Which of the following atoms will have an overall positive charge?
-
A
9 protons, 10 neutrons, 10 electrons
-
B
10 protons, 10 neutrons, 10 electrons
-
C
11 protons, 12 neutrons, 10 electrons
-
D
14 protons, 14 neutrons, 18 electrons
The atom with 11 protons, 12 neutrons, and 10 electrons will have an overall positive charge.
The overall charge is calculated by comparing the number of protons (positive charges) to the number of electrons (negative charges). The formula is: Net Charge = (Number of Protons) - (Number of Electrons). A positive result indicates a cation.
A) 9 protons, 10 neutrons, 10 electrons
Protons: 9. Electrons: 10. Net charge = 9 - 10 = -1. This results in a net negative charge (anion).
B) 10 protons, 10 neutrons, 10 electrons
Protons: 10. Electrons: 10. Net charge = 10 - 10 = 0. This atom is electrically neutral.
C) 11 protons, 12 neutrons, 10 electrons
Protons: 11. Electrons: 10. Net charge = 11 - 10 = +1. This results in a net positive charge (cation).
D) 14 protons, 14 neutrons, 18 electrons
Protons: 14. Electrons: 18. Net charge = 14 - 18 = -4. This results in a net negative charge.
Conclusion:
An atom carries a positive charge when it contains more protons than electrons. Performing the simple charge calculation for each option shows that only the atom with 11 protons and 10 electrons has a proton excess, yielding a net positive charge.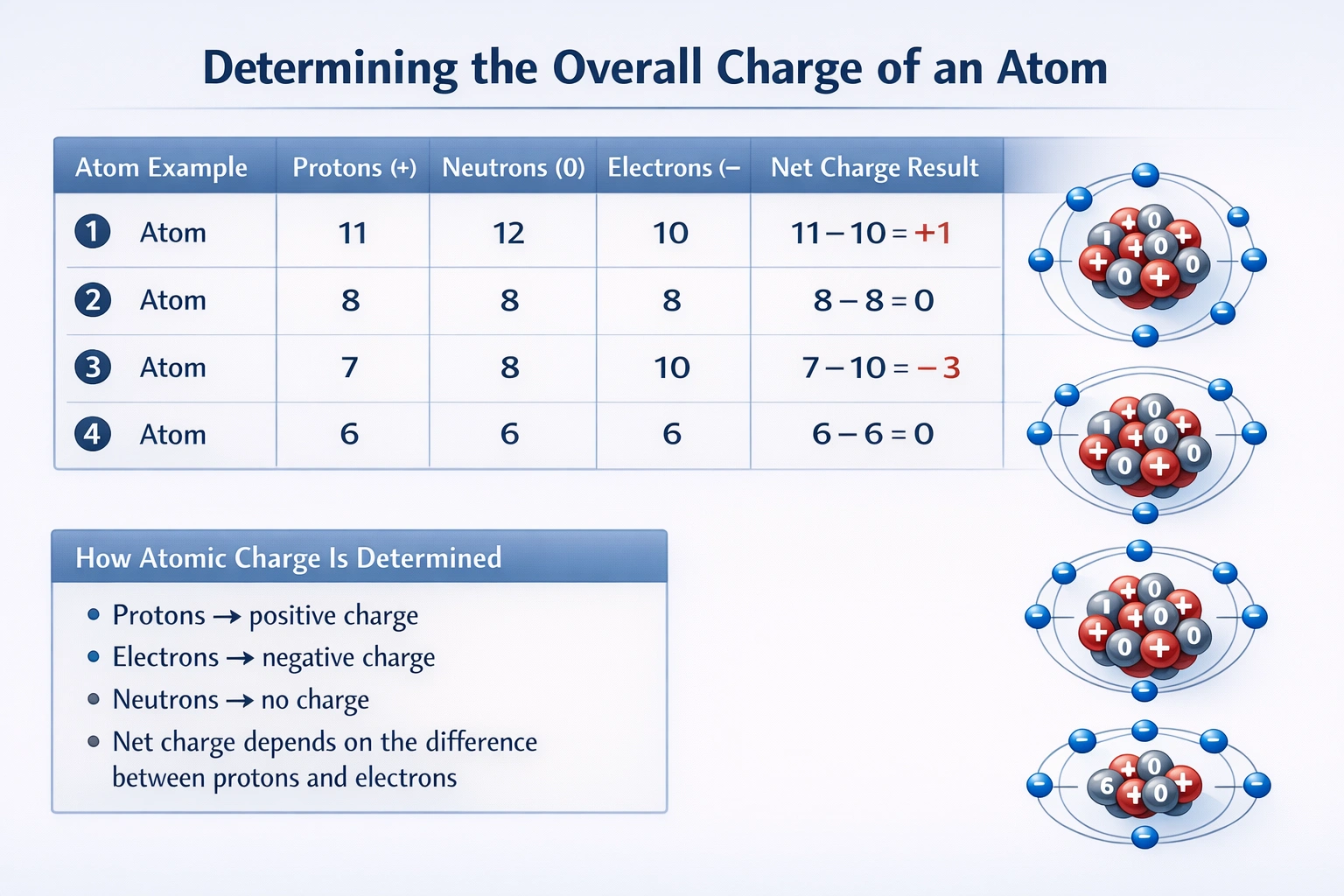
Topic Flashcards
Click to FlipWhat simple calculation determines the net charge of an atom or ion?
Net Charge = (Number of Protons) – (Number of Electrons).
An atom has 13 protons and 10 electrons. What is its net charge?
+3 (Charge = 13 - 10 = +3).
What is the name for an atom that has lost one or more electrons and therefore has a positive charge?
A cation.
In a neutral atom, the number of electrons must equal the number of ______.
Protons.
If an ion has a charge of +2 and has 12 protons, how many electrons does it have?
10 electrons (Charge = Protons – Electrons; +2 = 12 – e; e = 10).