Which of the following is a special property of water?
-
A
Water easily flows through phospholipid bilayers
-
B
A water molecule's oxygen atom allows fish to breathe.
-
C
Water is highly cohesive which explains its high melting point.
-
D
Water can self-hydrolyze and decompose into hydrogen and oxygen.
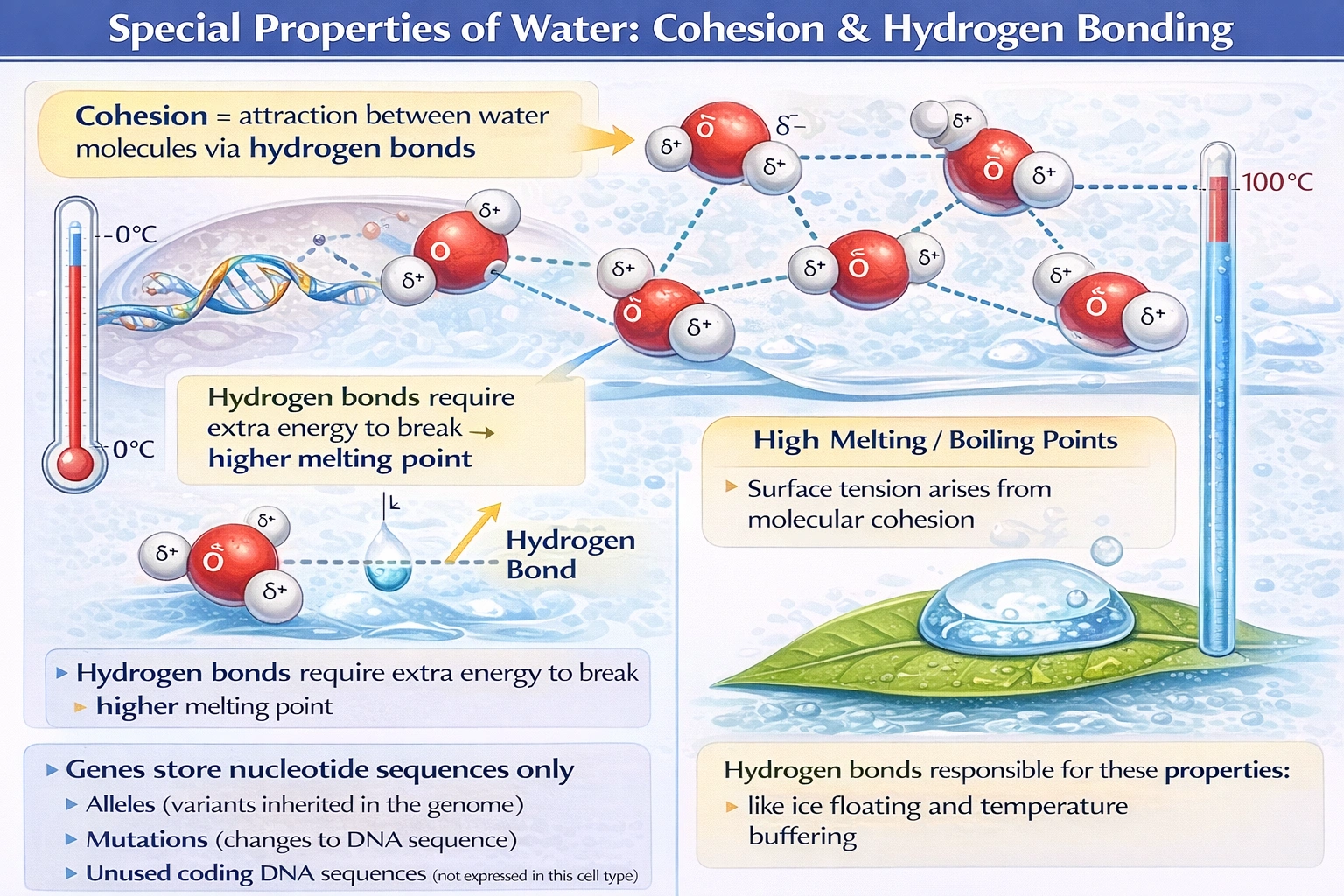
Water is highly cohesive which explains its high melting point.
Water molecules form up to four hydrogen bonds per molecule through electrostatic attraction between partially positive hydrogen atoms and partially negative oxygen atoms of adjacent molecules creating strong intermolecular cohesion that elevates phase transition temperatures far beyond expectations based on molecular mass alone.
A) Water easily flows through phospholipid bilayers
Water does not readily traverse intact phospholipid bilayers due to the hydrophobic fatty acid tails forming the membrane interior. Pure lipid bilayers exhibit low water permeability (approximately 10⁻³ cm/s), necessitating specialized aquaporin channel proteins to facilitate rapid osmotic water movement in biological systems. While small amounts of water diffuse slowly through membranes via transient defects, "easy flow" misrepresents membrane barrier function phospholipid bilayers specifically evolved to restrict uncontrolled water movement, maintaining cellular osmotic integrity.
B) A water molecule's oxygen atom allows fish to breathe
Fish extract dissolved molecular oxygen (O₂) from water—not oxygen atoms chemically bound within H₂O molecules. Water's oxygen atoms remain covalently bonded to hydrogen and are biologically unavailable for respiration; breaking these bonds requires substantial energy input (electrolysis). Aquatic organisms rely on O₂ gas dissolved in water from atmospheric exchange or photosynthesis—typically 5–10 mg/L concentration in oxygenated freshwater. Fish gills extract this dissolved O₂ through countercurrent exchange, but the oxygen atoms within water molecules themselves play no role in respiration and cannot be metabolically liberated by biological systems.
C) Water is highly cohesive which explains its high melting point
Water's melting point (0°C) and boiling point (100°C) are anomalously high compared to analogous group 16 hydrides: H₂S (mp = -85.5°C), H₂Se (mp = -65.7°C), and H₂Te (mp = -49°C) follow expected trends based on molecular weight, but water deviates dramatically upward due to hydrogen bonding. Each water molecule forms approximately 3.4 hydrogen bonds on average in liquid state, creating extensive intermolecular cohesion that requires substantial thermal energy to overcome during phase transitions. This cohesion also manifests in high surface tension (72.8 mN/m at 20°C), capillary action, and the density anomaly where ice floats—properties critical for aquatic ecosystem survival. While cohesion contributes significantly to elevated melting point, precise attribution requires acknowledging that both cohesion (intermolecular attraction) and the tetrahedral hydrogen-bonding network in ice collectively elevate phase transition temperatures.
D) Water can self-hydrolyze and decompose into hydrogen and oxygen
Pure water exhibits negligible spontaneous decomposition under standard conditions. The autoionization equilibrium (2H₂O ⇌ H₃O⁺ + OH⁻) produces only 10⁻⁷ M hydronium and hydroxide ions at 25°C—ionization, not decomposition into elemental gases. Thermodynamic stability prevents spontaneous H₂O → H₂ + ½O₂ decomposition (ΔG° = +237 kJ/mol), requiring continuous energy input via electrolysis, photolysis, or high-temperature catalysis. Water's exceptional stability enables its role as life's universal solvent; spontaneous decomposition would preclude aqueous biochemistry. Trace hydrogen production in natural waters results from radiolysis or microbial activity—not intrinsic water instability.
Conclusion:
Water's hydrogen-bonding network creates exceptional intermolecular cohesion responsible for multiple anomalous properties including elevated melting/boiling points, high specific heat capacity, density maximum at 4°C, and surface tension—all critical for terrestrial and aquatic life. While option C slightly oversimplifies by attributing high melting point solely to cohesion (the crystalline ice structure also contributes), it correctly identifies cohesion as a fundamental special property distinguishing water from similar molecules. Other options misrepresent membrane permeability (A), confuse dissolved O₂ with water's oxygen atoms (B), or falsely claim spontaneous decomposition (D). Water's cohesive hydrogen bonding remains its most biologically significant special property, enabling temperature buffering in organisms and environments, capillary transport in plants, and habitat stability through ice floating behavior.