Which have about the same mass?
-
A
Protons and neutrons
-
B
Neutrons and electrons
-
C
Electrons and alpha particles
-
D
Neutrons and beta particles
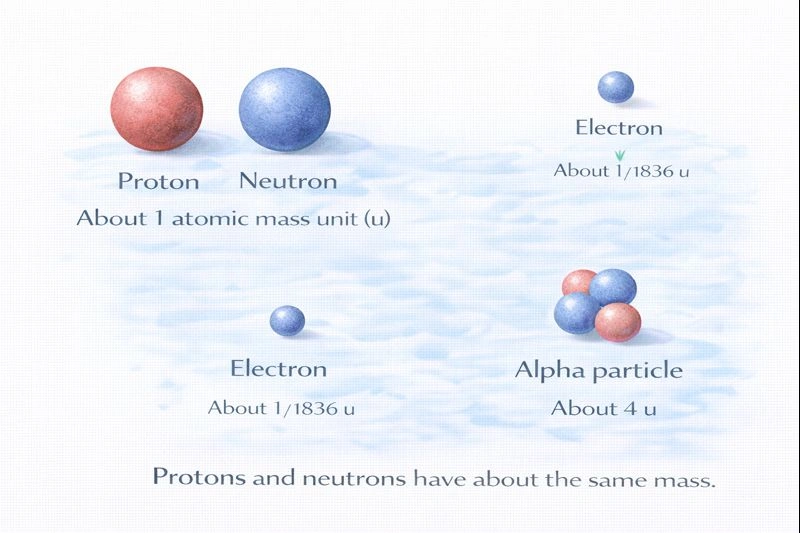
Protons and neutrons have about the same mass.
Subatomic particles differ greatly in mass, but two of them are nearly equal. Protons and neutrons are both nucleons located in the atomic nucleus, and their masses are almost identical. Each has a mass close to 1 atomic mass unit (u), which is why atomic mass is largely determined by the number of protons and neutrons rather than electrons.
A. Protons and neutrons
A proton has a mass of about 1.007 u, and a neutron has a mass of about 1.009 u. The difference between them is extremely small, so for most chemical and biological purposes, they are considered to have the same mass.
B. Neutrons and electrons
Electrons are much lighter than neutrons. An electron has a mass of about 1/1836 that of a proton or neutron, making the difference very large.
C. Electrons and alpha particles
An alpha particle consists of two protons and two neutrons, giving it a mass of about 4 atomic mass units. An electron’s mass is negligible in comparison.
D. Neutrons and beta particles
A beta particle is essentially an electron (or positron) emitted during radioactive decay. As with electrons in general, beta particles are far lighter than neutrons.
Conclusion
Among common subatomic particles, protons and neutrons are unique in having nearly identical masses. Their similar mass explains why they together account for almost all of an atom’s mass, making option A the correct choice.
Topic Flashcards
Click to FlipThese two nucleons are responsible for nearly all of an atom's mass.
Protons and neutrons.
The subatomic particle whose mass is so small it is considered negligible in atomic mass calculations.
The electron.
The mass of one proton is approximately equal to the mass of one ____.
Neutron.
An alpha particle has a mass about four times greater than that of a single one of these.
A proton or a neutron.
This type of radioactive emission (a beta particle) has a mass most similar to that of an electron.
A high-speed electron emitted from the nucleus.