What is polar molecule?
-
A
A molecule that contains oxygen
-
B
A molecule that contains oxygen
-
C
A molecule that is attracted to water
-
D
A molecule that is attracted to water
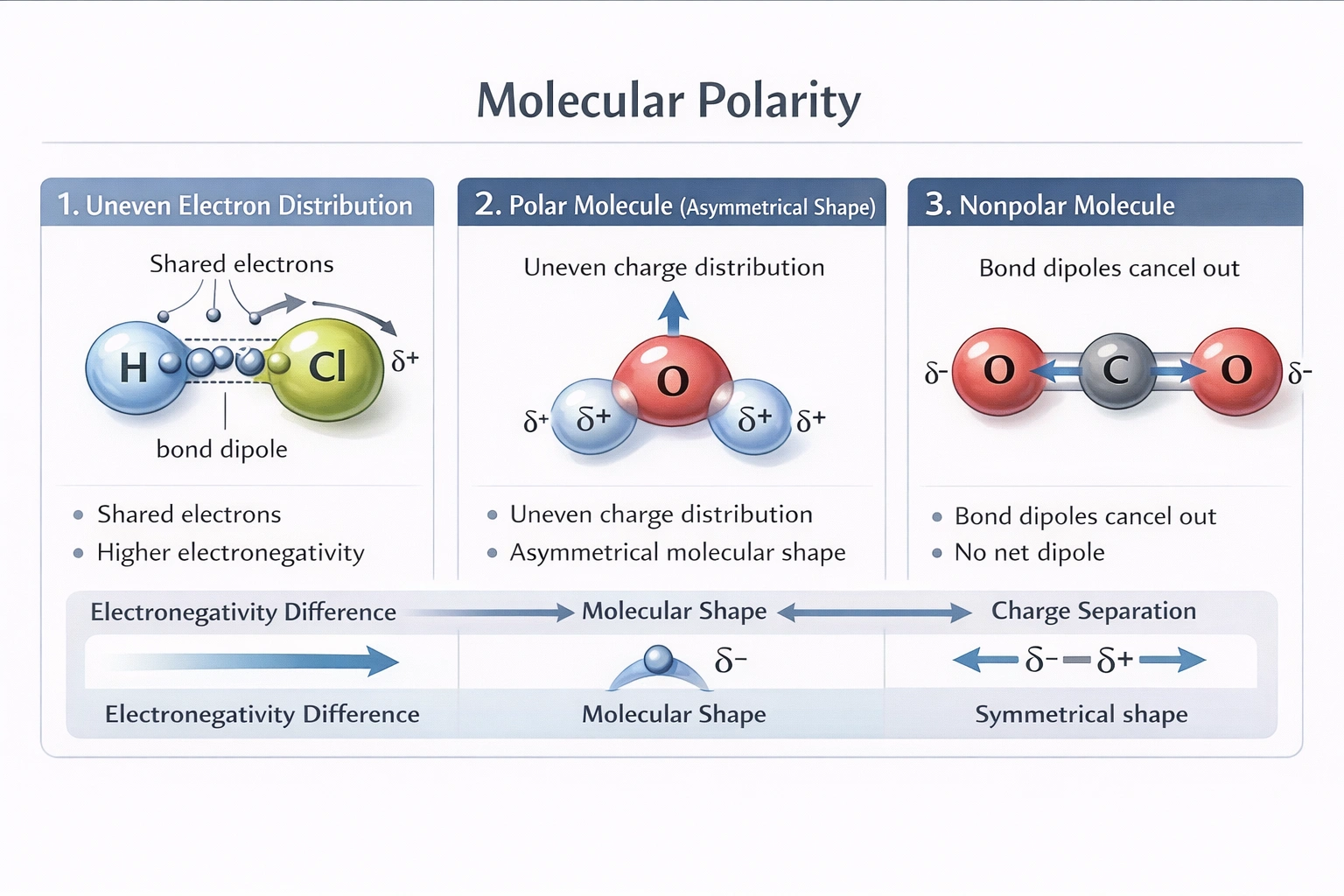
A polar molecule is a molecule that has slight charges on each end.
Molecular polarity arises from an uneven distribution of electron density within a molecule. This occurs when atoms with different electronegativities are bonded together, and the molecular shape prevents the cancellation of the resulting individual bond dipoles.
A) A molecule that contains oxygen
The presence of an oxygen atom does not guarantee polarity. While oxygen is electronegative and often participates in polar bonds (as in water, H₂O), the overall polarity of the molecule depends on its shape. For example, carbon dioxide (CO₂) contains oxygen but is linear; the polar C=O bonds are symmetrically opposed, canceling each other out and making CO₂ a nonpolar molecule. Conversely, many polar molecules (like HCl) do not contain oxygen at all.
B) A molecule that is repulsed by water
Molecules that are repelled by water are typically nonpolar and hydrophobic ("water-fearing"). They lack a charge separation that can interact favorably with water's polar molecules. Polar molecules, due to their charge separation, are generally attracted to water, not repulsed by it.
C) A molecule that is attracted to water
This describes a hydrophilic ("water-loving") molecule. While it is true that most polar molecules are hydrophilic because they can form dipole-dipole interactions or hydrogen bonds with water, this is a behavioral consequence of polarity, not its definition. Furthermore, some ions (which are charged but not molecules) and even some nonpolar substances can be attracted to water through other mechanisms (e.g., hydration of ions, dispersion forces). Attraction to water is a property, not the defining structural feature.
D) A molecule that has slight charges on each end
This is the core definition. In a polar covalent bond, the more electronegative atom pulls the shared electrons closer, acquiring a partial negative charge (δ-), while the other atom gets a partial positive charge (δ+). If the molecule's geometry is asymmetrical (like bent, trigonal pyramidal, or any shape where bond dipoles do not sum to zero), then one region of the molecule will have a net partial positive charge and another a net partial negative charge. This separation of charge creates a permanent dipole moment, making the molecule polar. Water is the classic example: the oxygen end is δ- and the hydrogen ends are δ+.
 Conclusion:
Conclusion:
Polarity is an intrinsic electrical property of a molecule defined by the presence of distinct, separated partial positive and partial negative poles due to uneven electron distribution. It is not defined by the presence of a specific element, by repulsion from water, or even solely by attraction to water, but by the existence of these molecular "ends" with opposite partial charges.
Topic Flashcards
Click to FlipWhat two conditions must be met for a molecule to be polar?
1) It must have polar covalent bonds (atoms with different electronegativities). 2) It must have an asymmetrical shape so the bond dipoles don't cancel out.
What is the term for the separation of partial positive (δ+) and partial negative (δ-) charges within a single molecule?
A dipole moment (or it is a dipole).
Water (H₂O) is polar, but carbon dioxide (CO₂) is not. Why is CO₂ nonpolar despite having polar C=O bonds?
Because its linear shape causes the two bond dipoles to point in opposite directions and cancel each other out.
True or False: A molecule must contain oxygen to be polar. Provide a counterexample.
False. Hydrogen chloride (HCl) is a polar molecule that does not contain oxygen.
What property of a molecule, resulting from its polarity, explains why it dissolves readily in water?
It is hydrophilic ("water-loving") because it can form favorable interactions (like hydrogen bonds) with water molecules.