How do zinc and sulfur react to form a compound?
-
A
Zinc and sulfur will share electrons.
-
B
Sulfur will transfer electrons to zinc.
-
C
Zinc will transfer electrons to sulfur.
-
D
Zinc and sulfur do not react with each other.
Zinc will transfer electrons to sulfur.
This question addresses the type of chemical bond formed between a metal (zinc) and a nonmetal (sulfur). The difference in electronegativity between these elements is large enough to favor the complete transfer of electrons, leading to ionic bond formation.
A) Zinc and sulfur will share electrons.
Electron sharing characterizes covalent bonding, which is typical between two nonmetals. While some metal-nonmetal bonds can have covalent character, the primary and most stable interaction between a reactive metal like zinc and a nonmetal like sulfur is ionic. Sharing implies a relatively small electronegativity difference, which is not the case here.
B) Sulfur will transfer electrons to zinc.
This reverses the natural tendency. Sulfur is a nonmetal with a relatively high electronegativity (tendency to attract electrons). It wants to gain electrons to achieve a stable, full valence shell (octet). Zinc is a metal with a lower electronegativity; it tends to lose electrons to achieve a stable electron configuration. It is energetically unfavorable for sulfur, which "wants" electrons, to give them away to zinc.
C) Zinc will transfer electrons to sulfur.
This is correct. Zinc, a metal in Group 12, has two valence electrons in its 4s orbital. It readily loses these two electrons to form a Zn²⁺ cation, achieving the stable electron configuration of the previous noble gas (argon). Sulfur, a nonmetal in Group 16, has six valence electrons. It gains the two electrons lost by zinc to fill its valence shell to eight electrons, forming an S²⁻ anion. The resulting oppositely charged ions (Zn²⁺ and S²⁻) are then held together by strong electrostatic forces, forming the ionic compound zinc sulfide (ZnS). This direct electron transfer is the hallmark of ionic bond formation.
D) Zinc and sulfur do not react with each other.
This is false. Zinc and sulfur react vigorously, often with the emission of heat and light, in a highly exothermic combination reaction to form zinc sulfide. This reaction is commonly demonstrated in chemistry education and is a practical method for producing the compound.
Conclusion:
The reaction between a metal and a nonmetal typically involves the metal atom donating electrons to the nonmetal atom. This electron transfer creates positively and negatively charged ions that attract each other, forming an ionic compound. For zinc and sulfur, zinc loses two electrons to sulfur.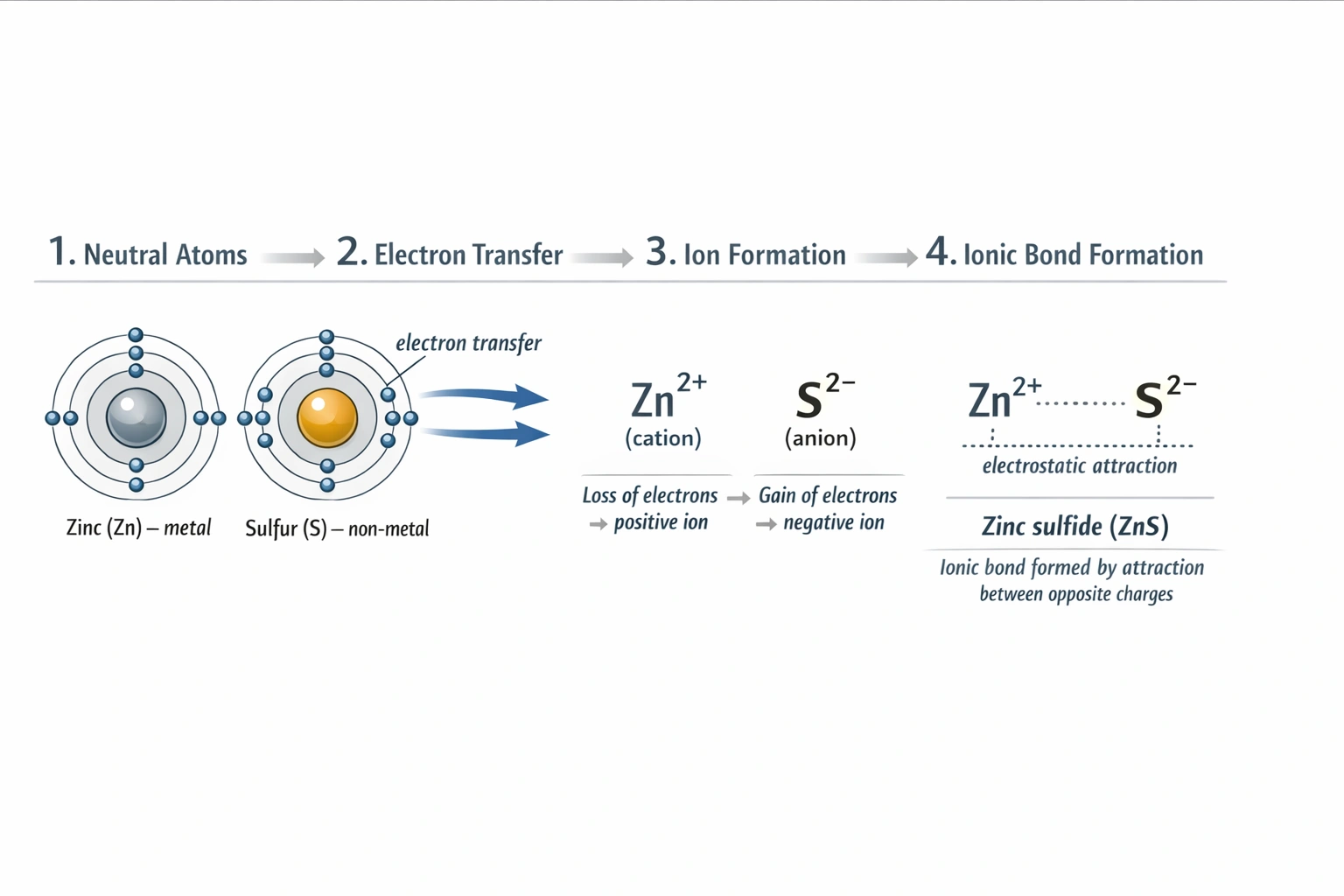
Topic Flashcards
Click to FlipWhat type of chemical bond is primarily formed when zinc (a metal) reacts with sulfur (a nonmetal)?
An ionic bond. This occurs through the transfer of electrons from the metal to the nonmetal, forming oppositely charged ions.
Why is the description "zinc and sulfur will share electrons" incorrect for this reaction?
Electron sharing defines covalent bonds, typical between two nonmetals. The large electronegativity difference between metal (Zn) and nonmetal (S) drives a complete electron transfer, not sharing.
What specific ions are formed when zinc reacts with sulfur, and what are their charges?
Zinc loses two electrons to form the Zn²⁺ cation. Sulfur gains those two electrons to form the S²⁻ anion.
What is the name and chemical formula of the compound produced in this reaction?
The compound is zinc sulfide, with the chemical formula ZnS.
What is the driving force for zinc to transfer electrons to sulfur?
Both elements achieve stable electron configurations. Zinc loses 2e⁻ to resemble argon (stable octet), and sulfur gains 2e⁻ to complete its valence shell (octet), fulfilling the octet rule.