What would you expect to happen when electrons in an excited state fall to lower energy levels?
-
A
Energy is absorbed.
-
B
Energy is released.
-
C
Energy is both released and absorbed.
-
D
Energy remains constant.
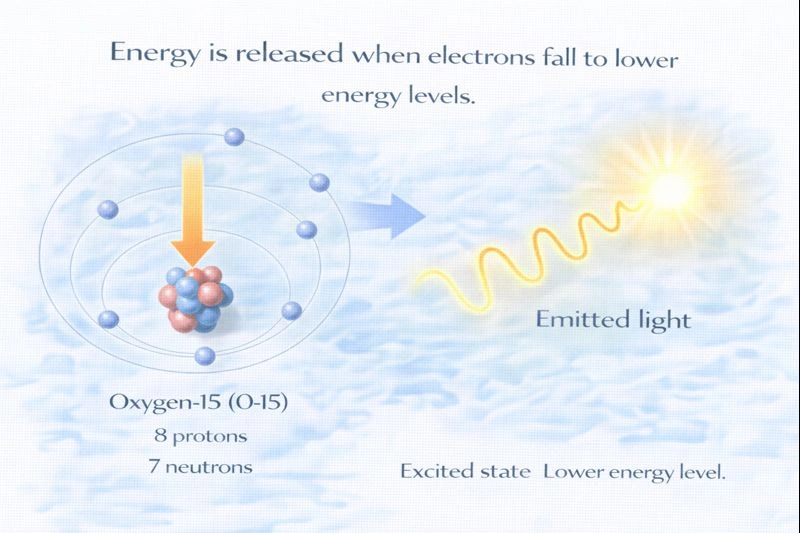
Energy is released when electrons fall from an excited state to a lower energy level.
Electrons occupy specific, quantized energy levels around the nucleus. When an electron is in an excited state, it has more energy than it does in its normal, lower-energy (ground) state. A system naturally tends toward lower energy and greater stability, so when an excited electron drops to a lower energy level, the excess energy must leave the atom. This energy is emitted as electromagnetic radiation, often in the form of light.
A. Energy is absorbed
Energy absorption occurs when an electron moves from a lower energy level to a higher one. That process requires an input of energy, such as heat or light, and is known as excitation. The question describes the opposite situation, where the electron is moving downward in energy.
B. Energy is released
When an excited electron falls to a lower energy level, it releases energy as a photon. The energy of the emitted photon is exactly equal to the difference between the two energy levels. This principle explains emission spectra, fireworks colors, neon lights, and many types of spectroscopy used in chemistry and physics.
C. Energy is both released and absorbed
A single electron transition involves only one direction of energy change. Moving up requires absorption of energy, while moving down results in the release of energy. Both cannot occur at the same time in a single transition.
D. Energy remains constant
If energy remained constant, the electron could not change energy levels. A transition between energy levels necessarily involves a change in energy, which must be conserved by releasing it to the surroundings.
Conclusion
When electrons move from higher to lower energy levels, the atom loses excess energy in the form of electromagnetic radiation. This release of energy is a fundamental process underlying light emission and atomic spectra, making option B the correct choice.
Topic Flashcards
Click to FlipThe term for the process that must happen before an electron can fall and emit light (i.e., how it gets to a high-energy state).
Excitation (or absorption of energy).
The specific name for the light particle whose energy equals the difference between two electron orbitals.
A photon.
The direct, observable result of many electrons in a sample falling from various excited states to lower ones.
An emission spectrum (a series of colored lines).
This fundamental law of physics explains why energy must be released as radiation when an electron moves to a lower level.
The Law of Conservation of Energy.
In a flame test, the characteristic color is produced by this specific atomic event.
Electrons falling from an excited state back to their ground state.