What molecule plays a direct role in chemiosmosis?
-
A
glucose
-
B
NADH
-
C
O₂
-
D
pyruvate
NADH plays a direct role in chemiosmosis.
Chemiosmosis is the mechanism by which cells generate the majority of their ATP. It involves the use of an electrochemical proton gradient across a membrane to drive ATP synthesis via the enzyme ATP synthase. In cellular respiration, this proton gradient is established by the electron transport chain (ETC) embedded in the inner mitochondrial membrane. The direct role in chemiosmosis is played by the molecules that donate high-energy electrons to the beginning of this chain. NADH (Nicotinamide Adenine Dinucleotide, reduced form) is a primary electron carrier. It is produced during glycolysis, the Krebs cycle, and other pathways. NADH donates its electrons to Complex I (NADH dehydrogenase) of the ETC. This donation initiates the flow of electrons through the chain, which provides the energy to pump protons (H⁺) from the mitochondrial matrix into the intermembrane space, creating the proton motive force essential for chemiosmosis.
A) glucose
Glucose is the initial fuel molecule for cellular respiration. However, it is several metabolic steps removed from the electron transport chain. Glucose must first be broken down through glycolysis and the Krebs cycle to produce the reduced electron carriers (NADH and FADH₂). Glucose itself does not directly interact with the ETC or contribute to proton pumping, making it an indirect, not direct, participant in chemiosmosis.
B) NADH
NADH is a direct electron donor to the ETC. When NADH transfers its electrons to Complex I, it is oxidized back to NAD⁺. The energy released from this exergonic electron transfer is used to pump protons, directly contributing to the establishment of the proton gradient. The chemiosmotic process is fundamentally coupled to the flow of electrons from donors like NADH.
C) O₂
Molecular oxygen (O₂) is the final electron acceptor in the ETC. At Complex IV, oxygen accepts electrons and combines with protons to form water. This role is absolutely critical because it removes low-energy electrons from the chain, allowing it to continue operating. However, O₂ does not itself create the proton gradient; it acts at the end of the process. Its role is terminal and accepting, not direct in the energy-coupling that builds the gradient for ATP synthesis.
D) pyruvate
Pyruvate is a key metabolic intermediate. In the presence of oxygen, it is transported into the mitochondrial matrix and converted to acetyl-CoA, which enters the Krebs cycle. The Krebs cycle generates NADH and FADH₂. Therefore, pyruvate is a crucial precursor to the electron carriers but, like glucose, does not directly interact with the electron transport chain or the proton pumps. Its role is in substrate provision, not in the chemiosmotic mechanism itself.
Conclusion
Chemiosmosis depends on a series of coupled reactions: electron transport drives proton pumping, and the resulting proton gradient drives ATP synthesis. The molecules that have a direct role are those that participate in the electron transport step. NADH (and FADH₂) are these direct participants, serving as the source of the electrons whose energy is harnessed to build the proton gradient. Substrates like glucose and pyruvate are upstream fuel sources, and oxygen is a downstream electron sink. NADH sits at the crucial interface between substrate metabolism and the proton-motive machinery, making it the molecule with a direct role in chemiosmosis.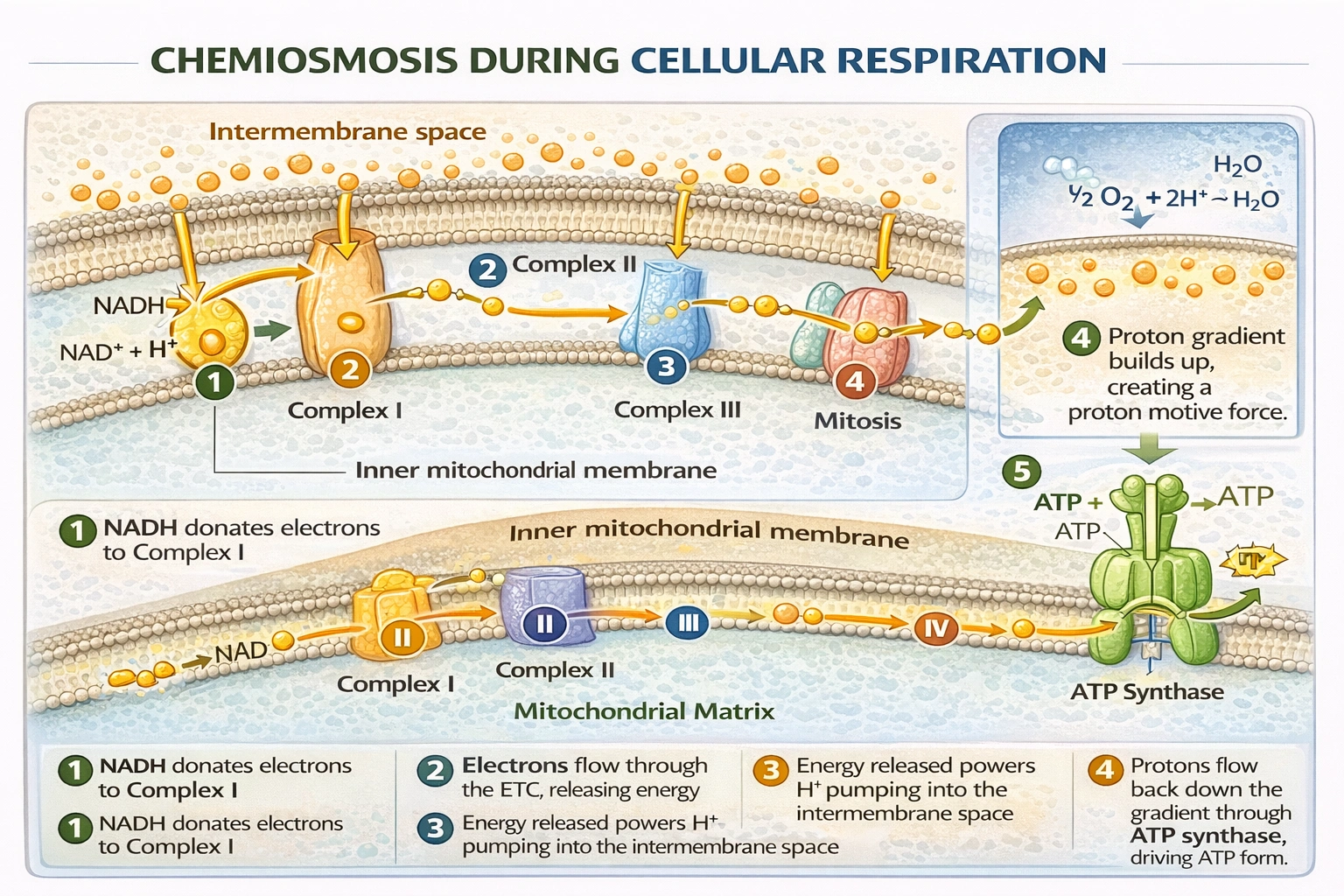
Topic Flashcards
Click to FlipWhich molecule plays a direct role in chemiosmosis?
NADH.
What does NADH donate during chemiosmosis?
High-energy electrons.
Where does NADH donate its electrons?
The electron transport chain.
What does the electron transport chain create that drives chemiosmosis?
A proton (H⁺) gradient.
What enzyme uses the proton gradient to synthesize ATP?
ATP synthase.