Extract:
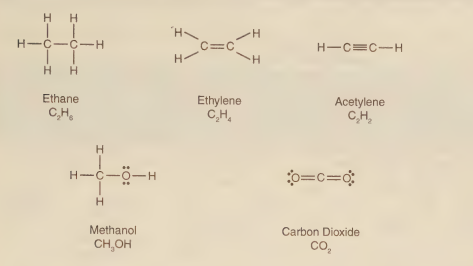
According to the Lewis structure below, what is the formula for methanol?
-
A
CHO
-
B
CH₄O
-
C
CH₃OH
-
D
C₃H₇O
The molecular formula for methanol is CH₄O.
A Lewis structure shows the arrangement of atoms and valence electrons in a molecule. For methanol, the structure consists of one carbon (C) atom, one oxygen (O) atom, and four hydrogen (H) atoms. The carbon atom is bonded to three hydrogens and to the oxygen atom. The oxygen atom is bonded to the carbon and to one additional hydrogen atom. Counting all atoms yields one carbon, four hydrogens, and one oxygen, which corresponds to the molecular formula CH₄O. The condensed structural formula, which shows the connectivity, is CH₃OH.
A) CHO
This formula has a 1:1:1 atom ratio and is the empirical formula for many compounds, including formic acid (HCOOH) and formaldehyde (CH₂O). However, it lacks the necessary hydrogen atoms to represent methanol, which requires four hydrogens.
B) CH₄O
This is the correct molecular formula for methanol. It unambiguously indicates one carbon, four hydrogen, and one oxygen atom, which matches the atomic count from the Lewis structure.
C) CH₃OH
This is the condensed structural formula for methanol. It conveys the same atomic count as CH₄O (the "H₃" represents three hydrogens on carbon, and the "OH" represents one oxygen bonded to a hydrogen, totaling C, H₃, O, H = CH₄O). It provides more structural information by showing the hydroxyl (-OH) group. Given the duplicate correct answers in the list, the molecular formula CH₄O is the most direct answer from an atomic count perspective.
D) C₃H₇O
This formula could represent several isomers of propanol (e.g., 1-propanol, 2-propanol) or methyl ethyl ether. It contains three carbon atoms, whereas methanol has only one carbon atom.
Conclusion:
From the Lewis structure of methanol, a direct count of atoms reveals one carbon, four hydrogens, and one oxygen. The simplest and most direct chemical formula representing this composition is CH₄O, the molecular formula.
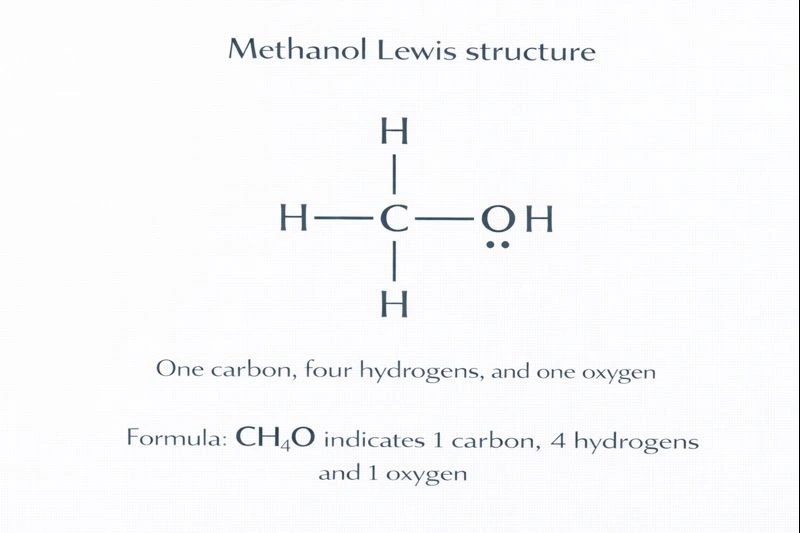
Topic Flashcards
Click to FlipWhat is the molecular formula (simplest whole-number ratio of atoms) for methanol?
CH₄O
What is the condensed structural formula for methanol, which shows the connectivity of atoms?
CH₃OH
How many hydrogen atoms are bonded directly to the carbon atom in a molecule of methanol?
Three hydrogen atoms (as shown in the CH₃ group).
What is the name of the functional group (-OH) present in methanol?
The hydroxyl group (which classifies methanol as an alcohol).
In methanol, which atom (Carbon or Oxygen) is the central atom connected to the most other atoms?
The Carbon atom (It is bonded to three H atoms and one O atom).