The first and second energy levels of a neutral atom are full, and the third energy level contains three electrons. To which group does this element belong?
-
A
Group 3
-
B
Group 8
-
C
Group 10
-
D
Group 13
This element belongs to Group 13.
The group number for main group elements (Groups 1, 2, and 13-18 in the modern IUPAC numbering) generally indicates the number of valence electrons. Valence electrons are those in the outermost principal energy level (highest n value) and are primarily responsible for an element's chemical properties.
A) Group 3
In the modern periodic table, Group 3 contains the scandium family (Sc, Y, La, Ac), which are transition metals. Their electron configurations involve filling inner d-orbitals, and their group number does not simply correspond to a count of s and p valence electrons. An atom with the described configuration is not a transition metal in Group 3.
B) Group 8
Group 8 contains iron, ruthenium, osmium, and hassium transition metals. The noble gases, which have full valence shells, are in Group 18. An atom with only three electrons in its third shell does not have a full outer shell, so it cannot be a noble gas. The number 8 does not correspond to the valence electron count of the described atom.
C) Group 10
Group 10 contains nickel, palladium, platinum, and darmstadtium more transition metals. Their chemistry is dominated by d-electrons. The described electron configuration points to a main group element, not a Group 10 transition metal.
D) Group 13
The description gives a specific electron configuration. "First and second energy levels are full" means: First level (n=1): holds max 2 electrons (1s²). Second level (n=2): holds max 8 electrons (2s²2p⁶). "Third energy level contains three electrons" means the configuration continues as 3s²3p¹. Therefore, the full electron configuration is 1s² 2s² 2p⁶ 3s² 3p¹. The valence electrons are those in the highest principal energy level (n=3): the 3s² and 3p¹ electrons, totaling 3 valence electrons. Elements with three valence electrons reside in Group 13. Examples include boron (B), aluminum (Al), gallium (Ga), etc. Aluminum, for instance, has the exact configuration [Ne] 3s² 3p¹.
Conclusion: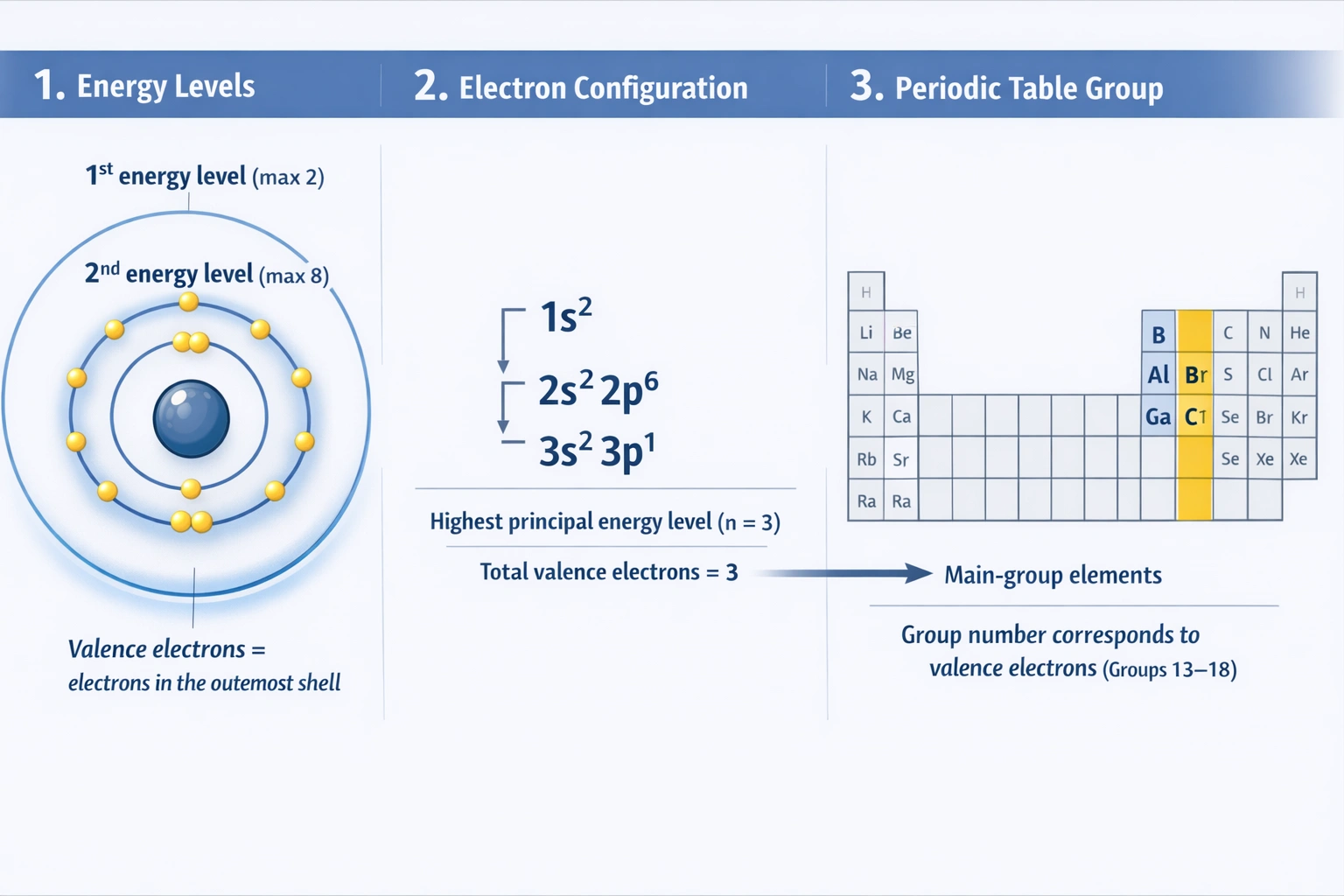
By determining the number of valence electrons from the described electron occupancy, we find the atom has three valence electrons. In the modern periodic table, elements with three valence electrons in their outermost s and p orbitals are classified in Group 13.
Topic Flashcards
Click to FlipThe first two energy levels of an atom are full, and the third level has three electrons. How many total valence electrons does this atom have?
Three (3) valence electrons. The valence electrons are in the outermost (third) level. The configuration ends as 3s²3p¹.
For main group elements (Groups 1, 2, 13-18), what does the group number tell you about the atom?
The group number indicates the number of valence electrons (e.g., Group 13 has 3, Group 17 has 7).
An element has the electron configuration 1s² 2s² 2p⁶ 3s² 3p¹. To which group in the periodic table does it belong?
Group 13. The highest level is n=3, which contains 3 electrons (3s²3p¹), corresponding to three valence electrons.
What common, lightweight metal has the electron configuration described (full n=1 & n=2, three electrons in n=3)?
Aluminum (Al). Its noble gas configuration is [Ne] 3s² 3p¹.
Why is the element with this configuration NOT in Group 3?
Group 3 contains transition metals (like Scandium) that are filling inner *d* orbitals. This element is a main-group metal filling *p* orbitals.