A subatomic particle is___ in relation to an atom.
-
A
faster
-
B
smaller
-
C
bigger
-
D
denser
While subatomic particles such as electrons move faster than the nuclei of atoms due to their small size and mass.
the question asks about the relative size of subatomic particles compared to the atom. Subatomic particles are smaller than the entire atom, and this is the focus of the question. The speed of subatomic particles is not directly relevant to their size in relation to an atom.
- A) Faster:
- While subatomic particles such as electrons move faster than the nuclei of atoms due to their small size and mass, the question asks about the relative size of subatomic particles compared to the atom. Subatomic particles are smaller than the entire atom, and this is the focus of the question. The speed of subatomic particles is not directly relevant to their size in relation to an atom.
- B) Smaller:
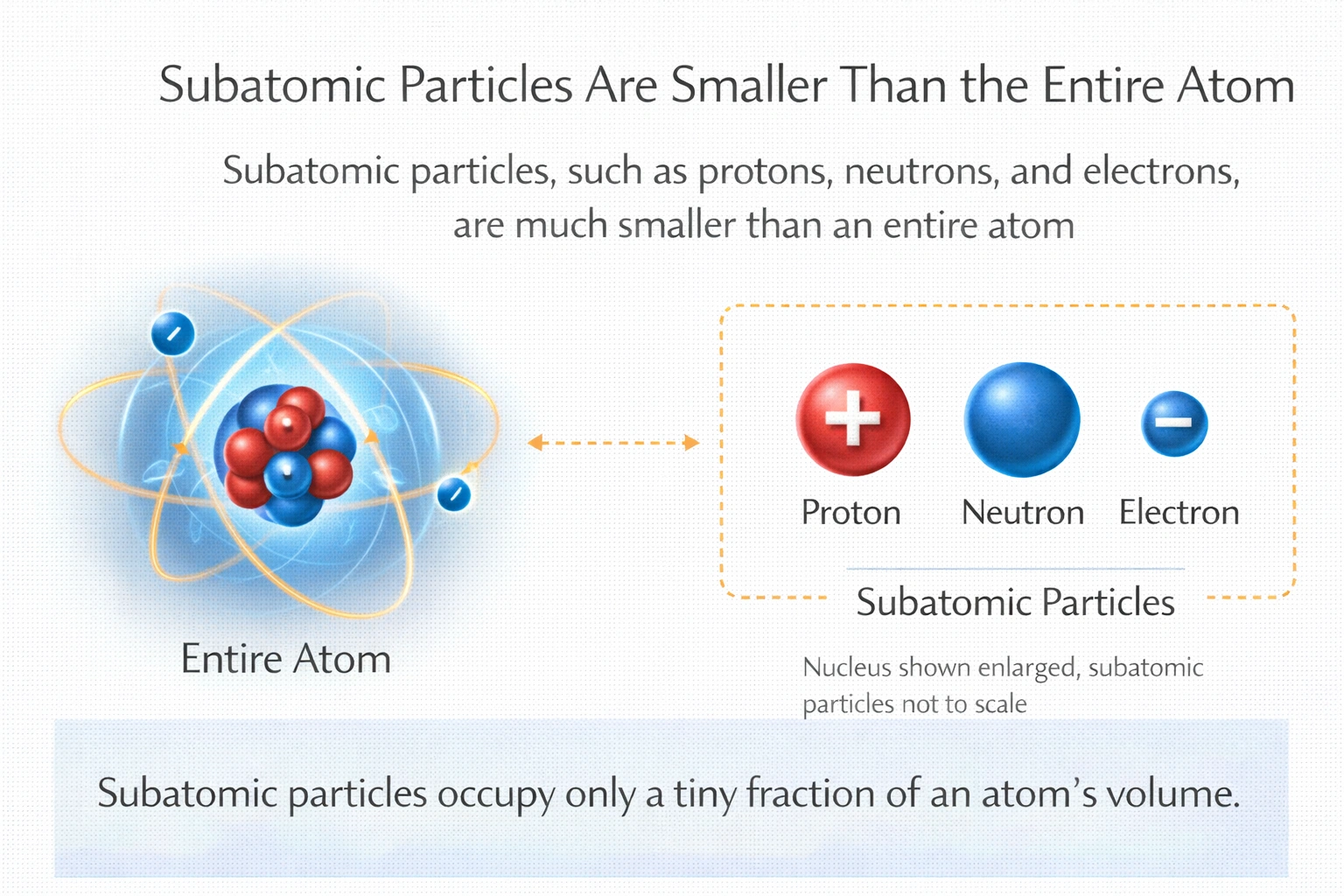
- Subatomic particles (such as protons, neutrons, and electrons) are much smaller than the atom itself. For example, electrons have a mass that is 1/1836 of a proton’s mass, and they occupy the space outside the nucleus of an atom. The size of an atom is determined by the space the electrons occupy in orbitals around the nucleus, whereas subatomic particles like protons and neutrons are found in the nucleus, which is only a tiny fraction of the total size of the atom.
- C) Bigger:
- This is incorrect. Subatomic particles are smaller than atoms, not larger. Atoms consist of a nucleus (composed of protons and neutrons) and surrounding electrons. The nucleus is extremely small compared to the overall size of the atom, making subatomic particles much smaller than the whole atom.
- D) Denser:
- While subatomic particles like protons and neutrons are dense in terms of mass, the size of subatomic particles is much smaller than that of an atom. The density of subatomic particles refers to how tightly packed their mass is, not their relative size. Therefore, this answer is not accurate in the context of the question.
Conclusion: Subatomic particles are smaller in relation to an atom, making B) Smaller the correct answer.
Topic Flashcards
Click to FlipWhat are the three primary subatomic particles that make up an atom?
Protons, neutrons, and electrons.
An atom is mostly empty space. What tiny, dense region contains most of its mass?
The nucleus.
True or False: The electron cloud (where electrons are found) defines the overall size of an atom.
True.
The nucleus of an atom is composed of which two types of subatomic particles?
Protons and neutrons.
What term describes a particle that is a constituent of an atom and is therefore smaller than the atom itself?
A subatomic particle.