Which pair are isotopes?
-
A
Co 60 and Kr 70
-
B
K 40 and Ar 40
-
C
O 15 and O 18
-
D
N 14 and C 14
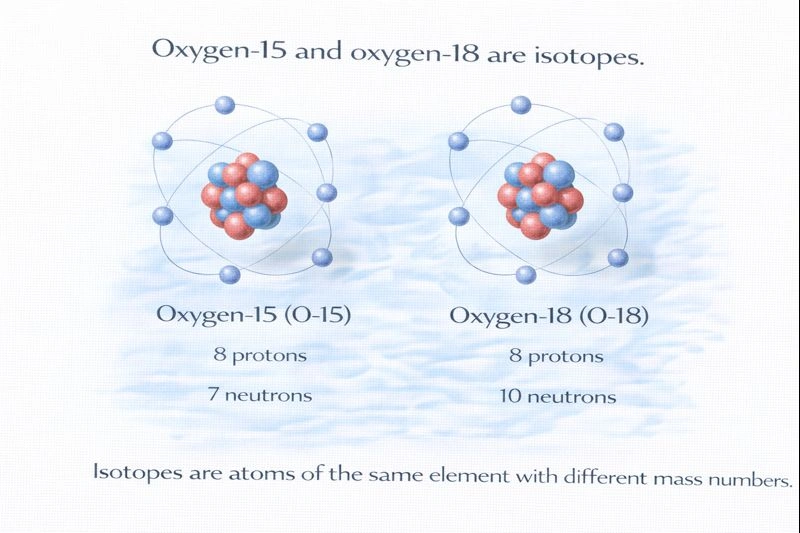
Oxygen-15 and oxygen-18 are isotopes.
Isotopes are atoms of the same element that have the same number of protons but different numbers of neutrons. Because the element is defined by its proton number (atomic number), isotopes must belong to the same element while differing in mass number.
A. Co-60 and Kr-70
Cobalt-60 and krypton-70 are different elements with different atomic numbers. Since isotopes must be atoms of the same element, this pair cannot be isotopes.
B. K-40 and Ar-40
Although potassium-40 and argon-40 share the same mass number, they are different elements with different atomic numbers. They are related through radioactive decay, but they are not isotopes of each other.
C. O-15 and O-18
Both atoms are oxygen, which means they each have 8 protons. Oxygen-15 has 7 neutrons, while oxygen-18 has 10 neutrons. The difference in neutron number gives them different mass numbers while remaining the same element.
D. N-14 and C-14
Nitrogen-14 and carbon-14 have the same mass number but different atomic numbers. Such atoms are called isobars, not isotopes.
Conclusion
Isotopes must be atoms of the same element with different mass numbers. Oxygen-15 and oxygen-18 meet this requirement, making option C the correct choice.
Topic Flashcards
Click to FlipAtoms of the same element that differ only in their number of neutrons.
Isotopes
The part of the atom that must be identical for two atoms to be isotopes of each other.
The number of protons (atomic number).
The term for atoms of different elements that have the same mass number (like N-14 and C-14).
Isobars.
Carbon-12 and Carbon-14 both have this many protons.
6.
The part of the atom that changes to create different isotopes of the same element.
The number of neutrons (and therefore the mass number).