Extract:
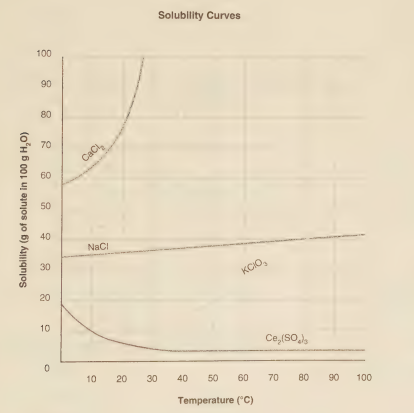
If 30 grams of KClO₃ is added to 100 grams of water at 0°C, some of the solute remains undissolved. What is the minimum temperature that must be attained to dissolve all the solute?
-
A
30°C
-
B
70°C
-
C
80°C
-
D
100°C
The minimum temperature required to dissolve all 30 grams of KClO₃ in 100 g of water is 70°C.
Solubility curves graphically represent the maximum amount of solute that can dissolve in a given amount of solvent (usually 100 g of water) at different temperatures. If 30 g of KClO₃ is added to 100 g of water at 0°C and some remains undissolved, it means the solubility at 0°C is less than 30 g/100g. To dissolve all 30 g, the temperature must be raised to the point where the solubility of KClO₃ is at least 30 g/100g. This point is found where a horizontal line at 30 g on the y-axis intersects the KClO₃ solubility curve; the corresponding temperature on the x-axis is the minimum required.
A) 30°C
At 30°C, the solubility of KClO₃ is typically less than 30 g/100g water (often around 10-15 g based on common solubility curves). Therefore, at 30°C, not all 30 grams would dissolve; some solid would still remain.
B) 70°C
On a standard solubility curve for KClO₃, the solubility line crosses the 30 g/100g water mark at approximately 70°C. This means at 70°C, the solution is exactly saturated with 30 g of KClO₃. At any temperature at or above 70°C, the solubility is greater than or equal to 30 g/100g, allowing all the solute to dissolve.
C) 80°C
At 80°C, the solubility of KClO₃ is greater than 30 g/100g water, so all the solute would certainly dissolve. However, 80°C is not the minimum temperature required; it is higher than necessary. The question asks for the lowest temperature at which no solid remains.
D) 100°C
At 100°C, the solubility is even higher, and dissolution would occur rapidly. Again, this temperature is sufficient but not the minimum required to achieve the goal.
Conclusion:
To find the minimum temperature to dissolve a specific mass of solute, one must reference the solubility curve for that substance. The minimum temperature corresponds precisely to the point where the solubility equals the mass of solute present per 100 g of solvent. For 30 g of KClO₃ in 100 g of water, this intersection occurs at 70°C.
Topic Flashcards
Click to FlipWhat is the term for the maximum amount of solute that can dissolve in 100 g of solvent at a given temperature?
Solubility.
On a solubility curve, how do you determine the minimum temperature needed to dissolve a specific mass of solute in 100 g of water?
Find the point where a horizontal line at that mass intersects the solubility curve; the corresponding temperature is the minimum required.
If adding 30 g of KClO₃ to 100 g of water at 0°C leaves solid undissolved, what is the state of the solution at 0°C?
It is a saturated solution (with excess solid).
For most solid solutes like KClO₃, how does solubility change as temperature increases?
Solubility generally increases with increasing temperature.
If the solubility of KClO₃ at 70°C is exactly 30 g/100 g water, what will happen if the same solution is cooled to 50°C?
Some KClO₃ will crystallize out of solution because the solubility at 50°C is lower.