You rub a neutral plastic cup with a cotton ball, giving the cup a positive charge. What is true of the cotton ball?
-
A
It has lost electrons.
-
B
It has a negative charge.
-
C
It has lost neutrons.
-
D
It has more protons than electrons.
The cotton ball has a negative charge.
When two neutral objects are rubbed together, electrons can be transferred from one material to the other through a process known as the triboelectric effect. In this scenario, the plastic cup ends up with a positive charge, which means it has lost electrons. Those electrons must have been transferred to the cotton ball. As a result, the cotton ball gains excess electrons and becomes negatively charged.
A) It has lost electrons.
Losing electrons would cause an object to become positively charged. Since the plastic cup is the object that becomes positive, it is the one that lost electrons. The cotton ball, therefore, did not lose electrons in this interaction.
B) It has a negative charge.
The transfer of electrons from the plastic cup to the cotton ball leaves the cotton with more electrons than protons. This excess of electrons gives the cotton ball a net negative charge, which is consistent with the observed charging of the cup.
C) It has lost neutrons.
Neutrons are tightly bound within atomic nuclei and are not involved in everyday electrical interactions. Rubbing objects together cannot remove neutrons, making this statement physically impossible.
D) It has more protons than electrons.
Having more protons than electrons describes a positively charged object. In this case, the cotton ball gains electrons, so it ends up with more electrons than protons, not the other way around.
Conclusion
Because the plastic cup becomes positively charged by losing electrons, the cotton ball must gain those electrons. This electron gain gives the cotton ball a net negative charge.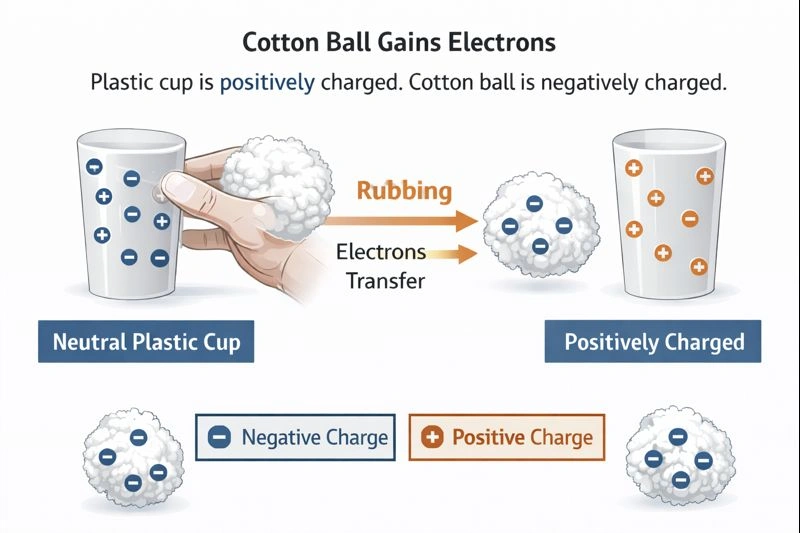
Topic Flashcards
Click to FlipWhat subatomic particle is physically transferred between objects during charging by friction (like rubbing)?
Electrons. (Protons are bound in the nucleus and do not move in everyday static electricity).
If a plastic cup becomes positively charged after rubbing, what must have happened to its electrons?
It must have lost electrons to the cotton ball.
In the triboelectric series, when two materials are rubbed, which one tends to become negative?
The material that is more likely to attract and hold electrons (higher on the series) becomes negative. In this case, the cotton ball.
What is the net charge of the plastic-cotton system before and after rubbing?
The total charge is conserved. It is zero (neutral) before and remains zero after. The cup gains positive charge (+), and the cotton gains an equal negative charge (–).
State the law of charge conservation in your own words.
Charge cannot be created or destroyed, only transferred from one object to another. The total charge in an isolated system remains constant.