If bacteria are placed in a strong solution of salt water, they will shrink as watermoves out of the bacteria. What is this process called?
-
A
Dehydration synthesis
-
B
Hydrolysis
-
C
Osmosis
-
D
Isotonic transport
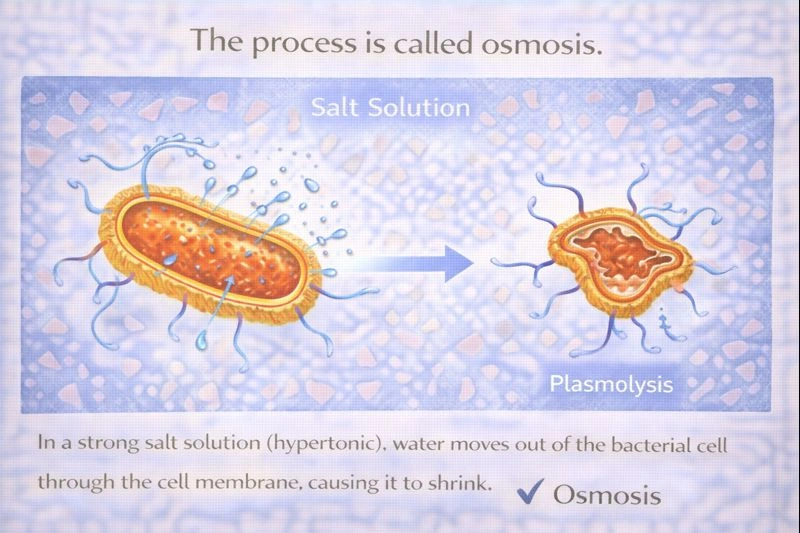
The process is called osmosis.
When bacteria are placed in a strong salt solution, the environment outside the cell has a much higher solute concentration than the inside of the cell. This condition is described as hypertonic. Cell membranes are selectively permeable, meaning water can move across them, but many solutes cannot.
Because of the difference in solute concentration, water moves out of the bacterial cell through the cell membrane. This movement occurs from an area of higher water concentration (inside the cell) to an area of lower water concentration (outside the cell). As water leaves the cell, the cytoplasm shrinks away from the cell wall, a condition known as plasmolysis. This shrinkage can slow bacterial metabolism or even kill the cell if severe.
A) Dehydration synthesis
This is a chemical reaction in which small molecules join together to form larger molecules while releasing water as a byproduct. For example, amino acids join to form proteins through dehydration synthesis. It does not involve water moving across cell membranes and does not explain why bacteria shrink in salt water.
B) Hydrolysis
Hydrolysis is the opposite of dehydration synthesis. It uses water to break large molecules into smaller ones, such as breaking starch into glucose molecules during digestion. It is a chemical process, not a membrane transport process, and does not describe water leaving cells.
C) Osmosis
Osmosis is the passive movement of water across a semipermeable membrane from an area of low solute concentration to an area of high solute concentration. In salty water, the outside solution is hypertonic, so water leaves the bacterial cell. This causes the cell to shrink, which matches the situation described in the question.
D) Isotonic transport
Isotonic conditions mean the solute concentration inside and outside the cell is equal. In this case, water would move in and out at equal rates, and the cell would remain the same size. Since the bacteria are shrinking, the solution is not isotonic.
Conclusion
Bacteria shrink in strong salt solutions because water moves out of the cell across the membrane due to a concentration gradient. This movement of water is called osmosis. Therefore, the correct answer is C) Osmosis.
Topic Flashcards
Click to FlipWhat is the term for the net movement of water across a semipermeable membrane from a region of low solute concentration to high solute concentration?
Osmosis. It is a passive process driven by the concentration gradient of water.
When a cell loses water and shrinks in a hypertonic solution, what is the specific term for this event in plant or bacterial cells?
Plasmolysis. The cytoplasm pulls away from the cell wall as water exits the cell.
True or False: Osmosis is a type of diffusion specifically for solvent molecules (like water), not solute particles.
True. Osmosis is the diffusion of water across a membrane. Diffusion typically refers to the movement of solutes.
What critical property must the membrane have for osmosis to occur?
It must be selectively permeable (semipermeable), allowing water to pass but restricting the movement of certain solutes.
If bacteria in a strong salt solution shrink, what would likely happen if they were placed in pure water instead?
They would swell as water moves into the cell (a hypertonic to hypotonic shift). In extreme cases, animal cells could burst, but bacterial cells with rigid walls are more resistant.