What is the electrical charge of the nucleus?
-
A
A nucleus always has a positive charge.
-
B
A stable nucleus has a positive charge, but a radioactive nucleus may have no charge and instead be neutral.
-
C
A nucleus always has no charge and is instead neutral.
-
D
A stable nucleus has no charge and is instead neutral, but a radioactive nucleus may have a charge.
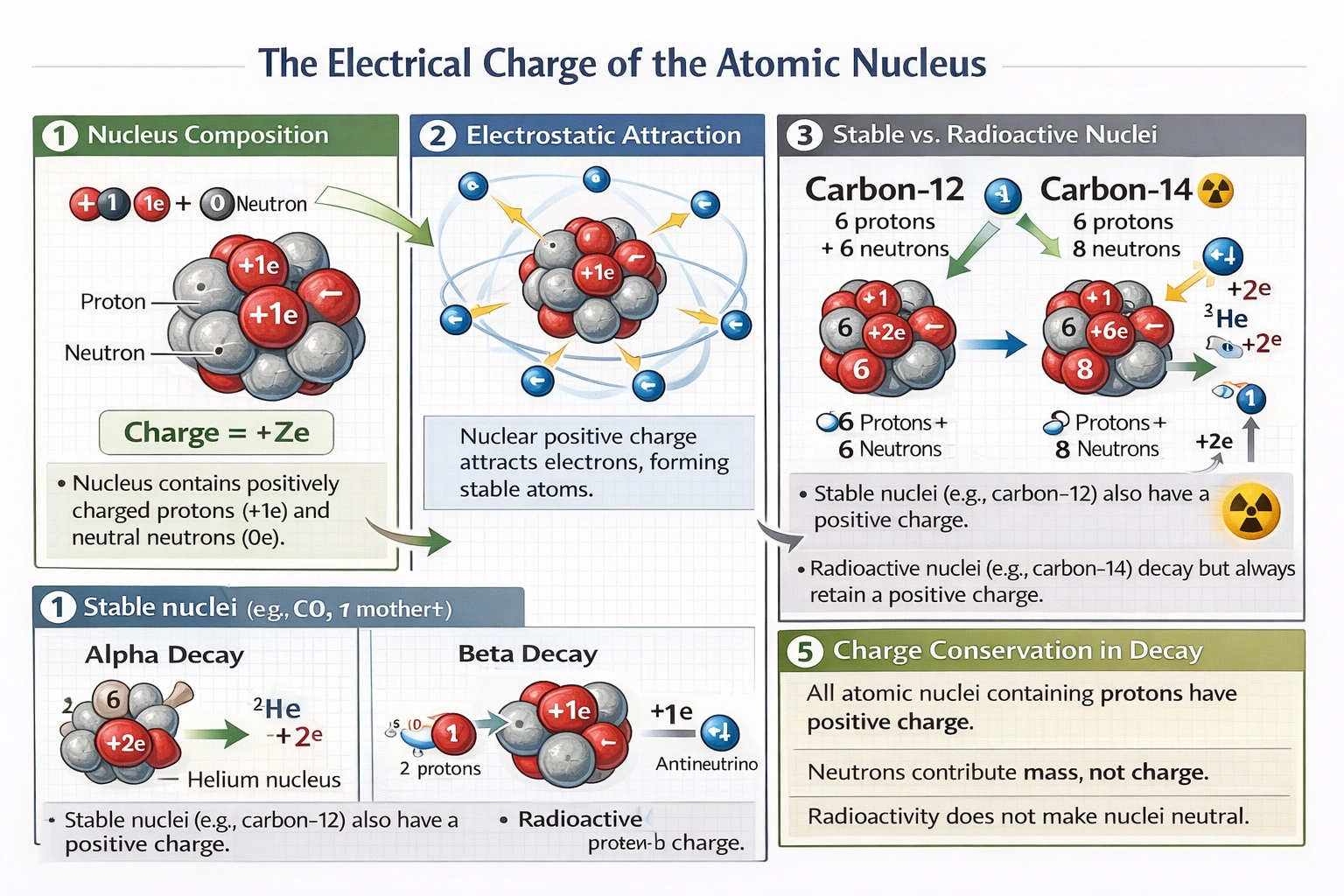
The atomic nucleus always possesses a net positive electrical charge due to its constituent protons, regardless of nuclear stability or radioactive properties.
Protons carry a fundamental positive charge (+1.602 × 10⁻¹⁹ C each) while neutrons remain electrically neutral; therefore, any nucleus containing protons—meaning all nuclei except the neutron itself—exhibits positive charge proportional to its atomic number (Z), with charge magnitude = +Ze where e represents elementary charge.
A) A nucleus always has a positive charge
All atomic nuclei except free neutrons contain protons and therefore carry positive charge. Hydrogen-1 (¹H) has +1e charge from its single proton; uranium-238 (²³⁸U) has +92e charge from 92 protons. Even radioactive nuclei like carbon-14 (⁶ protons = +6e charge) or polonium-210 (⁸₄ protons = +84e charge) maintain positive charge throughout decay processes—alpha decay reduces charge by +2e (losing helium nucleus), beta-minus decay increases charge by +1e (neutron → proton conversion), but the nucleus never becomes neutral or negative during these transformations. Nuclear charge stability is fundamental to atomic structure, enabling electron attraction that forms atoms.
B) A stable nucleus has a positive charge, but a radioactive nucleus may have no charge and instead be neutral
Radioactive nuclei maintain positive charge identical to stable isotopes of the same element. Carbon-12 (stable) and carbon-14 (radioactive) both possess six protons and therefore +6e nuclear charge. Radioactivity stems from nuclear instability due to proton-neutron ratio imbalances or excessive mass—not charge neutralization. During radioactive decay, charge conservation governs transformation products: alpha particles carry +2e charge away from parent nucleus, beta particles carry -1e charge (electrons) or +1e charge (positrons), but the daughter nucleus always retains positive charge appropriate to its new atomic number. No known radioactive decay pathway produces a neutral atomic nucleus.
C) A nucleus always has no charge and is instead neutral
This statement contradicts fundamental atomic theory established since Rutherford's gold foil experiment (1911). If nuclei were neutral, electrons would not experience electrostatic attraction to form stable atoms—matter as we know it could not exist. Cathode ray experiments, mass spectrometry, and particle accelerator data consistently demonstrate nuclear positive charge through deflection in electric and magnetic fields. The entire framework of chemistry depends on nuclear positive charge attracting electrons to form electron clouds with quantized energy levels. Neutral nuclei would eliminate chemical bonding and atomic structure.
D) A stable nucleus has no charge and is instead neutral, but a radioactive nucleus may have a charge
Both stable and radioactive nuclei possess positive charge proportional to proton count. Stability depends on the strong nuclear force balancing proton-proton electrostatic repulsion through optimal neutron mediation—not on charge presence or absence. Lead-208 (stable, 82 protons = +82e charge) and radium-226 (radioactive, 88 protons = +88e charge) both exhibit strong positive charge. Radioactive decay modes actually demonstrate charge conservation: when radium-226 undergoes alpha decay to radon-222, the +88e charge distributes as +86e (radon nucleus) + +2e (alpha particle). Charge neutrality never characterizes any atomic nucleus.
Conclusion:
Nuclear positive charge constitutes a universal, invariant property of all atomic nuclei containing protons—spanning stable and radioactive isotopes alike. This charge arises exclusively from protons (neutrons contribute mass without charge) and determines atomic number, electron count in neutral atoms, and chemical behavior. Radioactivity reflects nuclear instability from imbalanced proton-neutron ratios or excessive binding energy deficits—not charge variations. Options B, C, and D incorrectly suggest charge variability based on stability or radioactivity, contradicting conservation of charge principles and experimental evidence from over a century of nuclear physics. Option A correctly states the invariant positive charge of all atomic nuclei—a foundational principle enabling atomic structure, chemical bonding, and the existence of matter itself.