When a car is driven for a long time, the pressure of air in the tires increases. This is best explained by which of the following gas laws?
-
A
Boyle’s Law
-
B
Charles’s Law
-
C
Gay Lussac’s Law
-
D
Dalton’s Law
The increase in air pressure inside car tires after long driving is explained by Gay-Lussac’s Law.
When a car is driven for an extended period, friction between the tires and the road, as well as flexing of the tire rubber, causes the air inside the tires to heat up. The volume of a tire remains nearly constant, so an increase in temperature leads directly to an increase in pressure. This direct relationship between temperature and pressure at constant volume is described by Gay-Lussac’s Law.
A) Boyle’s Law
Boyle’s Law describes the inverse relationship between pressure and volume at constant temperature. In a car tire, the volume does not significantly change during driving, so this law does not explain the observed increase in pressure.
B) Charles’s Law
Charles’s Law relates volume and temperature at constant pressure. Because the tire’s volume is constrained and pressure is not held constant, this law does not apply to the situation.
C) Gay Lussac’s Law
Gay-Lussac’s Law states that pressure is directly proportional to temperature when volume is constant. As the air inside the tire heats up, the kinetic energy of the gas molecules increases, causing more frequent and forceful collisions with the tire walls. This raises the pressure, matching what happens during prolonged driving.
D) Dalton’s Law
Dalton’s Law concerns the total pressure of a mixture of gases and the contribution of each gas’s partial pressure. The composition of air inside the tire does not change during driving, so this law is not relevant.
Conclusion
An increase in temperature at constant volume leads to an increase in pressure. This relationship explains why tire pressure rises during long drives and is described by Gay-Lussac’s Law.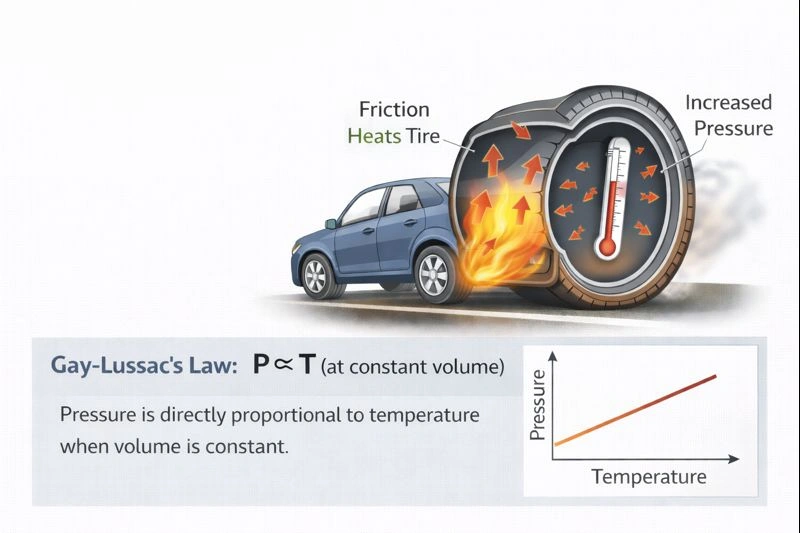
Topic Flashcards
Click to FlipState Gay-Lussac's Law in your own words.
The pressure of a fixed amount of gas at constant volume is directly proportional to its absolute temperature (P ∝ T).
Write the mathematical relationship for Gay-Lussac's Law.
P₁/T₁ = P₂/T₂, where P is pressure and T is absolute temperature (in Kelvin), provided volume and amount of gas are constant.
Why does the volume of a car tire remain approximately constant during a drive, making Gay-Lussac's Law applicable?
The tire's structure (rubber and rigid wheel rim) constrains its shape and volume, even as the air inside heats up and pressure increases.
If the air in a tire heats from 20°C (293 K) to 40°C (313 K) and the initial pressure was 32 psi, what is the approximate new pressure? (Use P₁/T₁ = P₂/T₂)
P₂ = P₁ * (T₂/T₁) = 32 psi * (313 K / 293 K) ≈ 32 psi * 1.068 ≈ 34.2 psi.
Which two gas laws are combined to form the Combined Gas Law, and what does Gay-Lussac's Law contribute to it?
Boyle's Law (P-V inverse relationship), Charles's Law (V-T direct relationship), and Gay-Lussac's Law (P-T direct relationship). The Combined Gas Law is P₁V₁/T₁ = P₂V₂/T₂.