Which of the following is a chief difference between evaporation and boiling?
-
A
Liquids boil only at the surface while they evaporate equally throughout the liquid.
-
B
Evaporating substances change from gas to liquid while boiling substances change from liquid to gas.
-
C
Evaporation happens in nature while boiling is a manmade phenomenon.
-
D
Evaporation can happen below a liquid's boiling point.
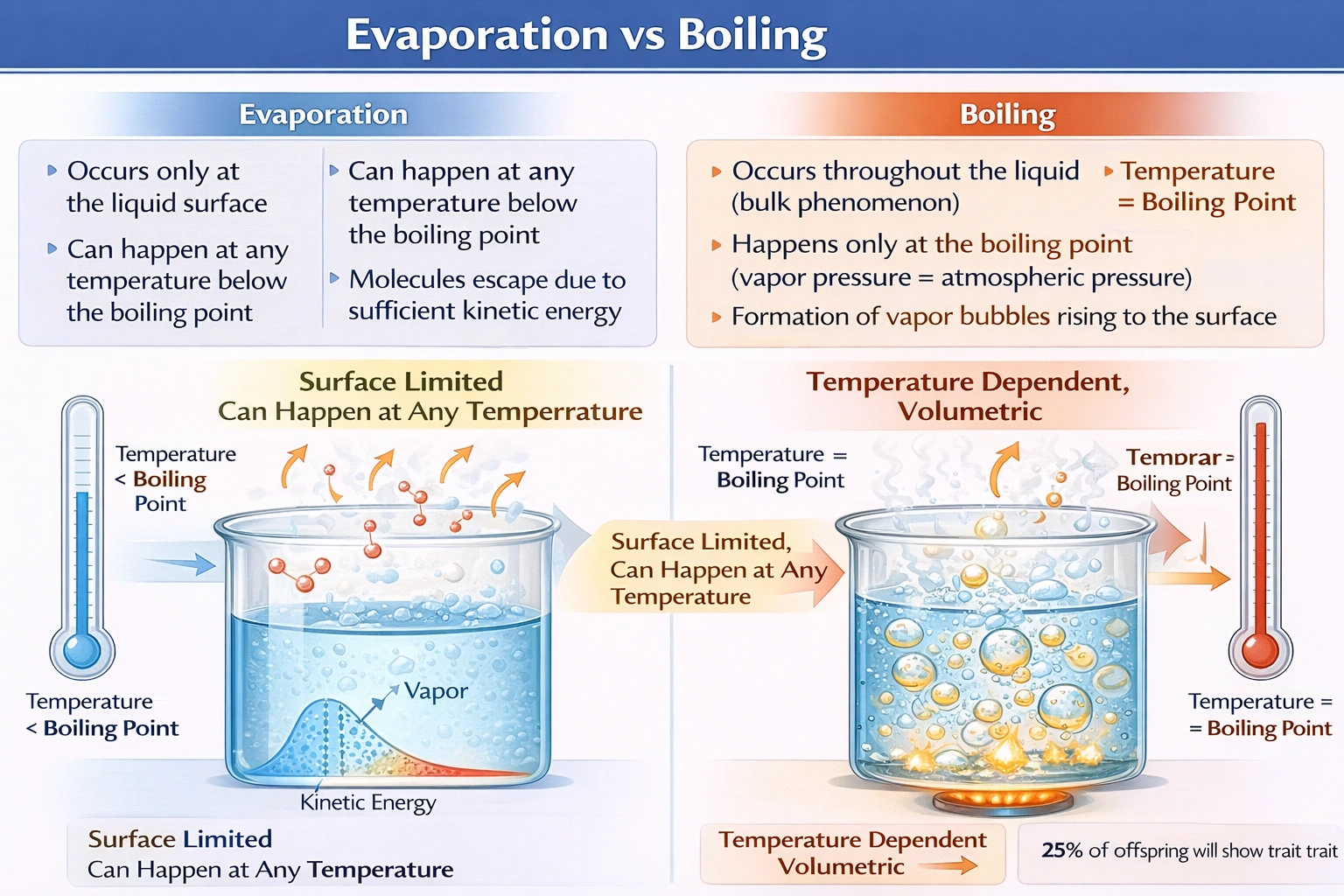
Evaporation can occur at temperatures below a liquid's boiling point, while boiling requires the liquid to reach its specific boiling temperature.
Evaporation represents a surface phenomenon where molecules with sufficient kinetic energy escape the liquid phase into vapor at any temperature, whereas boiling occurs throughout the liquid volume only when vapor pressure equals atmospheric pressure—requiring the liquid to attain its characteristic boiling point temperature under given pressure conditions.
A) Liquids boil only at the surface while they evaporate equally throughout the liquid
This option reverses the actual mechanisms. Evaporation occurs exclusively at the liquid-air interface where surface molecules gain enough kinetic energy to overcome intermolecular forces and escape into the vapor phase. Boiling, by contrast, involves vapor bubble formation throughout the liquid's bulk volume—not just at the surface—as vapor pressure exceeds atmospheric pressure internally. During boiling, bubbles nucleate at nucleation sites within the liquid and rise to the surface, demonstrating bulk-phase transformation rather than surface-limited escape.
B) Evaporating substances change from gas to liquid while boiling substances change from liquid to gas
This option fundamentally misrepresents phase change directions. Both evaporation and boiling describe liquid-to-gas transitions (vaporization), not condensation (gas-to-liquid). Condensation occurs when vapor molecules lose kinetic energy and return to the liquid phase—opposite to both evaporation and boiling. The distinction between evaporation and boiling lies in temperature requirements and location within the liquid, not in the direction of phase change, which remains identical for both processes.
C) Evaporation happens in nature while boiling is a manmade phenomenon
Both processes occur naturally without human intervention. Evaporation drives the water cycle as solar energy causes ocean and lake water to vaporize below 100°C. Boiling occurs naturally in geothermal features like hot springs and geysers where subsurface heating raises water temperature to its boiling point at local pressure conditions—Old Faithful geyser demonstrates natural boiling without artificial heat sources. Human cooking represents only one context for boiling; the physical process itself is governed by thermodynamic principles independent of human activity.
D) Evaporation can happen below a liquid's boiling point
Evaporation occurs continuously at temperatures below boiling point because liquid molecules possess a distribution of kinetic energies described by the Maxwell-Boltzmann distribution. Even at room temperature, a small fraction of surface molecules acquire sufficient energy to overcome intermolecular attractions and escape as vapor—explaining why puddles dry on cool days. Boiling, however, requires the entire liquid to reach a temperature where its vapor pressure equals external pressure, enabling vapor bubble formation throughout the liquid volume. This temperature threshold defines the boiling point (100°C for water at sea level), making evaporation possible at any temperature while boiling demands specific thermal conditions.
Conclusion:
The fundamental distinction between evaporation and boiling centers on temperature dependence and spatial occurrence within the liquid. Evaporation operates as a surface phenomenon possible at any temperature due to kinetic energy distribution among molecules, while boiling requires the liquid to reach its boiling point temperature to enable bulk vaporization through bubble formation. Option D correctly identifies this critical thermodynamic difference—evaporation's ability to occur below boiling point—whereas other options misrepresent spatial mechanisms, phase change directions, or natural occurrence patterns. Understanding this distinction proves essential for applications ranging from climate science (evaporative cooling) to industrial processes (distillation requiring controlled boiling).