Aluminum (Al) has 13 protons in its nucleus. What is the number of electrons in an Al³⁺ ion?
-
A
16
-
B
13
-
C
10
-
D
3
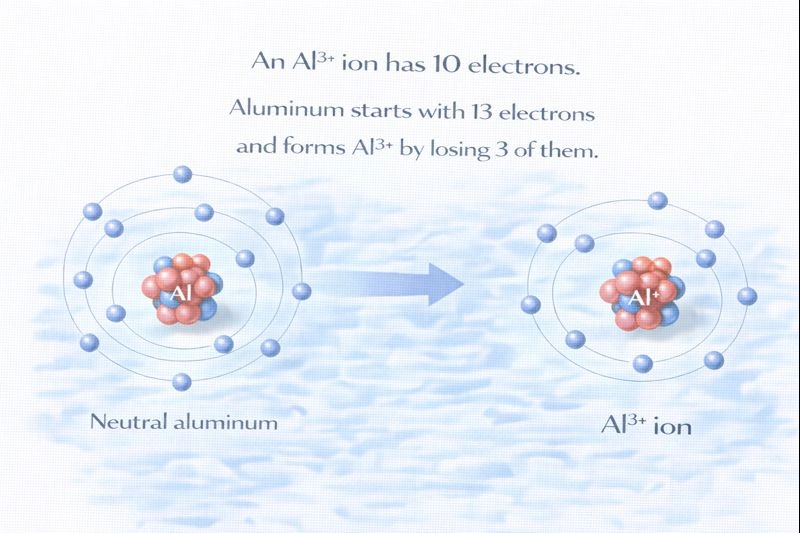
The Al³⁺ ion contains 10 electrons.
Aluminum has an atomic number of 13, which means a neutral aluminum atom contains 13 protons and 13 electrons. When aluminum forms an Al³⁺ ion, it becomes positively charged by losing electrons. A 3+ charge indicates that three electrons have been removed from the neutral atom. As a result, the total number of electrons decreases while the number of protons in the nucleus remains unchanged.
A. 16
Having 16 electrons would mean aluminum gained three additional electrons, producing a 3− charge (Al³⁻), not a 3+ charge. The question specifically refers to a positively charged ion.
B. 13
Thirteen electrons correspond to a neutral aluminum atom. Once aluminum forms an Al³⁺ ion, it no longer has the same number of electrons as protons.
C. 10
An Al³⁺ ion has lost three electrons. Starting from 13 electrons in the neutral atom, subtracting three electrons results in 10 electrons remaining.
D. 3
The value 3 represents the magnitude of the charge, not the number of electrons. An aluminum ion with only three electrons would be extremely unstable and would not correspond to Al³⁺.
Conclusion
The charge on an ion indicates how many electrons have been gained or lost relative to the neutral atom. Aluminum starts with 13 electrons, and forming a 3+ ion means losing three of them. This leaves the Al³⁺ ion with 10 electrons, while the nucleus still contains 13 protons.
Topic Flashcards
Click to FlipThe term for the process where an electron moves from a higher to a lower energy level.
Emission (or relaxation).
The form in which excess energy is released during electron relaxation.
A photon of electromagnetic radiation (e.g., light).
The specific, quantized packet of light energy emitted during an electron transition.
A photon.
The phenomenon where unique sets of colored lines are produced by elements, caused by electron energy level transitions.
An emission spectrum (or line spectrum).
The process that provides the energy needed for an electron to jump to a higher energy level.
Absorption of energy (e.g., from heat or light).