Why does potential energy increase as particles approach each other?
-
A
Attractive forces increase.
-
B
Attractive forces decrease.
-
C
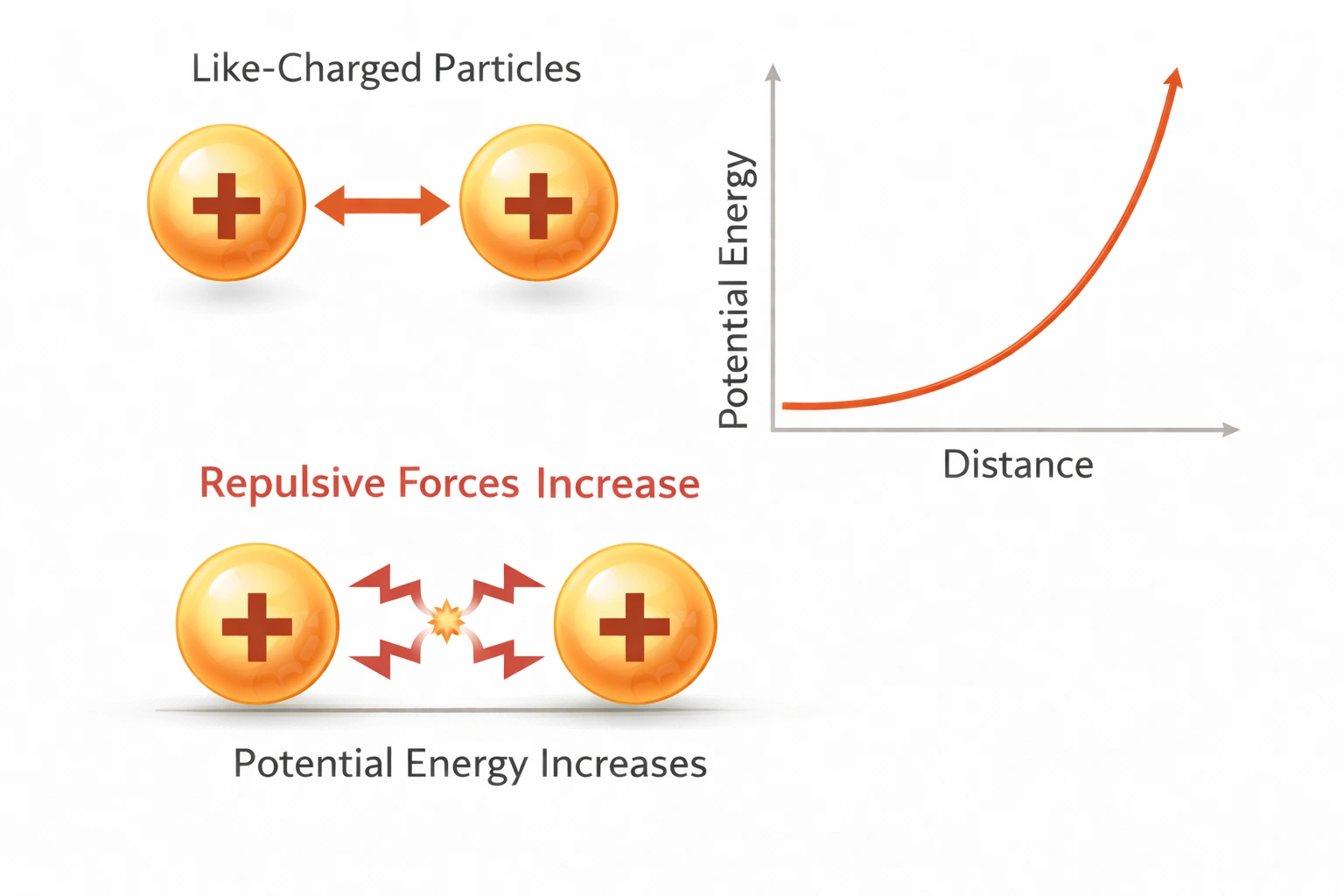
Repulsive forces increase.
-
D
Repulsive forces decrease.
Potential energy increases as particles approach each other when repulsive forces dominate, such as when two like-charged particles or compressed atomic nuclei come close.
In such cases, work must be done against the repulsive force, increasing stored potential energy.
A) Attractive forces increase
If attractive forces increase (e.g., opposite charges), potential energy decreases as particles approach, becoming more negative. This does not explain an increase in potential energy.
B) Attractive forces decrease
Decreasing attraction would reduce the rate of potential energy decrease but would not cause it to increase. This is inconsistent with energy trends in attractive systems.
C) Repulsive forces increase
This is correct. When particles with like charges or overlapping electron clouds approach, strong repulsion develops. Work must be done to push them together, increasing potential energy, similar to compressing a spring.
D) Repulsive forces decrease
Decreasing repulsion would lower potential energy, not raise it. This contradicts the behavior of systems where potential energy rises at short distances.
Conclusion
Potential energy increases during approach only when repulsive interactions dominate. The correct answer is C) Repulsive forces increase
Topic Flashcards
Click to FlipThe relationship between force and potential energy is given by F = -dU/dr (force is the negative gradient of potential energy). If the potential energy U increases as particles get closer (r decreases), what must be true about the sign and nature of the force F?
If U increases as r decreases, then dU/dr is negative (because U goes up when the change in r is down). Therefore, F = - (negative) = positive. A positive force is repulsive. This directly links the mathematical definition to the physical conclusion.
Compare two systems: (A) Two protons approaching each other. (B) A proton and an electron approaching each other. Sketch a graph for each showing Potential Energy (U) vs. Distance (r). Where does U increase with decreasing r? Explain the fundamental difference.
(A) Proton-Proton: U starts near zero at large r and increases sharply (curves upward) as r decreases due to strong electrostatic repulsion and later nuclear repulsion. (B) Proton-Electron: U is negative at large r (bound system) and decreases (becomes more negative) as r decreases due to electrostatic attraction. U increases on approach only for the repulsive (proton-proton) case.
In nuclear physics, when two nuclei are fused, they must overcome a tremendous potential energy "Coulomb barrier" to get close enough for the attractive strong nuclear force to bind them. Describe the work-energy process as they are pushed together.
As the positively charged nuclei approach, the repulsive electrostatic force increases (F ∝ 1/r²). External work must be done (e.g., via high temperature/kinetic energy in a fusion reactor) to push them together against this repulsive force. This work is stored as increased electric potential energy until they tunnel through or pass the barrier's peak.
A common analogy is compressing a spring. Explain how compressing a spring (where potential energy increases) is analogous to pushing two repelling particles together. Identify what represents the "force" and the "potential energy" in the particle scenario.
Analogy: The spring's restoring force (which pushes your hands apart) is like the interparticle repulsive force. The work you do to compress the spring against this force is stored as increased elastic potential energy in the spring, just as work done to push particles together is stored as increased interaction potential energy in the system.
Imagine a hypothetical pair of particles where the potential energy is given by U(r) = +A/r, where A is a positive constant. Is the force between them attractive or repulsive? Calculate the force function F(r) and determine if it increases or decreases as they approach.
1) Find Force: F = -dU/dr = -d/dr (A/r) = -(-A/r²) = +A/r². A positive force indicates repulsion. 2) As r decreases, F(r) = A/r² increases. This matches the condition in the original problem: increasing potential energy on approach correlates with an increasing repulsive force.