.
Conclusion
Lithium's three electrons distribute as two in the 1s orbital and one in the 2s orbital, yielding the configuration 1ss²2s². This arrangement satisfies the Aufbau principle, Pauli exclusion principle, and observed atomic number constraints. Alternative configurations either contain incorrect electron counts or violate orbital energy ordering rules. The single 2s valence electron defines lithium's chemical behavior and periodic table placement as the lightest alkali metal.
To the nearest whole number, what is the mass of one mole of water?
A.
16 g/mol
B. 18 g/mol
C. 20 g/mol
D. 22 g/mol
Rationale
The molar mass of water is 18 g/mol to the nearest whole number. Water's molecular formula H?O contains two hydrogen atoms (1.008 g/mol each) and one oxygen atom (16.00 g/mol). Summing these values yields 2.016 + 16.00 = 18.016 g/mol, which rounds to 18 g/mol when expressed as a whole number.
A) 16 g/mol
This value corresponds to the atomic mass of oxygen alone. Oxygen constitutes the majority of water's mass but does not account for hydrogen's contribution. Water molecules contain two additional hydrogen atoms contributing approximately 2 g/mol to the total. Using 16 g/mol ignores the complete molecular composition of H?O. This mass represents elemental oxygen, not the compound water.
B) 18 g/mol
Hydrogen contributes 1.008 g/mol per atom, yielding 2.016 g/mol for the two hydrogen atoms in water. Oxygen contributes 16.00 g/mol as a single atom per water molecule. The precise sum equals 18.016 g/mol, which rounds to 18 g/mol at whole-number precision. This value aligns with experimental measurements of water's molecular weight using mass spectrometry and colligative property determinations. Standard chemistry references consistently list water's molar mass as 18.0 g/mol for stoichiometric calculations.
C) 20 g/mol
This value exceeds water's actual molar mass by approximately 2 g/mol. No standard isotopic composition of water yields this mass, deuterium oxide (D?O) reaches only 20.028 g/mol with heavy hydrogen isotopes. Ordinary water contains protium (²H), not deuterium, as its predominant hydrogen isotope. Adding two extra mass units lacks chemical basis for naturally occurring water molecules.
D) 22 g/mol
This value substantially overestimates water's molecular mass by 4 g/mol. It might arise from incorrectly summing atomic masses or incorporating non-existent atoms into the formula. No common water isotopologue, whether H?O, HDO, or D?O, approaches 22 g/mol under normal conditions. Such a mass would require incorporation of additional oxygen or heavy isotopes not present in standard water.
Conclusion
Water's molar mass derives directly from summing the atomic masses of its constituent atoms: two hydrogens and one oxygen. The calculated value of 18.016 g/mol rounds conventionally to 18 g/mol for whole-number representation. This mass underpins stoichiometric relationships throughout aqueous chemistry and thermodynamics. Precise knowledge of this value enables accurate solution preparation, reaction balancing, and quantitative analysis in laboratory practice.

What is the oxidation state of the chlorine atom in the compound HCl?
A.
1
B. -1
C. 2
D. -2
Rationale
The chlorine atom in HCl exhibits a -1 oxidation state. Hydrogen assumes a +1 oxidation state when bonded to nonmetals, and the neutral compound requires the sum of oxidation states to equal zero. Chlorine's higher electronegativity (3.16 versus hydrogen's 2.20) draws the bonding electron pair toward itself, resulting in the negative oxidation state.
A) +1
A +1 oxidation state occurs when chlorine bonds to more electronegative elements such as oxygen in hypochlorous acid (HClO). Hydrogen ranks lower in electronegativity than chlorine, making electron withdrawal by hydrogen impossible. Positive chlorine oxidation states require oxygen or fluorine as bonding partners. Binary hydrogen-chlorine compounds cannot support chlorine in positive oxidation states under standard conditions.
B) -1
Hydrogen contributes +1 to the oxidation state sum in compounds with nonmetals. The neutral HCl molecule requires chlorine to balance this with -1 to satisfy the zero-sum rule. Chlorine's position in Group 17 gives it seven valence electrons and strong electron affinity. The bonding pair localizes closer to chlorine, effectively assigning both electrons to chlorine in oxidation state formalism. This -1 state represents chlorine's most common and stable oxidation state across ionic and covalent compounds.
C) +2
Chlorine achieves +2 oxidation state only in rare compounds like chlorine dioxide (ClO?) under specific conditions. This state requires multiple bonds to highly electronegative oxygen atoms. Hydrogen cannot oxidize chlorine to positive states due to its lower electronegativity. Binary hydrogen-chlorine systems lack the electronic environment necessary for chlorine oxidation beyond zero or negative values.
D) -2
The -2 oxidation state characterizes Group 16 elements like oxygen and sulfur in many compounds. Chlorine's seven valence electrons require gaining only one electron to achieve noble gas configuration. Gaining two electrons would produce an unstable Cls²? ion with unfavorable electron-electron repulsion. No common chlorine compounds exhibit -2 oxidation state under normal chemical conditions.
Conclusion
Oxidation state assignment follows electronegativity hierarchy and charge balance requirements in neutral molecules. Hydrogen's +1 state combined with HCl's neutrality mandates chlorine at -1. This assignment aligns with chlorine's position as the more electronegative partner and its tendency to gain one electron for stable octet completion. The -1 oxidation state represents the thermodynamically favored electron distribution in hydrogen chloride.

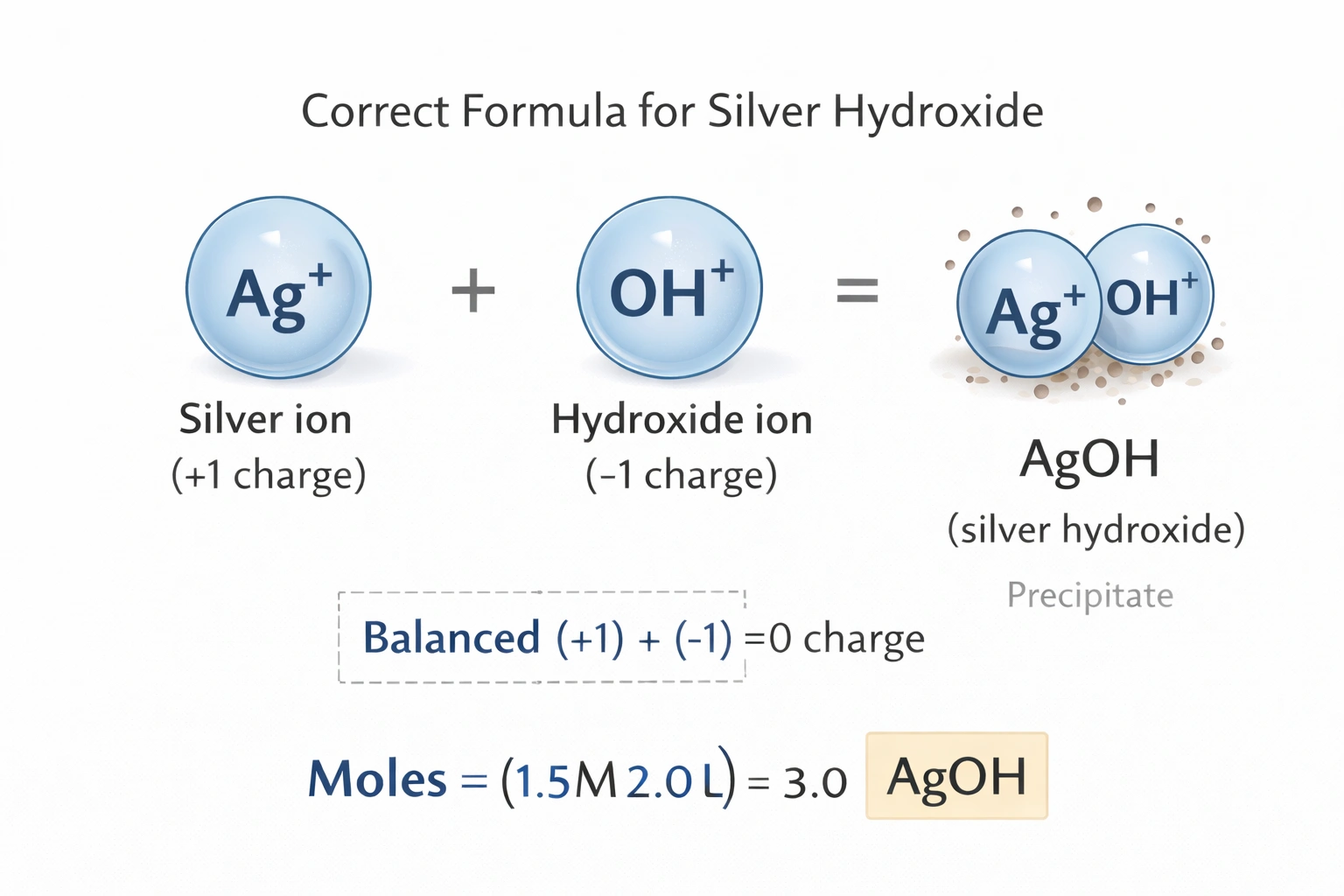
What is the correct formula for silver hydroxide?
A.
AgO?
B. AgOH
C. AgOH?
D. AgH?O
Rationale
Silver hydroxide has the chemical formula AgOH. Silver commonly exhibits a +1 oxidation state, forming Ag? cations that combine with hydroxide anions (OH?) in a one-to-one ratio to achieve charge neutrality. This stoichiometry produces a simple ionic compound where electrostatic attraction balances the single positive and single negative charges.
A) AgO?
This formula lacks hydrogen atoms entirely, contradicting hydroxide's essential H-O composition. AgO? might represent silver peroxide containing O?s²? ions or silver(II) oxide with mixed oxidation states. Hydroxide specifically requires the hydroxyl functional group (-OH) with covalently bonded hydrogen and oxygen. No stable silver compound with formula AgO? corresponds to hydroxide chemistry or standard silver oxidation states.
B) AgOH
Silver predominantly forms +1 cations (Ag?) in ionic compounds due to its electron configuration [Kr]4d²°5s° after ionization. The hydroxide ion carries a -1 charge (OH?) with oxygen covalently bonded to hydrogen. Charge balance requires exactly one Ag? to pair with one OH?, yielding the empirical formula AgOH. This compound exists as a dark brown precipitate formed when silver salts react with strong bases, though it decomposes readily to silver oxide and water.
C) AgOH?
This notation incorrectly suggests two hydrogen atoms bonded to oxygen in the anion, contradicting hydroxide's fixed OH? structure. AgOH? might imply Ags²? paired with two hydroxides, but silver(II) is rare and unstable in simple hydroxides. Water ligands in coordination complexes use notation like [Ag(H?O)?]?, not AgOH?. No recognized silver hydroxide compound adopts this stoichiometry under standard conditions.
D) AgH?O
This arrangement resembles a coordination compound with water ligands rather than hydroxide ions. H?O represents neutral water molecules, not the anionic OH? required for hydroxide formation. Silver aqua complexes exist as [Ag(H?O)‚™]? cations in solution but do not constitute silver hydroxide. Hydroxide specifically requires deprotonated water (OH?), not intact H?O molecules bonded to silver.
Conclusion
Silver's consistent +1 oxidation state combines with the monovalent hydroxide anion to produce a 1:1 ionic compound. The formula AgOH satisfies charge balance requirements and aligns with experimental observations of silver-base reactions. Alternative formulations either misrepresent hydroxide's chemical identity or invoke unstable silver oxidation states not characteristic of simple hydroxide compounds. Precise formula notation distinguishes hydroxide (OH?) from water (H?O) and other oxygen-containing anions.
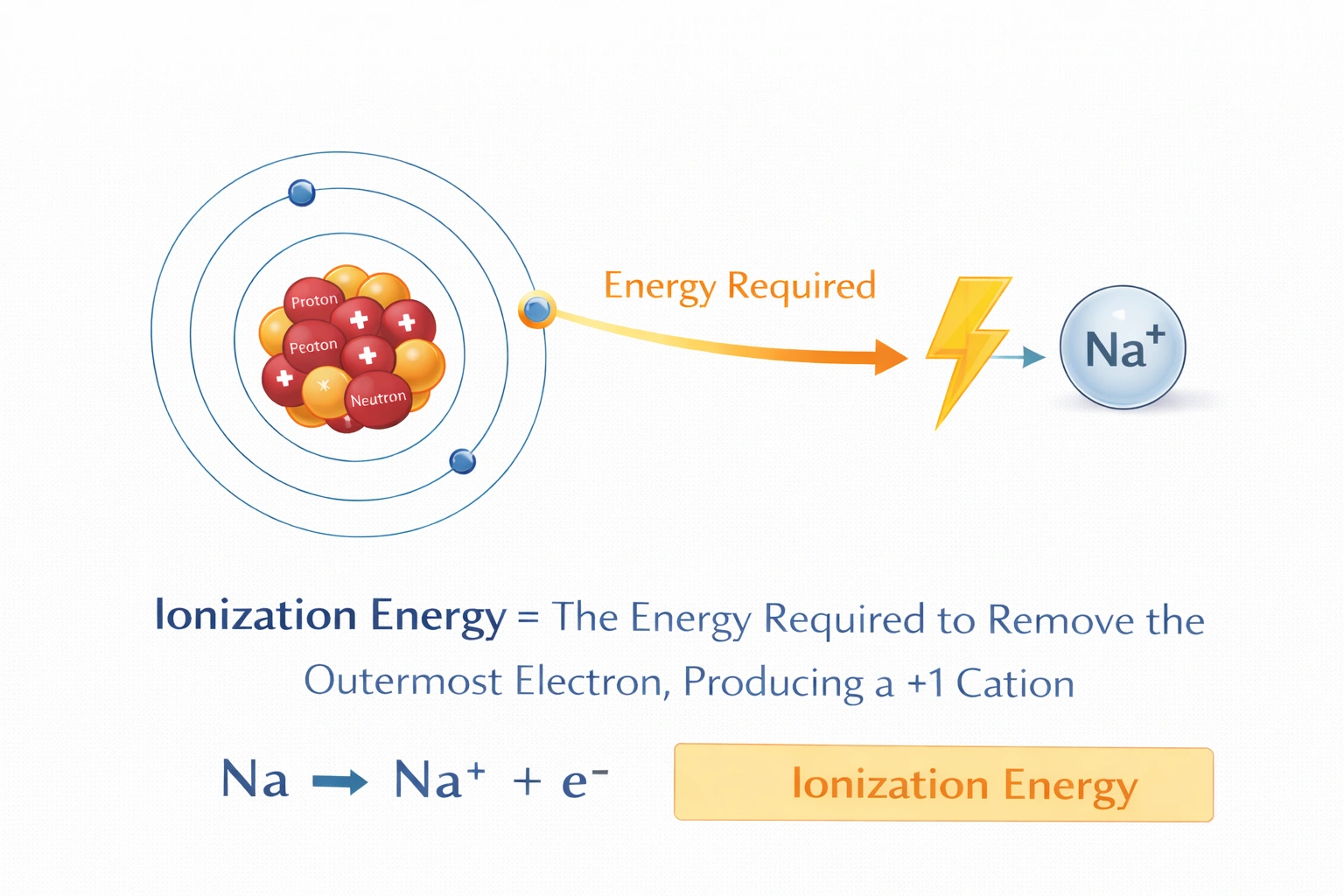
The energy required to remove the outermost electron from an atom is called __________.
A.
covalent bonding
B. electronegativity
C. atomic radius
D. ionization energy
Rationale
The energy required to remove the outermost electron from an atom is called ionization energy. This quantity represents the minimum energy input needed to overcome electrostatic attraction between the nucleus and a valence electron in a gaseous, ground-state atom. First ionization energy specifically refers to removal of the most loosely bound electron, producing a +1 cation.
A) Covalent bonding
Covalent bonding describes electron sharing between atoms to achieve stable electron configurations. This interaction forms discrete molecules through orbital overlap and shared electron pairs. Bond formation releases energy rather than requiring electron removal. Covalent bonds involve mutual electron attraction between nuclei, not complete electron ejection from an atom. This concept addresses molecular stability, not atomic ionization processes.
B) Electronegativity
Electronegativity quantifies an atom's tendency to attract shared electrons within a chemical bond. Measured on relative scales such as Pauling or Mulliken, it predicts bond polarity and electron distribution in molecules. Electronegativity describes electron attraction during bonding events, not energy requirements for electron removal. This property influences bond character but does not represent an absolute energy value for ionization.
C) Atomic radius
Atomic radius measures the approximate distance from an atomic nucleus to its outermost electron shell boundary. Values typically range from 30 to 300 picometers depending on element and measurement method. Radius trends inversely with ionization energy across the periodic table due to varying nuclear attraction distances. While radius influences ionization energy magnitude, it represents spatial dimension rather than energy quantity. This parameter describes atomic size, not energy thresholds.
D) Ionization energy
Ionization energy is defined as the minimum energy required to remove an electron from an isolated gaseous atom in its ground electronic state. The first ionization energy specifically targets the outermost valence electron, producing a singly charged positive ion. This endothermic process requires energy input to overcome Coulombic attraction between the positively charged nucleus and negatively charged electron. Periodic trends show ionization energy increasing left to right across periods and decreasing down groups, reflecting nuclear charge and electron shielding effects.
Conclusion
Ionization energy provides the precise thermodynamic measure for electron removal from neutral atoms. This fundamental atomic property governs chemical reactivity, metallic character, and periodic trends in elemental behavior. Alternative terms describe bonding interactions, electron attraction tendencies, or spatial dimensions rather than ionization thresholds. Recognition of ionization energy as the specific descriptor for electron removal energy remains essential for understanding atomic structure and chemical periodicity.
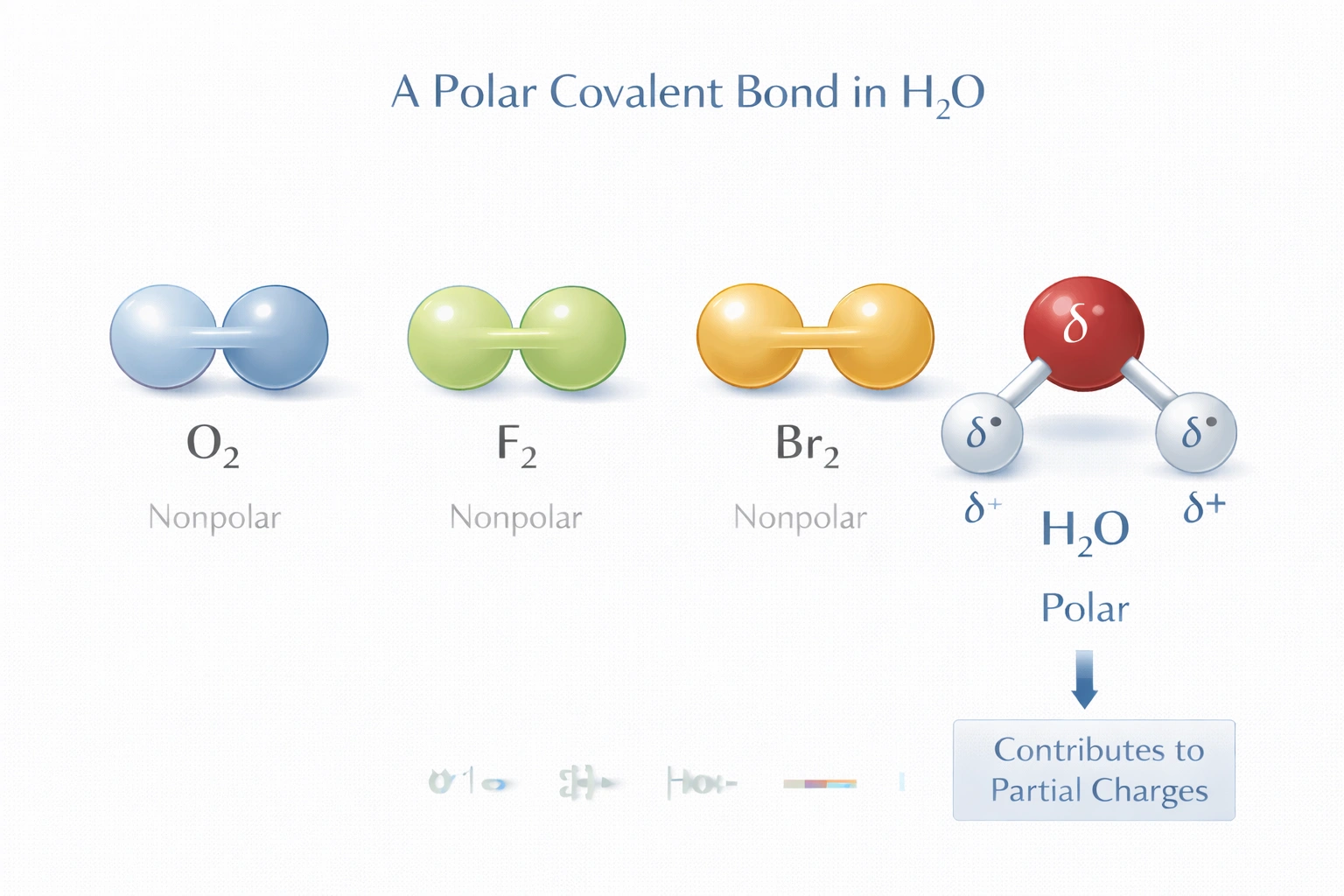
Which compound contains a polar covalent bond?
A.
O?
B. F?
C. Br?
D. H?O
Rationale
Polar covalent bonds form when atoms with different electronegativities share electrons unequally. Water (H?O) contains polar covalent bonds between oxygen and hydrogen atoms due to oxygen's significantly higher electronegativity. This unequal electron distribution creates partial charges within the molecule, with oxygen carrying a partial negative charge and each hydrogen carrying a partial positive charge.
A) O?
Oxygen molecules consist of two identical oxygen atoms bonded together. Both atoms possess identical electronegativity values of 3.44 on the Pauling scale. The bonding electron pair distributes symmetrically between the atoms with no charge separation. This equal sharing produces a nonpolar covalent bond characteristic of homonuclear diatomic molecules. No dipole moment exists in the O? molecule.
B) F?
Fluorine molecules contain two identical fluorine atoms with matching electronegativity values of 3.98. The shared electron pair occupies a symmetrical bonding orbital centered between the nuclei. Equal electron distribution prevents formation of partial charges or bond polarity. Despite fluorine's high absolute electronegativity, the absence of an electronegativity difference between identical atoms yields a nonpolar covalent bond. F? exhibits zero dipole moment.
C) Br?
Bromine molecules comprise two identical bromine atoms each with electronegativity 2.96. The bonding electrons distribute evenly across the internuclear region without preferential accumulation near either atom. Symmetrical electron sharing eliminates charge separation and bond polarity. Like other homonuclear diatomic elements, Br? forms a pure nonpolar covalent bond with no permanent dipole. The molecule remains electrically neutral throughout its structure.
D) H?O
Water molecules contain two O-H bonds where oxygen (electronegativity 3.44) exceeds hydrogen's value (2.20) by 1.24 units. This difference causes bonding electrons to concentrate closer to oxygen, creating δ? on oxygen and δ? on each hydrogen. The bent molecular geometry (104.5° bond angle) prevents dipole cancellation, yielding a substantial net molecular dipole moment of 1.85 D. These polar covalent bonds govern water's high boiling point, surface tension, and solvent properties.
Conclusion
Polar covalent bonding requires both electron sharing and significant electronegativity differences between bonded atoms. Homonuclear diatomic molecules (O?, F?, Br?) lack electronegativity differences and form nonpolar bonds exclusively. Water uniquely combines heteronuclear bonding with sufficient electronegativity disparity to generate polar covalent bonds. This fundamental distinction explains water's anomalous physical properties compared to nonpolar molecular substances.
The three important allotropes of phosphorus are red, white, and __________.
A.
green
B. gray
C. black
D. silver
Rationale
The three important allotropes of phosphorus are white, red, and black. Black phosphorus is the most thermodynamically stable form at room temperature and exhibits a layered, graphite-like structure with semiconducting properties. It contrasts sharply with the highly reactive, toxic white phosphorus and the amorphous, moderately stable red phosphorus used in safety matches.
A) Green
No scientifically recognized allotrope of phosphorus is designated as "green phosphorus." While certain phosphorus compounds or impure samples may appear greenish under specific lighting, this does not represent a distinct structural form. The term lacks basis in crystallographic or chemical literature and does not correspond to any known phase of elemental phosphorus.
B) Gray
Although black phosphorus can exhibit a grayish metallic luster due to its layered structure, the correct and universally accepted name for this allotrope is black phosphorus. The term "gray phosphorus" is occasionally used informally but is not standard in chemical nomenclature or textbooks. IUPAC and major reference sources consistently refer to it as black phosphorus, distinguishing it by its orthorhombic or rhombohedral crystalline forms.
C) Black
Black phosphorus is produced by heating white phosphorus under high pressure or by catalytic conversion of red phosphorus. Its atoms arrange in puckered layers held by van der Waals forces, allowing anisotropic electrical conductivity and semiconductor behavior. Unlike white phosphorus (which ignites in air) or red phosphorus (which is air-stable but amorphous), black phosphorus is the least reactive and most dense allotrope, making it valuable in materials science and electronics.
D) Silver
"Silver phosphorus" is not a valid allotrope. The descriptor may arise from confusion with the shiny appearance of black phosphorus or with metallic elements like silver itself. Elemental phosphorus does not form a silvery allotrope; even its most metallic-looking form, black phosphorus, is classified by its structure and reactivity, not by color alone. No peer-reviewed source recognizes a silver-colored or silver-named phosphorus allotrope.
Conclusion
The three principal allotropes of phosphorus, white, red, and black, are defined by distinct molecular structures, stabilities, and physical properties. White phosphorus consists of P? tetrahedra, red phosphorus forms polymeric chains, and black phosphorus adopts a layered lattice. Among the options, only black correctly completes the triad of major phosphorus allotropes recognized in chemistry.

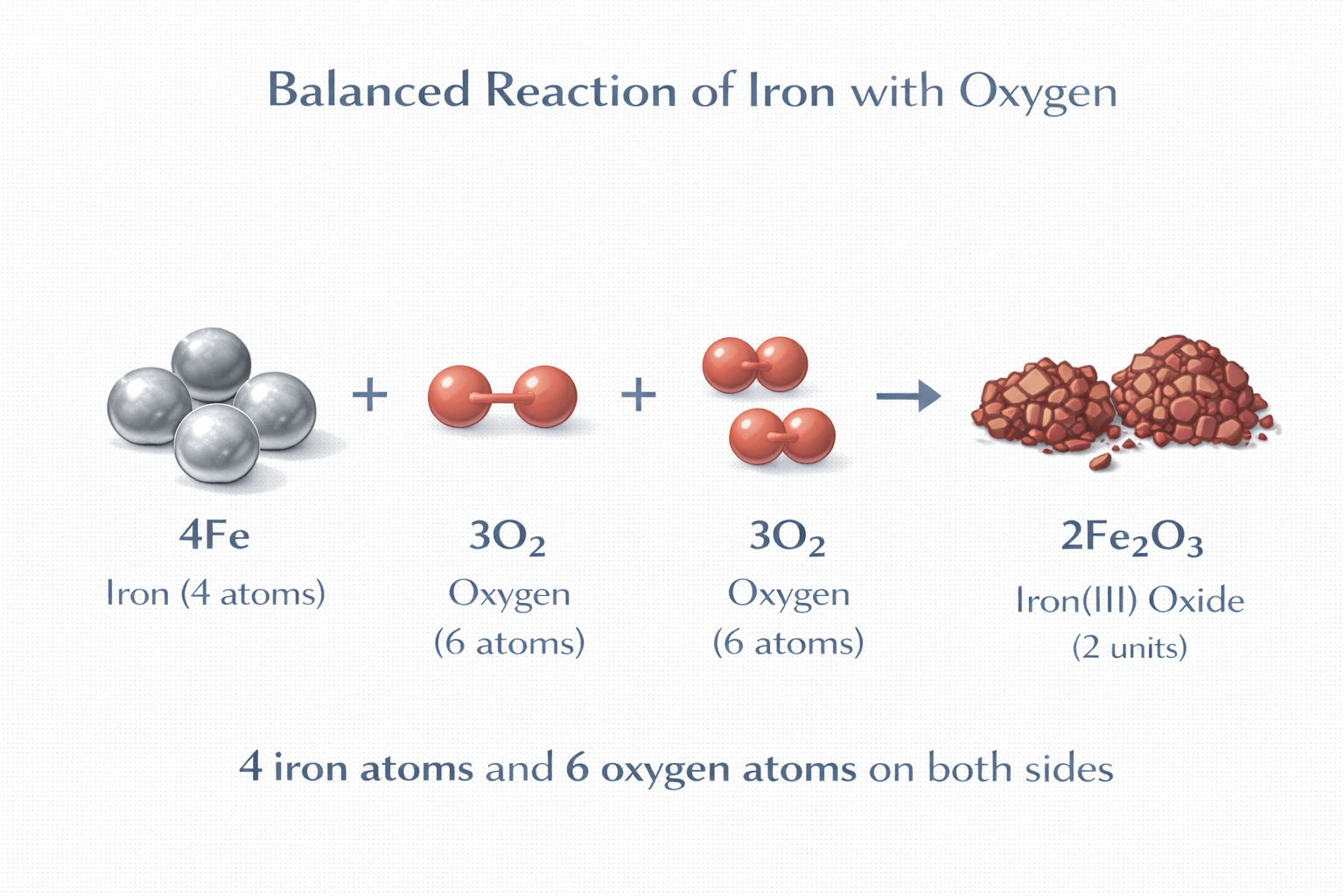
When balanced, the reaction Fe + O? ? Fe?O? will be
A.
2Fe + 2O? ? 3Fe?O?
B. 4Fe + 6O? ? 6Fe?O?
C. 2Fe + 3O? ? 2Fe?O?
D. 4Fe + 3O? ? 2Fe?O?
Rationale
The balanced equation for the reaction is 4Fe + 3O? ? 2Fe?O?. This stoichiometry ensures conservation of mass by equalizing the number of iron and oxygen atoms on both sides: 4 Fe atoms and 6 O atoms appear as reactants and products. The coefficients represent the smallest whole-number ratio that satisfies atomic balance for the formation of iron(III) oxide.
A) 2Fe + 2O? ? 3Fe?O?
Iron atoms are unbalanced: 2 Fe on the left versus 6 Fe (3 x 2) on the right. Oxygen atoms also mismatch: 4 O (2 x 2) on the left versus 9 O (3 x 3) on the right. This equation violates the law of conservation of mass and does not reflect any valid stoichiometric relationship for this reaction.
B) 4Fe + 6O? ? 6Fe?O?
This equation contains 4 Fe atoms on the left but 12 Fe atoms (6 x 2) on the right, clearly unbalanced. Oxygen shows 12 atoms (6 x 2) on the left versus 18 (6 x 3) on the right. Although the Fe:O ratio within Fe?O? is preserved, the overall atom counts are inconsistent. This appears to be a scaled-up version of an incorrect intermediate step, not a valid balanced equation.
C) 2Fe + 3O? ? 2Fe?O?
Oxygen is balanced (6 atoms on each side), but iron is not: 2 Fe on the left versus 4 Fe (2 x 2) on the right. This imbalance arises from insufficient iron atoms to form two formula units of Fe?O?, which require four iron atoms total. While oxygen stoichiometry is correct, the iron deficit renders the equation invalid.
D) 4Fe + 3O? ? 2Fe?O?
Iron atoms: 4 on the left, and 4 on the right (2 x 2 in two Fe?O? units). Oxygen atoms: 6 on the left (3 x 2 from O?), and 6 on the right (2 x 3 in two Fe?O? units). All atoms are conserved, and the coefficients are in the simplest whole-number ratio. This matches the known oxidation of iron to form rust (iron(III) oxide) under ambient conditions.
Conclusion
Balancing chemical equations requires equal numbers of each type of atom on both sides while using the smallest possible integer coefficients. Option D achieves this for the synthesis of Fe?O? from elemental iron and oxygen gas. The other choices fail due to atom imbalances or non-minimal, inconsistent scaling. Thus, D) 4Fe + 3O? ? 2Fe?O? is the only chemically and mathematically correct representation.
How many neutrons are in an atom of carbon-12?
A.
2
B. 4
C. 6
D. 24
Rationale
An atom of carbon-12 contains 6 neutrons. The mass number (12) represents the total number of protons and neutrons in the nucleus. Carbon's atomic number is 6, meaning it always has 6 protons. Subtracting the proton count from the mass number gives 12 ˆ’ 6 = 6 neutrons, which defines the carbon-12 isotope.
A) 2
This value does not correspond to any standard calculation for carbon-12. It may arise from confusion with helium (which has 2 protons and typically 2 neutrons), but carbon isotopes always contain 6 protons. No known carbon isotope has only 2 neutrons; even the lightest stable isotope, carbon-12, requires 6 neutrons for nuclear stability. This number lacks chemical or nuclear basis for carbon.
B) 4
Four neutrons would imply a mass number of 10 (6 protons + 4 neutrons), corresponding to carbon-10, a highly unstable, short-lived radioisotope not found in nature. Carbon-12, the most abundant and stable carbon isotope, must have 6 neutrons to achieve its observed mass and nuclear properties. This value misrepresents the composition of the standard reference isotope used in atomic mass definitions.
C) 6
Carbon-12 consists of 6 protons and 6 neutrons, giving it a mass number of 12. This balanced nucleon count contributes to its exceptional nuclear stability, making it the basis for the atomic mass unit (amu). The equal number of protons and neutrons is characteristic of light, stable nuclei. This neutron count distinguishes carbon-12 from other isotopes like carbon-13 (7 neutrons) and carbon-14 (8 neutrons).
D) 24
Twenty-four neutrons would produce a mass number of 30 (6 protons + 24 neutrons), far beyond any naturally occurring carbon isotope. Such a neutron-rich nucleus would be extremely unstable and decay almost instantly. This value likely results from doubling the mass number or misapplying multiplication instead of subtraction. It has no relevance to real carbon isotopes.
Conclusion
The neutron count in an isotope is determined by subtracting the atomic number (proton count) from the mass number. For carbon-12, this yields 12 ˆ’ 6 = 6 neutrons. This precise composition underpins carbon-12's role as the standard for atomic weights and its prevalence in organic chemistry. Only option C reflects this fundamental nuclear arithmetic accurately.

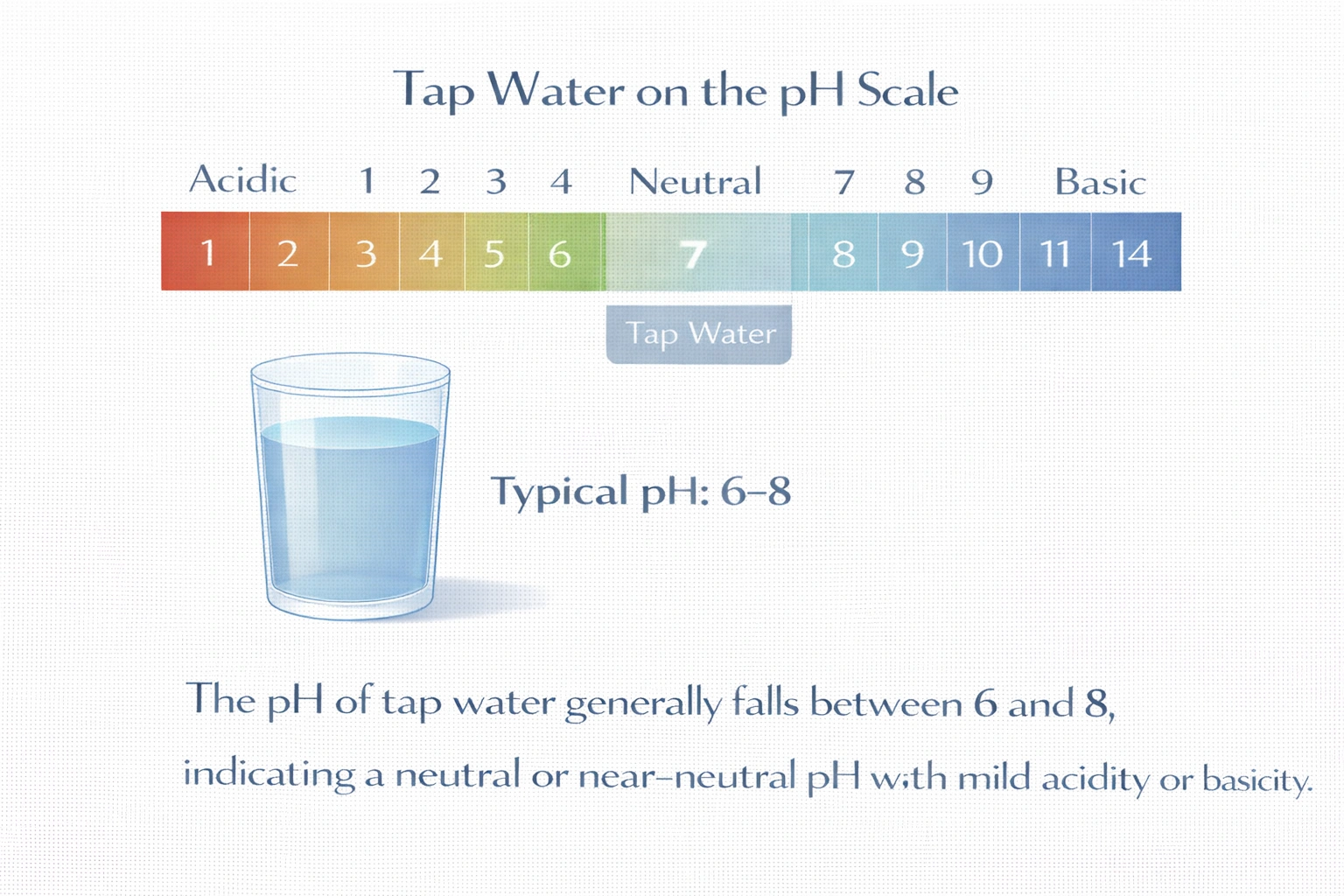
Where would you expect tap water to fall on the pH scale?
A.
Between 1 and 3
B. Between 4 and 6
C. Between 6 and 8
D. Between 8 and 10
Rationale
Tap water typically falls between pH 6 and 8 on the pH scale. This range encompasses neutral (pH 7) and reflects slight variations due to dissolved minerals, carbon dioxide, and municipal treatment processes. Regulatory standards, such as those from the U.S. Environmental Protection Agency (EPA), recommend a pH between 6.5 and 8.5 for drinking water to ensure safety, minimize pipe corrosion, and maintain palatability.
A) Between 1 and 3
This highly acidic range corresponds to substances like gastric acid (pH ~1.5-3.5) or undiluted vinegar. Water at this pH would be corrosive to plumbing, harmful to human tissue, and unsafe for consumption. Municipal water systems rigorously avoid such acidity through buffering and pH adjustment during treatment. Tap water never naturally or safely reaches this level of acidity.
B) Between 4 and 6
This moderately acidic range includes beverages like coffee (pH ~5) and soda (pH ~2.5-4). While rainwater can be slightly acidic (pH ~5.6) due to dissolved CO? forming carbonic acid, treated tap water is adjusted upward to prevent corrosion in distribution systems. Most municipal supplies buffer acidity using limestone or alkaline additives, keeping pH above 6.0 in nearly all regulated systems.
C) Between 6 and 8
This range includes neutral water (pH 7.0) and aligns with typical tap water measurements worldwide. Slight acidity (pH 6.5-7.0) may occur from dissolved CO?, while mild alkalinity (pH 7.0-8.0) often results from bicarbonate and carbonate ions in hard water. The EPA secondary standard sets acceptable pH between 6.5 and 8.5, and most utilities target 7.0-7.8 for optimal stability and taste. This interval represents safe, potable water under normal conditions.
D) Between 8 and 10
This basic range includes seawater (pH ~8.1), baking soda solutions (pH ~8.3), and ammonia (pH ~11). While some groundwater sources with high calcium carbonate content may reach pH 8.5, values above 8.5 are uncommon in municipal tap water. Excessively alkaline water can cause scaling in pipes and impart a bitter taste, so treatment plants avoid sustained pH above 8.5. This range overestimates typical tap water alkalinity.
Conclusion
Tap water is carefully managed to remain near neutral to ensure safety, infrastructure compatibility, and consumer acceptability. The range 6 to 8 accurately captures the vast majority of treated drinking water samples globally, accommodating minor natural and engineered variations while excluding extremes. Thus, C) Between 6 and 8 is the correct and scientifically supported answer.
What is the correct name of ZnSO4?
A.
Zinc sulfate
B. Zinc sulfide
C. Zinc sulfur
D. Zinc oxide
Rationale
Zinc sulfate is the correct name for ZnSO?. This ionic compound consists of the zinc cation (Zns²?) and the sulfate anion (SO?s²?), a polyatomic ion composed of one sulfur atom covalently bonded to four oxygen atoms in a tetrahedral arrangement. Standard IUPAC nomenclature for ionic compounds pairs the cation name with the anion name, yielding "zinc sulfate."
A) Zinc sulfate
Zinc commonly exhibits a +2 oxidation state, forming Zns²? ions in solution and solid compounds. The sulfate ion (SO?s²?) carries a -2 charge and is one of the most stable polyatomic anions. Charge balance requires a 1:1 ratio of Zns²? to SO?s²?, resulting in the formula ZnSO?. This compound appears as a white crystalline solid, often hydrated (e.g., ZnSO?·7H?O), and is used in agriculture, medicine, and electroplating.
B) Zinc sulfide
Zinc sulfide has the formula ZnS and contains no oxygen atoms. It forms when zinc ions combine with sulfide ions (Ss²?), not sulfate. ZnS is a white or yellowish solid used in luminescent paints and semiconductors. The presence of four oxygen atoms in ZnSO? definitively rules out this name, as sulfide implies only sulfur without oxygen.
C) Zinc sulfur
This is not valid chemical nomenclature. Elemental names like "sulfur" refer to neutral atoms (S), not ions or polyatomic groups. Ionic compounds must use the proper anion name, "sulfate" for SO?s²?, "sulfide" for Ss²?, not the elemental form. "Zinc sulfur" fails to specify charge, composition, or bonding, violating naming conventions.
D) Zinc oxide
Zinc oxide is ZnO, consisting of Zns²? and Os²? ions. It contains only one oxygen atom per zinc and lacks sulfur entirely. ZnO is a white powder used in sunscreens and rubber manufacturing. In contrast, ZnSO? includes both sulfur and four oxygen atoms as part of the sulfate group, making "zinc oxide" chemically inaccurate.
Conclusion
The formula ZnSO? unambiguously identifies a compound containing zinc and the sulfate ion. Correct naming follows the rule: [cation] + [polyatomic anion name]. Only zinc sulfate accurately reflects the composition and charge balance of ZnSO?. Misidentifying the anion as sulfide, elemental sulfur, or oxide ignores the presence of the SO?s²? group and violates fundamental naming principles.

What is the correct formula for potassium chloride?
A.
NaCl
B. KCl
C. KCl?
D. ClK
Rationale
The correct formula for potassium chloride is KCl. Potassium, a Group 1 alkali metal, forms a +1 cation (K?), while chlorine, a Group 17 halogen, forms a -1 anion (Cl?). The charges balance perfectly in a 1:1 ratio, and by convention, the cation is written before the anion in ionic compound formulas.
A) NaCl
This is the formula for sodium chloride, not potassium chloride. Although it follows the same 1:1 stoichiometry, it contains sodium (Na) instead of potassium (K). The two compounds are chemically similar but distinct; substituting one for the other in reactions or biological systems can lead to significant differences due to varying ionic sizes and reactivities.
B) KCl
Potassium readily loses its single valence electron to form K?, and chlorine gains one electron to form Cl?. Electrostatic attraction between these oppositely charged ions creates a stable ionic lattice. The formula KCl reflects both charge neutrality and standard chemical notation, cation first, anion second. This compound is widely used in fertilizers, medicine, and food processing.
C) KCl?
This formula implies either a Ks²? ion (which potassium does not form) or an imbalance of two Cl? ions per K?, resulting in a net -1 charge. Potassium exclusively exhibits a +1 oxidation state in compounds due to its [Ar]4s² electron configuration. No stable compound exists with the formula KCl? under normal conditions.
D) ClK
While this contains the correct elements, it violates the universal convention for writing ionic formulas: the cation is always written first. "ClK" is not accepted in chemical nomenclature, even though it might be understood informally. Proper notation ensures clarity and consistency across scientific communication.
Conclusion
Ionic compound formulas must reflect both charge balance and standard naming conventions. Potassium chloride consists of K? and Cl? in a 1:1 ratio, correctly written as KCl. All other options either use the wrong element, misrepresent stoichiometry, or violate notation rules. Thus, B) KCl is the only correct answer.

Aluminum (Al) has 13 protons in its nucleus. What is the number of electrons in an Al?? ion?
A.
16
B. 13
C. 10
D. 3
Rationale
An Al³? ion contains 10 electrons. Aluminum has 13 protons, and in its neutral atomic state, it also has 13 electrons. The 3+ charge indicates the loss of 3 electrons, leaving 13 ˆ’ 3 = 10 electrons. This electron count gives Al³? the same configuration as neon (1s²2s²2p?), a stable noble gas arrangement.
A) 16
This would imply a net charge of -3 (13 protons vs. 16 electrons), corresponding to Al³?, a species that does not exist under normal chemical conditions. Aluminum, being a metal, readily loses electrons to form cations, not anions. This option misinterprets the meaning of the superscript "3+".
B) 13
Thirteen electrons describe a neutral aluminum atom (Al°), not the Al³? ion. The question explicitly refers to the ion with a +3 charge, which requires electron removal. Retaining all 13 electrons would result in zero net charge, contradicting the given ion symbol.
C) 10
Correct. Removal of 3 valence electrons from neutral aluminum (electron configuration [Ne]3s²3p²) yields Al³? with the configuration 1s²2s²2p?identical to neon. This stable, closed-shell structure explains why aluminum commonly forms the +3 oxidation state in compounds like Al?O? or AlCl?.
D) 3
Three electrons would produce a +10 charge (13 ˆ’ 3 = +10), which is physically implausible for aluminum. No known chemical process removes 10 electrons from aluminum under standard conditions; even highly charged ions like Al³? are already energetically costly to form. This value likely results from confusing the charge magnitude with the electron count.
Conclusion
Ion charge reflects the difference between protons and electrons:
Charge = protons ˆ’ electrons ‡’ electrons = protons ˆ’ charge
For Al³?: electrons = 13 ˆ’ 3 = 10. This aligns with periodic trends and observed chemistry. Thus, C) 10 is the correct answer.

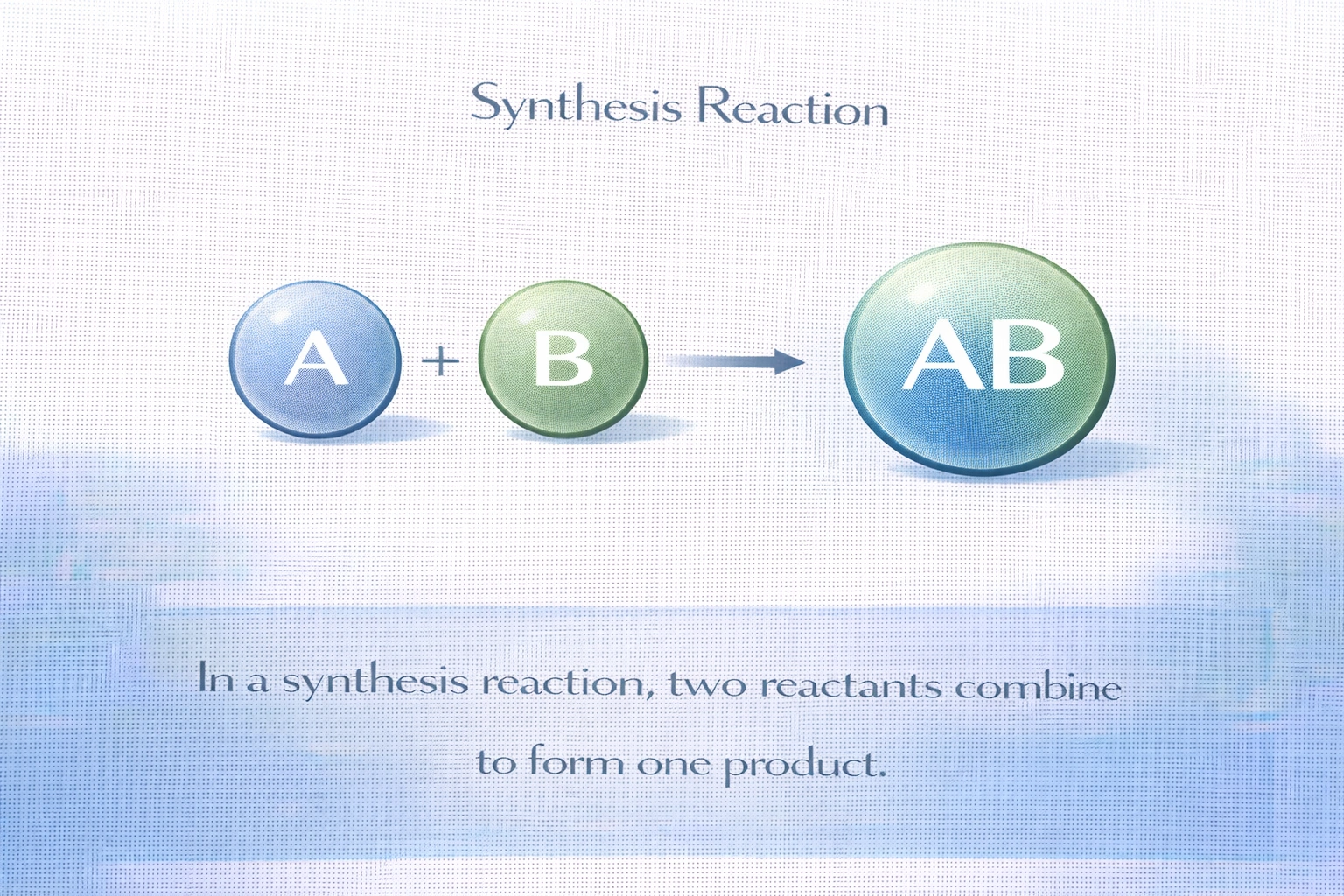
Name the reaction shown. A + B ? AB
A.
Single displacement
B. Double displacement
C. Synthesis
D. Acid-base
Rationale
The reaction A + B ? AB represents a synthesis (or combination) reaction. In this type of chemical process, two or more simpler substances, elements or compounds, combine to form a single, more complex product. The defining feature is multiple reactants yielding one product, which matches the given general equation exactly.
A) Single displacement
Single displacement reactions follow the pattern A + BC ? AC + B, where one element displaces another in a compound. This requires one element and one compound as reactants, and produces a new element and a new compound. The given reaction shows two reactants forming one product, with no evidence of replacement, so this is incorrect.
B) Double displacement
Double displacement (metathesis) reactions involve two ionic compounds exchanging ions: AB + CD ? AD + CB. These always produce two new compounds as products. Since the given reaction yields only one product, it cannot be a double displacement reaction.
C) Synthesis
Synthesis reactions are characterized by the general form A + B ? AB (or more reactants, e.g., A + B + C ? ABC). Examples include:
1. 2H? + O? ? 2H?O
2. CaO + H?O ? Ca(OH)?
These illustrate the core idea: simpler substances combine into a more complex one. The given equation fits this pattern precisely, making C) Synthesis correct.
D) Acid-base
Acid-base reactions specifically involve proton (H?) transfer, typically producing water and a salt (e.g., HCl + NaOH ? NaCl + H?O). They usually yield two products, not one. While some synthesis reactions might involve acids and bases, the general form A + B ? AB does not specify proton transfer, so this classification is unjustified.
Conclusion
The hallmark of a synthesis reaction is the formation of a single product from multiple reactants. The equation A + B ? AB is the textbook representation of this class. No other reaction type matches this stoichiometric pattern. Therefore, C) Synthesis is the correct answer.
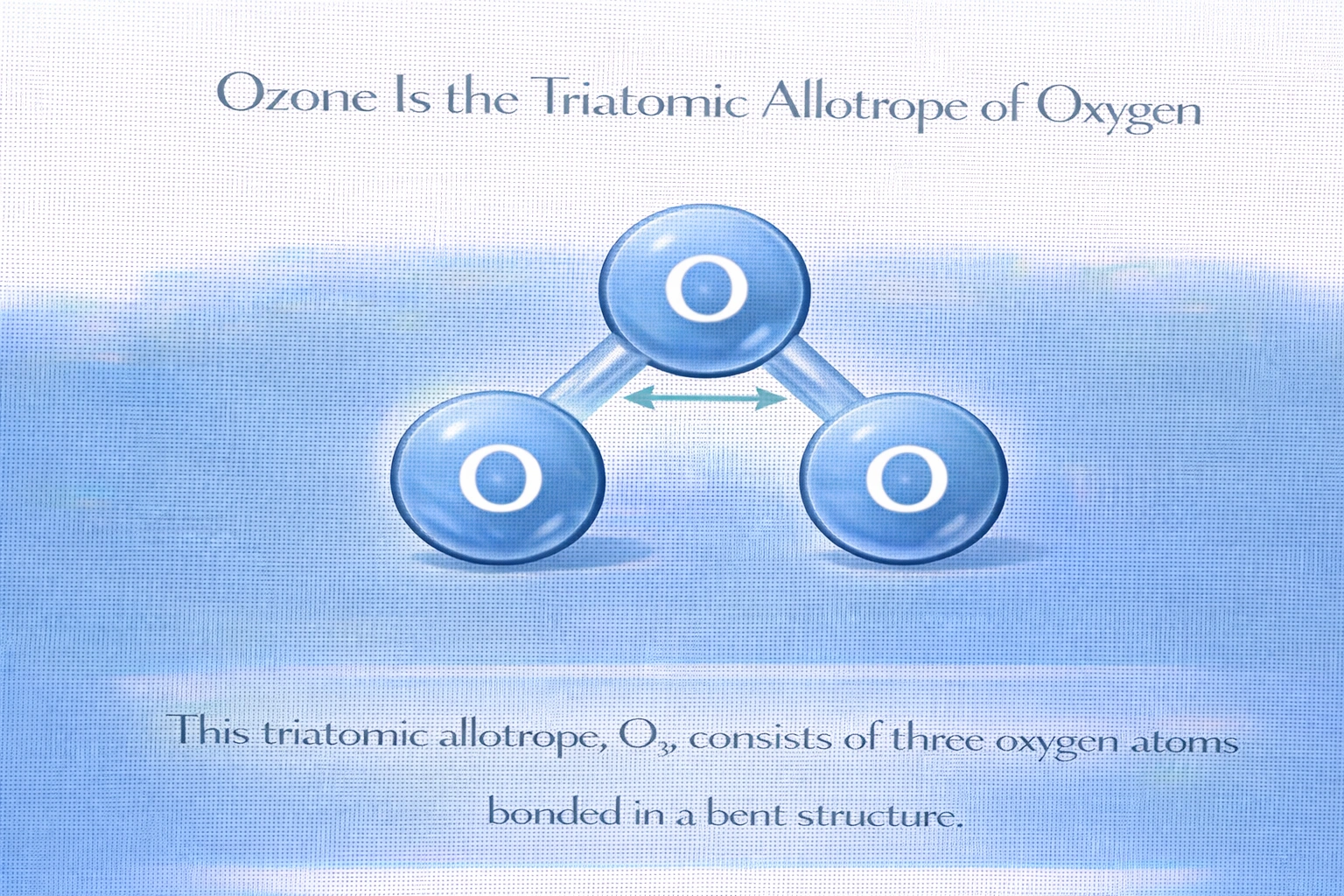
Which is a triatomic allotrope of oxygen?
A.
Ozone
B. Water
C. Acidic oxide
D. Carbon dioxide
Rationale
Ozone (O?) is the triatomic allotrope of oxygen. An allotrope is a different structural form of the same element in the same physical state. Ozone consists of three oxygen atoms bonded together in a bent, resonance-stabilized structure, distinct from the more common diatomic oxygen (O?). The prefix tri- denotes three atoms, and since ozone contains only oxygen atoms, it qualifies as a triatomic allotrope.
A) Ozone
Correct. O? is a pale blue gas with a sharp, pungent odor, formed naturally in the stratosphere by UV radiation. It is chemically distinct from O? and plays a critical role in absorbing harmful ultraviolet light. As a pure elemental form composed of three oxygen atoms, it is the only triatomic allotrope of oxygen.
B) Water
Water (H?O) is a compound, not an allotrope, because it contains two different elements: hydrogen and oxygen. While it is triatomic (three atoms total), allotropes must consist of only one element. Thus, water does not qualify.
C) Acidic oxide
This is a chemical classification, not a specific substance. Acidic oxides (e.g., CO?, SO?) are compounds that react with water to form acids. They are not pure oxygen and often contain other elements. This term does not refer to any allotrope of oxygen.
D) Carbon dioxide
CO? is a linear triatomic molecule, but it is a compound of carbon and oxygen, not a form of pure oxygen. Allotropes must be composed exclusively of a single element. Therefore, CO? is not an allotrope of oxygen.
Conclusion
Only ozone (O?) meets both criteria: it is made entirely of oxygen atoms and contains three atoms per molecule. Thus, A) Ozone is the correct answer.
Balance this equation: Zn + HCl ? ZnCl? + H?
A.
Zn + 2HCl ? ZnCl? + H?
B. Zn + HCl ? 2ZnCl? + H?
C. 2Zn + HCl ? 2ZnCl? + H?
D. Zn + 4HCl ? ZnCl? + H?
Rationale
The balanced chemical equation for the reaction between zinc and hydrochloric acid is Zn + 2HCl ? ZnCl? + H?. This single-displacement reaction involves zinc metal reducing hydrogen ions to form hydrogen gas while oxidizing itself to Zns²?, which combines with chloride ions to yield zinc chloride. The equation must obey the law of conservation of mass, ensuring equal numbers of each atom on both sides.
A) Zn + 2HCl ? ZnCl? + H?
This equation is fully balanced:
1. Zinc: 1 atom on left, 1 in ZnCl? on right
2. Hydrogen: 2 atoms from 2HCl on left, 2 in H? on right
3. Chlorine: 2 atoms from 2HCl on left, 2 in ZnCl? on right
All atoms are conserved, and coefficients are in the smallest whole-number ratio. This correctly represents the stoichiometry of the reaction and follows standard balancing rules.
B) Zn + HCl ? 2ZnCl? + H?
Zinc is unbalanced (1 vs. 4 atoms), chlorine (1 vs. 4), and hydrogen (1 vs. 2). This violates conservation of mass and misrepresents product formation, zinc cannot produce more zinc atoms than consumed. The coefficient "2" before ZnCl? is chemically impossible without additional zinc reactant.
C) 2Zn + HCl ? 2ZnCl? + H?
Zinc atoms: 2 on left, 4 on right (from 2ZnCl?), unbalanced. Chlorine: 1 on left, 4 on right. Hydrogen: 1 on left, 2 on right. This equation fails to conserve any element except possibly charge, but even then, it is invalid due to atom imbalance.
D) Zn + 4HCl ? ZnCl? + H?
Zinc is balanced (1 = 1), but hydrogen (4 vs. 2) and chlorine (4 vs. 2) are not. Excess HCl would remain unreacted, but the equation implies all 4HCl are consumed to form only one H? molecule, which contradicts reaction stoichiometry. The correct ratio requires exactly 2 HCl per Zn atom.
Conclusion
Balancing chemical equations relies on three key principles: (1) Conservation of mass, which mandates equal atom counts on both sides; (2) Strategic balancing, often starting with the most complex compound (here, ZnCl?); and (3) Coefficient adjustment only, never altering subscripts that define chemical identity. Applying these, Zn + 2HCl ? ZnCl? + H? is the only valid representation. Thus, A) Zn + 2HCl ? ZnCl? + H? is the correct answer.

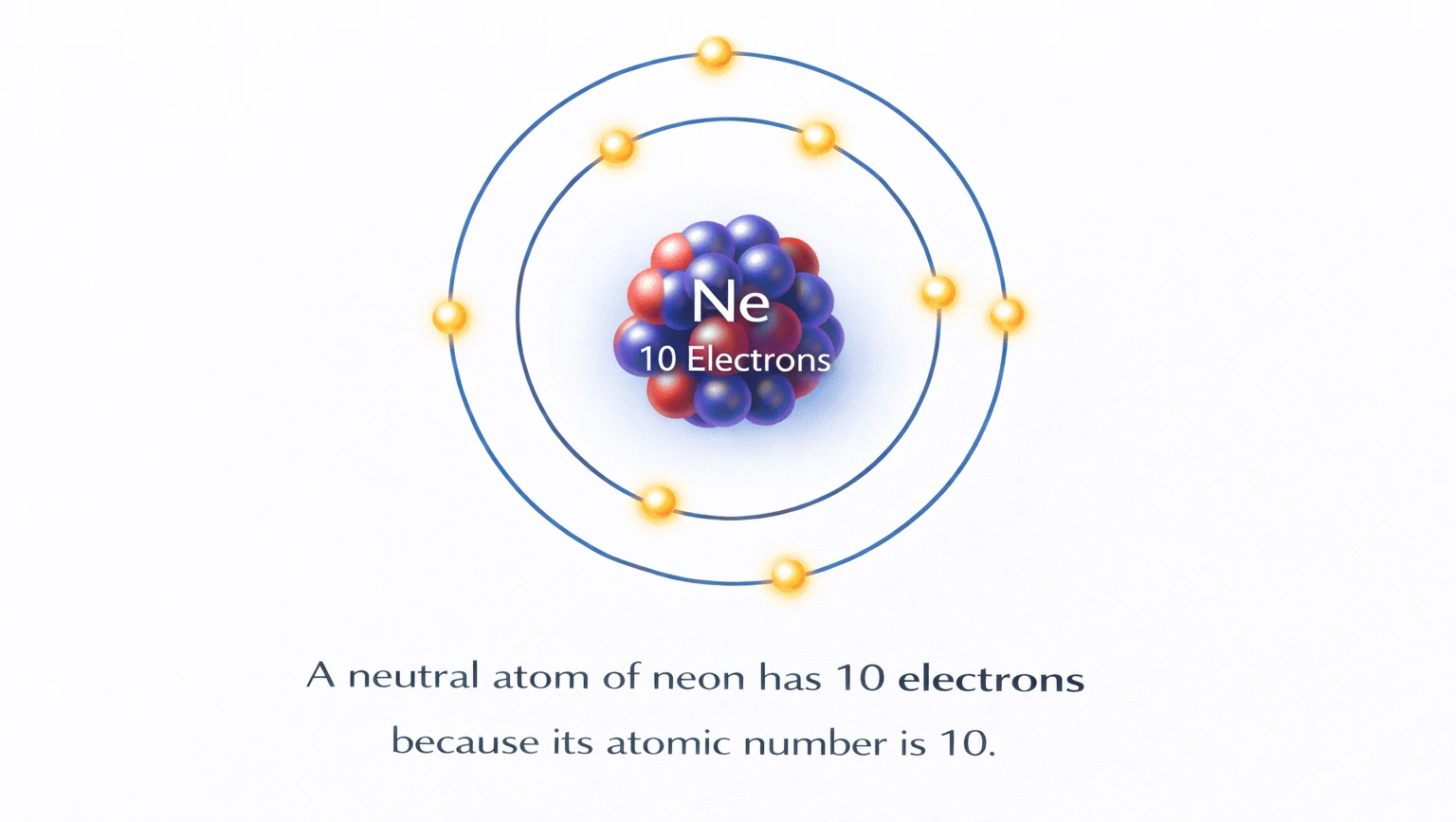
How many electrons are in a neutral atom of neon?
A.
9
B. 10
C. 11
D. 12
Rationale
A neutral atom of neon contains 10 electrons. Neon has an atomic number of 10, which defines the number of protons in its nucleus. In a neutral atom, the number of electrons equals the number of protons to ensure zero net charge. Thus, neon's electron configuration is 1ss²2ss²2p?, completing the second energy level and giving it the stable electron arrangement of a noble gas.
A) 9
This corresponds to fluorine (atomic number 9), which has one fewer proton and electron than neon. Fluorine's electron configuration ends in 2pµ, making it highly reactive as it seeks one more electron to achieve stability. Neon, by contrast, is inert due to its complete valence shell. This option misidentifies the element and is incorrect.
B) 10
Correct. Neon's atomic number is 10, so a neutral atom contains exactly 10 protons and 10 electrons. The filled 1s, 2s, and 2p orbitals result in a closed-shell configuration, explaining neon's lack of chemical reactivity and its classification as a noble gas. This matches both atomic theory and experimental observation.
C) 11
This corresponds to sodium (atomic number 11), which has one more electron than neon. Sodium's extra electron occupies the 3s orbital (configuration: [Ne]3s²), making it highly reactive as it readily loses that electron to form Na?. This value exceeds neon's electron count and is incorrect.
D) 12
This corresponds to magnesium (atomic number 12), with two more electrons than neon. Magnesium's configuration is [Ne]3ss², and it forms Mgs²? ions to achieve noble gas configuration. This is not relevant to neon and is incorrect.
Conclusion
The atomic number uniquely determines the number of protons and, in a neutral atom, the number of electrons. For neon (atomic number 10), this results in 10 electrons, giving it a stable, fully occupied electron shell. Therefore, B) 10 is the correct answer.
A drop of one of these substances rapidly turns litmus dye from blue to red. What substance is it?
A.
Milk
B. Sea water
C. Ammonia
D. Lemon juice
Rationale
Litmus is a pH indicator that turns red in acidic solutions and blue in basic solutions. A rapid color change from blue to red indicates the presence of a strong acid. Among the options, only lemon juice is sufficiently acidic to produce this immediate response due to its high concentration of citric acid.
A) Milk
Milk has a pH of approximately 6.5-6.7, making it only slightly acidic, close to neutral. It lacks the proton concentration needed for a rapid litmus response. Any color change would be slow and faint, not the sharp transition described. This is incorrect.
B) Sea water
Sea water is slightly basic (pH ‰ˆ 8.1) due to dissolved carbonates and bicarbonates. Basic solutions turn red litmus blue, not the reverse. Exposure to sea water would either leave blue litmus unchanged or deepen its blue hue. This produces the opposite effect and is incorrect.
C) Ammonia
Ammonia solution (e.g., household ammonia) is strongly basic (pH ‰ˆ 11-12). It readily accepts protons and increases OH? concentration, turning red litmus blue. It cannot turn blue litmus red; instead, it reinforces the blue color. This contradicts the observation and is incorrect.
D) Lemon juice
Lemon juice contains citric acid and has a pH of 2-3, classifying it as a strong organic acid. Its high [H?] rapidly protonates the litmus dye, causing an immediate structural change that shifts its absorption spectrum to appear red. This matches the described "rapid" color change from blue to red. This is correct.
Conclusion
The rapid blue-to-red litmus transition is diagnostic of a strong acid. Lemon juice, with its low pH and high acidity, is the only substance listed capable of producing this effect. Milk is too weakly acidic, while sea water and ammonia are basic and would not cause this color change. Therefore, D) Lemon juice is the correct answer.

What is the correct name of MgO?
A.
Manganese oxide
B. Magnesium oxide
C. Magnesium oxate
D. Magnesium hydroxide
Rationale
Magnesium oxide is the correct name for MgO. This ionic compound forms when magnesium (a Group 2 metal) loses two electrons to become Mgs²?, and oxygen (a nonmetal) gains two electrons to become Os²?. The resulting electrostatic attraction creates a neutral compound with a 1:1 ratio of ions, named using standard ionic nomenclature: the metal cation followed by the anion with the suffix "-ide."
A) Manganese oxide
Manganese is represented by the symbol Mn, not Mg. Mg is the chemical symbol for magnesium, a different element in Group 2. Manganese oxide would have formulas like MnO or MnO?, depending on oxidation state. This option confuses elemental symbols and is incorrect.
B) Magnesium oxide
Correct. The compound MgO consists of Mgs²? and Os²? ions. According to IUPAC naming rules for binary ionic compounds, the cation (magnesium) retains its elemental name, and the anion (oxygen) is modified to "oxide." Thus, magnesium oxide is the proper name. This compound is a white solid used in refractory materials and antacids.
C) Magnesium oxate
"Oxate" is not a recognized polyatomic ion or anion in chemical nomenclature. The correct name for the Os²? ion is oxide. Terms like "oxalate" (C?O?s²?) exist, but "oxate" has no chemical basis. This appears to be a fabricated term and is incorrect.
D) Magnesium hydroxide
Magnesium hydroxide has the formula Mg(OH)?, containing hydroxide ions (OH?), not oxide ions. It is a different compound altogether, commonly known as milk of magnesia, used as an antacid and laxative. MgO lacks hydrogen and hydroxide groups, so this name does not apply.
Conclusion
The name of an ionic compound is derived from its constituent ions: magnesium for Mgs²? and oxide for Os²?. No other naming convention applies to MgO. Therefore, B) Magnesium oxide is the correct answer.

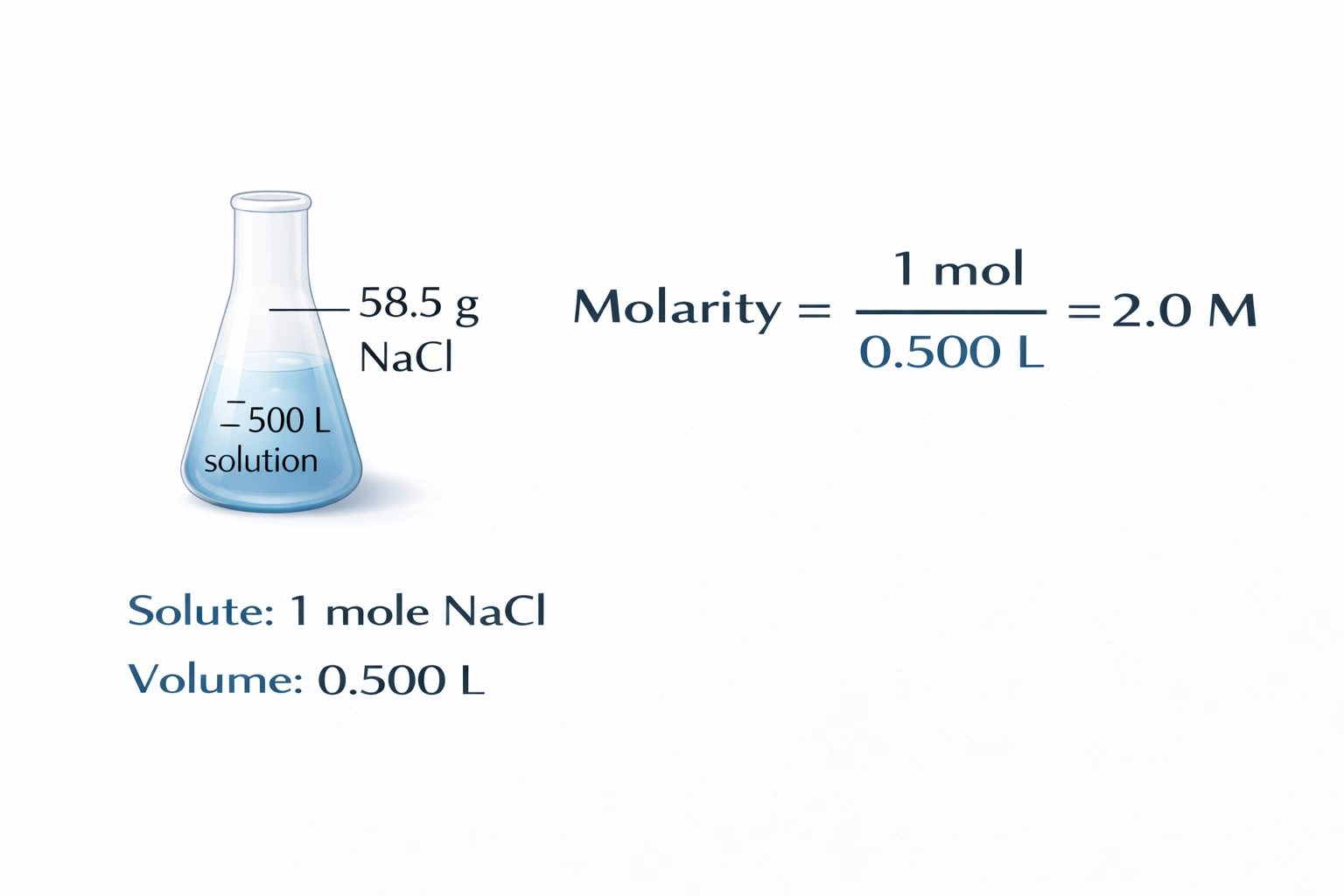
If 58.5 g of NaCl (1 mole of NaCl) are dissolved in enough water to make 0.500 L of solution, what is the molarity of the solution?
A.
1.0 M
B. 2.0 M
C. 11.7 M
D. The answer cannot be determined from the information given.
Rationale
The molarity of a solution is defined as the number of moles of solute per liter of solution. Here, 58.5 g of NaCl corresponds to exactly 1 mole (since the molar mass of NaCl is 58.5 g/mol), and it is dissolved in enough water to make 0.500 L of solution. Thus, the molarity is calculated as:
Molarity = 1 mol / 0.500 L = 2.0 M
A) 1.0 M
This would be correct only if the final volume were 1.0 L. However, the solution is prepared in half that volume (0.500 L), so the same amount of solute is concentrated in a smaller volume, doubling the molarity. This option ignores the given volume and is incorrect.
B) 2.0 M
Correct. The calculation directly follows from the definition of molarity: 1 mole of NaCl in 0.500 L yields a concentration of 2.0 mol/L, or 2.0 M. The problem provides both the exact moles (via mass and known molar mass) and the exact final volume, allowing precise determination.
C) 11.7 M
This value may arise from mistakenly dividing the mass (58.5 g) by 5 or confusing molarity with other concentration units. It does not correspond to any valid application of M = n/V. The correct computation gives 2.0 M, not 11.7 M. This is incorrect.
D) The answer cannot be determined from the information given
All necessary data are provided: the mass of NaCl (58.5 g), its molar mass (implied by stating it equals 1 mole), and the final solution volume (0.500 L). No additional information is needed. This option falsely claims insufficiency and is incorrect.
Conclusion
Molarity depends on moles of solute and total solution volume. With 1 mole of NaCl in 0.500 L, the concentration is unambiguously 2.0 M. Therefore, B) 2.0 M is the correct answer.
HESI A2 Exams
Biology Quizzes
3 Practice Tests
Biology Quizzes
3 Practice Tests
Anatomy Quizzes
3 Practice Tests
Anatomy Quizzes
3 Practice Tests
Reading Quizzes
4 Practice Tests
Reading Quizzes
4 Practice Tests
Grammar Quizzes
3 Practice Tests
Grammar Quizzes
3 Practice Tests
Vocabulary Quizzes
3 Practice Tests
Vocabulary Quizzes
3 Practice Tests
Physics Quizzes
3 Practice Tests
Physics Quizzes
3 Practice Tests
HESI Quizzes
3 Practice Tests
HESI Quizzes
3 Practice Tests