HESI A2 ANATOMY AND PHYSIOLOGY PRACTICE EXAM
This HESI A2 Anatomy and Physiology practice exam provides a comprehensive quiz experience covering the major systems of the human body. It is ideal for final preparation and self-assessment before taking the actual exam.
Topics Covered
Organ Systems
Physiological Processes
Structure and Function
Body Regulation
00:00
A man has a blood pressure of 142/78. The 142 is indicative of
A.
peak pressure applied against artery walls during contraction of the heart
B. the contraction phase brought on by depolarization and contraction of the ventricles
C. remaining pressure within the arterial system during cardiac relaxation
D. the volume of blood pumped out of the right and left ventricles
Rationale
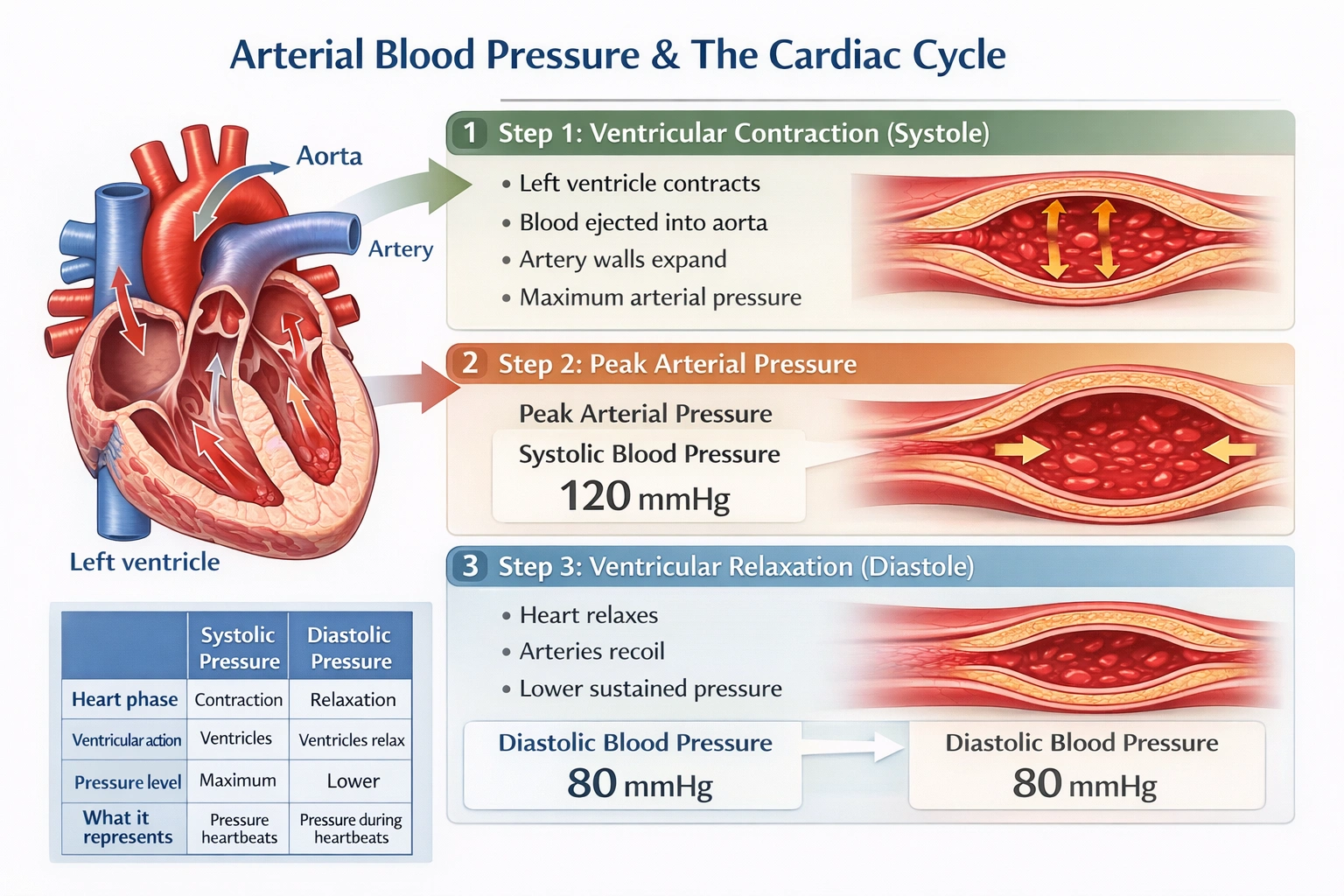
The 142 represents the peak pressure applied against artery walls during contraction of the heart.
In a blood pressure reading, such as 142/78 mmHg, the first number represents the systolic pressure the peak force exerted against the walls of the arteries when the heart contracts. The second number represents the diastolic pressure the residual force maintained in the arteries when the heart is at rest between beats. Understanding these two values is essential for interpreting cardiovascular health and identifying potential risks.
A. Peak pressure applied against artery walls during contraction of the heart
The value 142 mmHg is the systolic blood pressure, which measures the maximum force exerted on arterial walls during ventricular contraction (systole). This peak occurs when the left ventricle pushes blood into the aorta and through the circulatory system. A systolic reading of 142 mmHg falls within the elevated or Stage 1 hypertension range, indicating increased cardiovascular strain.
B. The contraction phase brought on by depolarization and contraction of the ventricles
This option describes the physiological event of systole, which is the phase of the cardiac cycle when the ventricles contract after electrical depolarization. While systole is responsible for generating systolic pressure, the number 142 itself quantifies the resulting pressure not the phase of contraction. Thus, this choice inaccurately defines what the numerical value represents.
C. Remaining pressure within the arterial system during cardiac relaxation
This accurately defines diastolic pressure, the second number in a blood pressure reading. In 142/78 mmHg, the diastolic value is 78 mmHg, reflecting the steady pressure maintained in the arteries when the heart is relaxed between beats. Therefore, this description does not correspond to the first number (142).
D. The volume of blood pumped out of the right and left ventricles
This describes stroke volume (or collectively, cardiac output), which is the amount of blood ejected by the ventricles per beat. Blood pressure, however, measures force per unit area (in mmHg), not volume. Although stroke volume influences blood pressure, the numerical value 142 represents pressure, not volume.
Conclusion:
The first number in a blood pressure reading (142 in this case) specifically denotes systolic pressure the peak force exerted on arterial walls during heart contraction. It does not refer to the phase of contraction itself, the resting diastolic pressure, or the volume of blood ejected. Accurate interpretation of these values is crucial for clinical assessment and management of cardiovascular health.
Mineralocorticoids
A.
control sodium and water transport
B. influence secondary sex hormones
C. regulate blood pressure, respiration, and temperature
D. regulate sugar and protein metabolism
Rationale
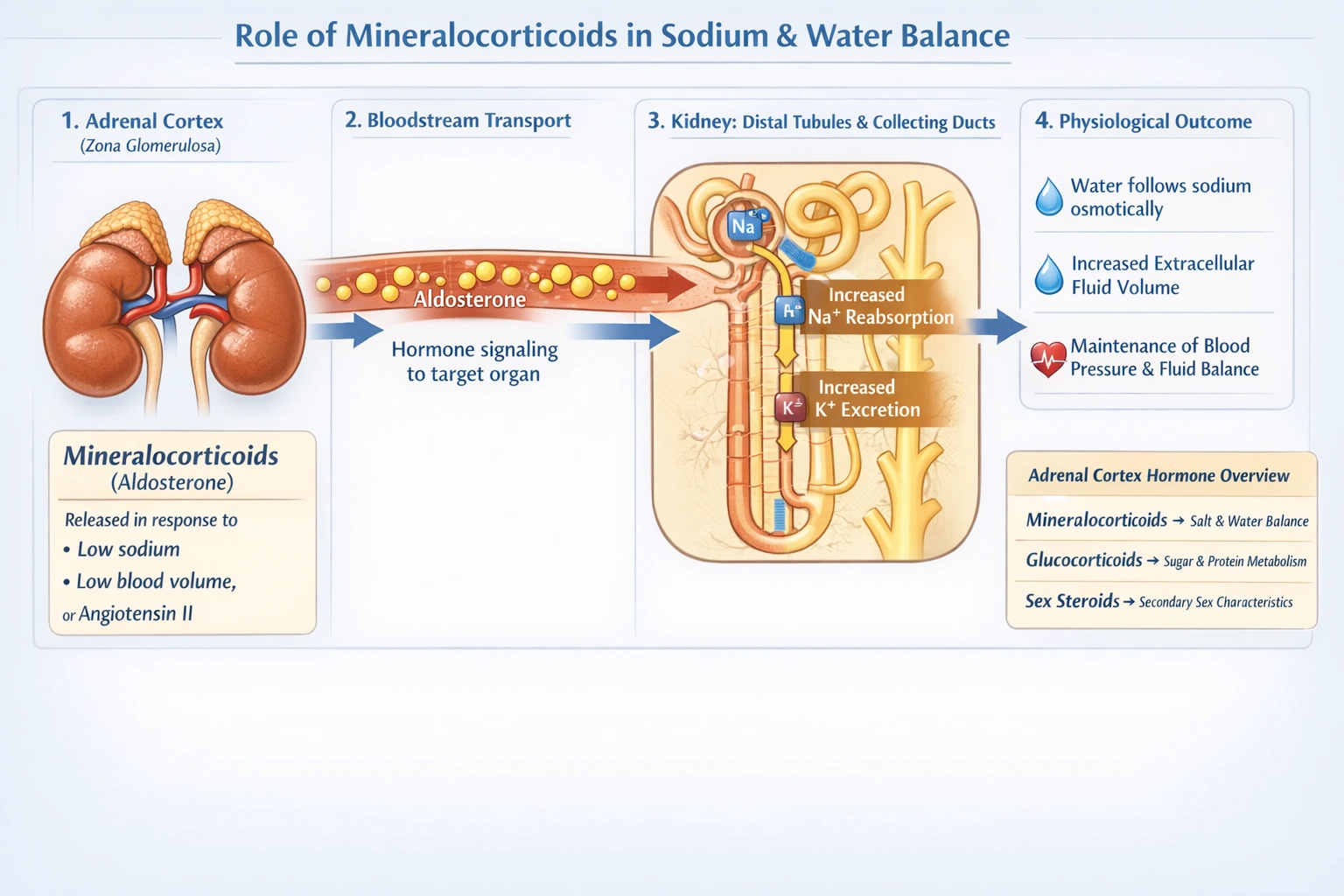
Mineralocorticoids control sodium and water transport.
Mineralocorticoids are a class of steroid hormones whose primary and defining physiological role is the regulation of sodium and potassium balance, which directly governs water transport and extracellular fluid volume. The principal mineralocorticoid, aldosterone, acts on the kidneys' distal tubules and collecting ducts to promote sodium reabsorption and potassium excretion, with water following sodium osmotically.
A) Control sodium and water transport
Mineralocorticoids, chiefly aldosterone, directly target ion transport in the kidneys. By increasing sodium reabsorption, they are the primary hormonal regulators of sodium balance. This action secondarily determines water retention and total body fluid volume, making this function central to their name and role.
B) Influence secondary sex hormones
Secondary sex characteristics are influenced by androgens, estrogens, and progesterone. While the adrenal cortex produces some weak androgens, these are not classified as mineralocorticoids. Mineralocorticoids like aldosterone have no significant role in the development or regulation of secondary sex characteristics.
C) Regulate blood pressure, respiration, and temperature
Mineralocorticoids contribute indirectly to long-term blood pressure regulation through their effect on blood volume. However, they do not regulate respiration, which is controlled by brainstem centers and chemoreceptors, or core body temperature, which is managed by the hypothalamus. This option incorrectly attributes broad autonomic functions to this specific hormone class.
D) Regulate sugar and protein metabolism
The regulation of sugar and protein metabolism is the primary function of glucocorticoids, such as cortisol. These hormones promote gluconeogenesis and protein catabolism. Mineralocorticoids have negligible effects on these metabolic pathways, as their function is specialized for electrolyte balance.
Conclusion:
The question asks for the specific function of mineralocorticoids. Their essential and dedicated role is in controlling sodium reabsorption and consequent water transport, which distinguishes them from other steroid hormone classes. Therefore, the correct answer is that mineralocorticoids control sodium and water transport.
The __________ is a hollow tube or passageway that extends from the pharynx to the stomach.
A.
duodenum
B. esophagus
C. oral cavity
D. pyloric sphincter
Rationale
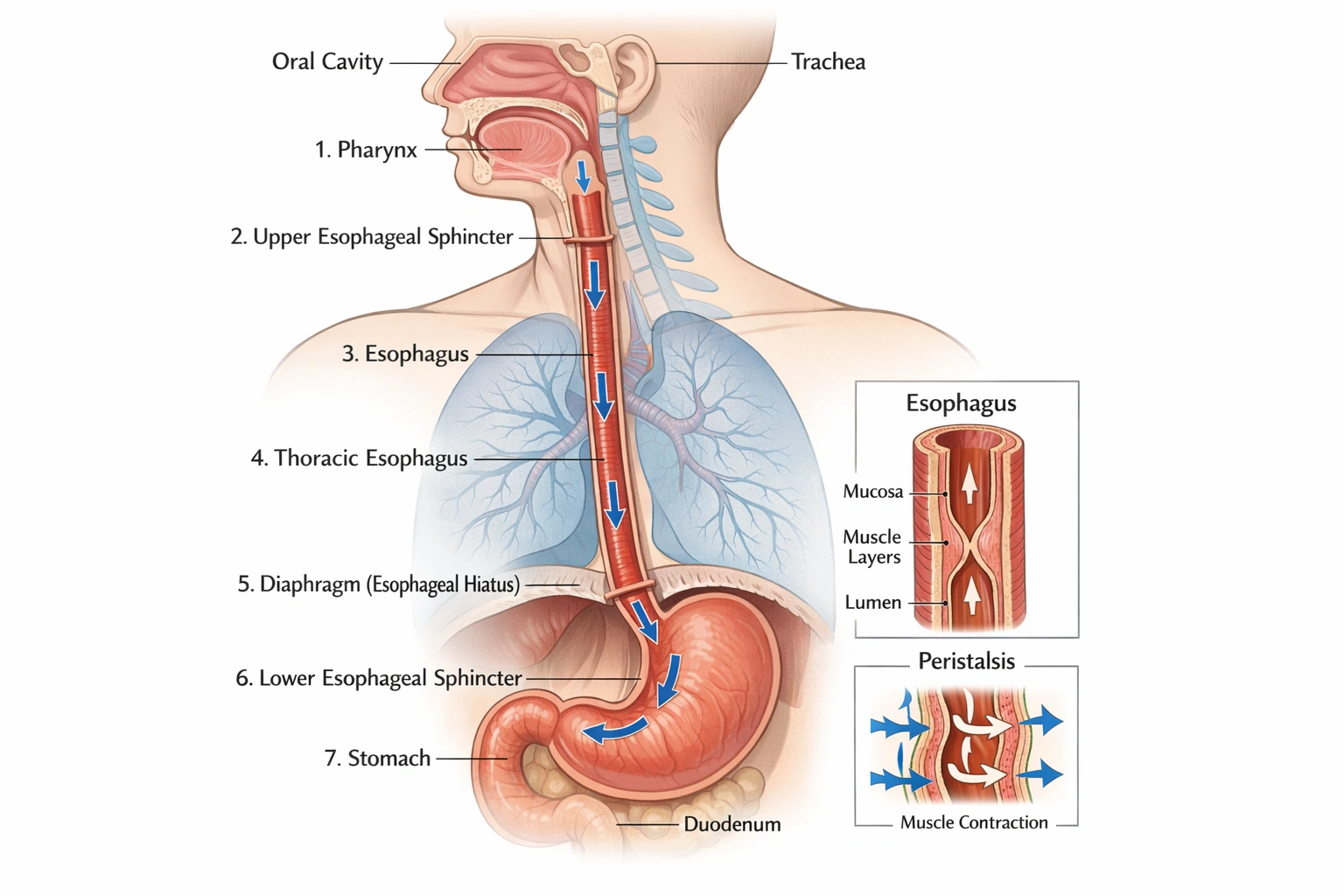
The esophagus is the hollow tube that extends from the pharynx to the stomach.
The esophagus is the muscular conduit that specifically transports food and liquids from the pharynx to the stomach the duodenum is the first part of the small intestine the oral cavity is the mouth which is anterior to the pharynx and the pyloric sphincter is a muscular valve controlling passage from the stomach to the small intestine.
A) Duodenum
The duodenum is the first and shortest segment of the small intestine. It receives partially digested food, or chyme, from the stomach via the pyloric sphincter. Its role is critical in chemical digestion, but it is located after the stomach, not between the pharynx and the stomach.
B) Esophagus
The esophagus perfectly matches the description of a hollow tube extending from the pharynx to the stomach. It is a distinct anatomical structure within the digestive tract whose sole function is transit, connecting the throat to the stomach for the passage of ingested material.
C) Oral cavity
The oral cavity, or mouth, is the initial chamber of the digestive tract where ingestion and mechanical digestion begin. It is bounded by the lips, cheeks, palate, and tongue. It does not extend to the stomach but rather leads into the oropharynx, which then connects to the esophagus.
D) Pyloric sphincter
The pyloric sphincter is a powerful ring of smooth muscle that forms the valve between the lower end of the stomach, the pylorus, and the duodenum. Its function is to regulate the release of gastric contents into the small intestine. It is a sphincter muscle, not a tube, and it is situated completely downstream from the stomach.
Conclusion:
The question describes the anatomical pathway for swallowed material prior to entering the stomach. The only structure that fulfills the role of a hollow tube connecting the pharynx directly to the stomach is the esophagus. Therefore, the correct answer is the esophagus.
__________ is an example of passive acquired immunity.
A.
Acquired from having the disease
B. Gamma globulin injection
C. Opsonization
D. Vaccination
Rationale
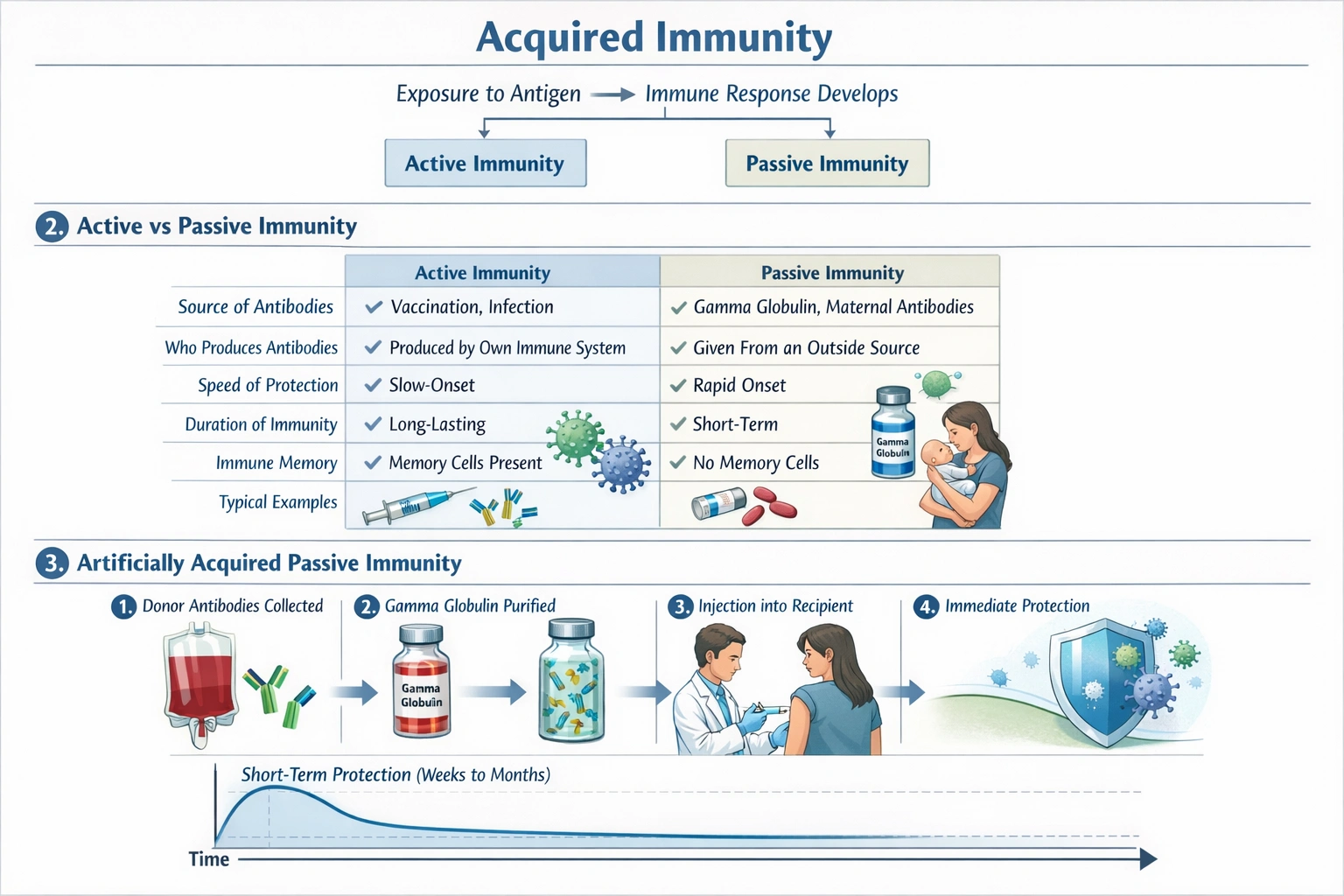
Gamma globulin injection is a definitive example of artificially acquired passive immunity.
This involves the direct administration of preformed antibodies, harvested from the pooled plasma of many donors, into a recipient. These antibodies provide immediate, short term protection against specific pathogens or toxins by neutralizing them directly in the bloodstream. The recipient's own immune system is not stimulated to produce a response; it merely receives the temporary benefit of these borrowed defenses, which last only weeks to months before being naturally broken down.
A) Acquired from having the disease
This describes naturally acquired active immunity. When a person contracts an infection, their own immune system actively responds by producing antibodies and generating memory cells. This process requires time to develop but results in long lasting protection. It is considered "active" because the individual's body produces the immune response, and "natural" because the exposure occurs through normal environmental contact, not a medical procedure.
B) Gamma globulin injection
A gamma globulin injection delivers a concentrated solution of preformed antibodies. This provides immediate immunity without any requirement for the recipient's immune system to be activated. It is "passive" because antibodies are transferred, not made by the host, and "artificially acquired" because the transfer is achieved through a clinical intervention rather than a natural process like placental antibody transfer from mother to fetus.
C) Opsonization
Opsonization is a biological process, not a category of acquired immunity. It refers to the coating of a pathogen by antibodies or complement proteins, which marks it for more efficient engulfment and destruction by phagocytic cells like macrophages. While opsonization is a crucial function of antibodies provided by either active or passive immunity, it is not itself a method of acquiring immunity.
D) Vaccination
Vaccination is the primary method for inducing artificially acquired active immunity. A vaccine introduces an antigen, such as a weakened virus or a protein subunit, to safely stimulate the recipient's own immune system. This leads to the production of memory cells and antibodies. The immunity is "active" because the body generates its own response, and "artificial" because the exposure is deliberate and mediated by medical science.
Conclusion:
The question asks for an example of passive acquired immunity, which is characterized by the transfer of preformed antibodies to provide immediate, temporary protection. Only the gamma globulin injection fits this precise definition, as it involves the artificial transfer of antibodies without activating the recipient's immune system. Therefore, the correct answer is gamma globulin injection.
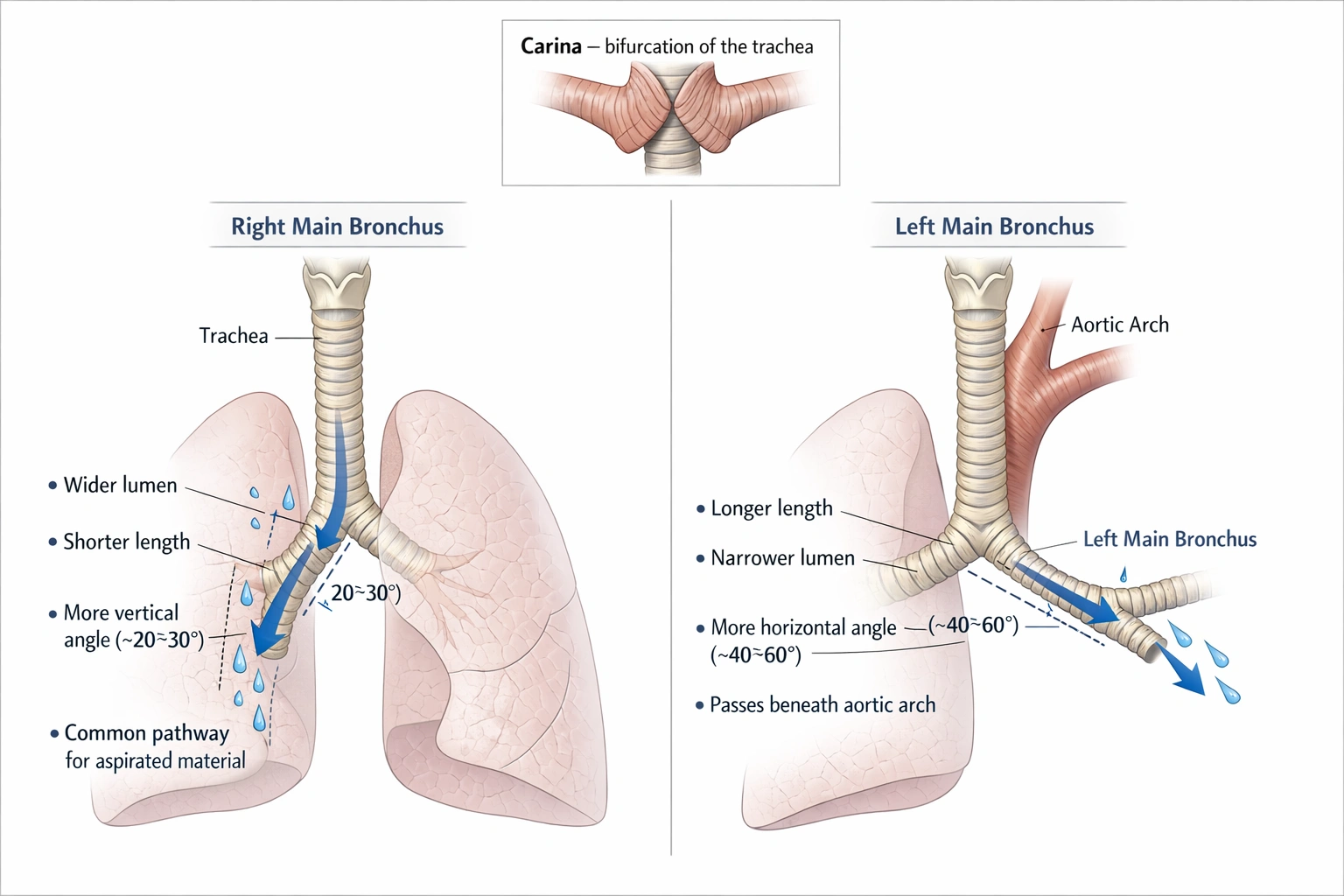
The risk of aspiration is greater in the right main stem bronchus because
A.
of the difference in pressure gradients between the left and right stem bronchi
B. it does not participate in gas exchange
C. it is shorter, wider, and more vertical
D. of its association with the trachea
Rationale
The right main stem bronchus presents a greater risk for aspiration due to its distinct anatomical configuration.
Compared to the left main bronchus, the right bronchus is shorter in length, has a wider diameter, and branches from the trachea at a more vertical angle. This combination creates a direct, low-resistance pathway. During aspiration, gravity and momentum more easily carry fluids or foreign objects straight down this wider, more vertically aligned conduit, often leading to material entering the right lower lobe.
A) Of the difference in pressure gradients between the left and right stem bronchi
Airway pressures within the tracheobronchial tree equalize rapidly during normal breathing and even during aspiration events. There is no sustained, significant pressure differential between the two main bronchi that would selectively pull material into the right side. The movement of aspirated material is governed more by anatomy, gravity, and the force of the aspiration event than by pressure gradients.
B) It does not participate in gas exchange
This is a true statement but not a differentiating factor. Both the right and left main stem bronchi are part of the conducting zone of the respiratory system, meaning neither participates in actual gas exchange. That function is reserved for the alveoli. Since this characteristic is shared, it cannot explain why aspiration is more common on the right side.
C) It is shorter, wider, and more vertical
The right main bronchus is approximately 2.5 cm long, wider in diameter, and deviates from the trachea at an angle of about 20 to 30 degrees, making it a near-vertical continuation. In contrast, the left main bronchus is longer (about 5 cm), narrower, and takes a more horizontal course at a sharper angle of 40 to 60 degrees. This anatomy makes the right bronchus the path of least resistance.
D) Of its association with the trachea
Both main bronchi are directly associated with the trachea as its two primary branches. This vague statement does not provide a specific reason for the increased risk on the right side, as the association is common to both structures.
Conclusion:
Aspirated material is more likely to enter the right lung due to the inherent anatomical design of the right main bronchus. Its shorter length, larger diameter, and more vertical orientation facilitate the direct passage of foreign substances, a key consideration in clinical settings like emergency medicine and anesthesiology.
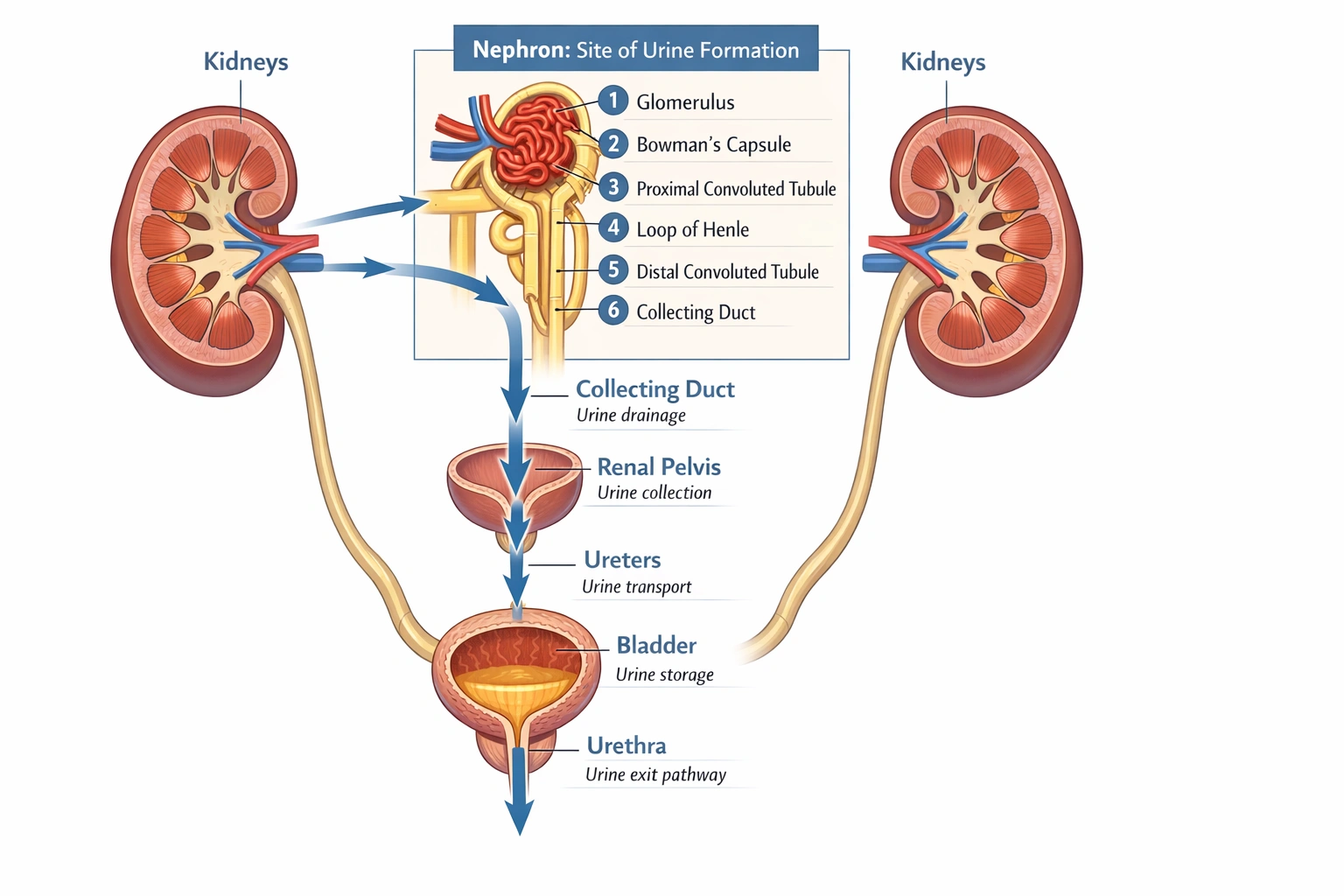
Urine is expelled from the body via the
A.
collecting duct
B. tubules
C. ureters
D. urethra
Rationale
Urine is expelled from the body through the urethra.
This muscular tube serves as the final conduit of the urinary system, extending from the urinary bladder to the external urethral orifice. The process of micturition involves the coordinated relaxation of urethral sphincters and contraction of the bladder's detrusor muscle to force urine out through this passage.
A) Collecting duct
The collecting duct is a microscopic structure within the kidney's nephron system. It is where final urine concentration occurs through water reabsorption regulated by antidiuretic hormone. Its function is to deliver formed urine into the renal pelvis, not to transport it out of the body. It remains entirely within the kidney.
B) Tubules
Renal tubules, including the proximal and distal convoluted tubules and the loop of Henle, are the sites of filtration, reabsorption, and secretion during urine formation. They are essential for modifying the filtrate into urine but are internal processing structures located entirely within the kidney. They do not convey urine outside the organ or the body.
C) Ureters
The ureters are paired muscular tubes that transport urine from the renal pelves of the kidneys to the urinary bladder via peristaltic contractions. They function as conduits between the sites of urine production and storage. However, they are not involved in the final expulsion of urine from the body; that role belongs to the urethra.
D) Urethra
The urethra is the terminal duct of the urinary system, responsible for discharging urine from the bladder to the exterior. In males, it also serves as a passage for semen, but its primary urinary function is universal. Its location and function make it the definitive structure for expulsion.
Conclusion:
The urinary system employs a series of specialized structures for the formation, transport, storage, and elimination of urine. While the collecting ducts and tubules are involved in urine production within the kidney, and the ureters transport it to the bladder, only the urethra serves as the final pathway for expelling urine from the body.
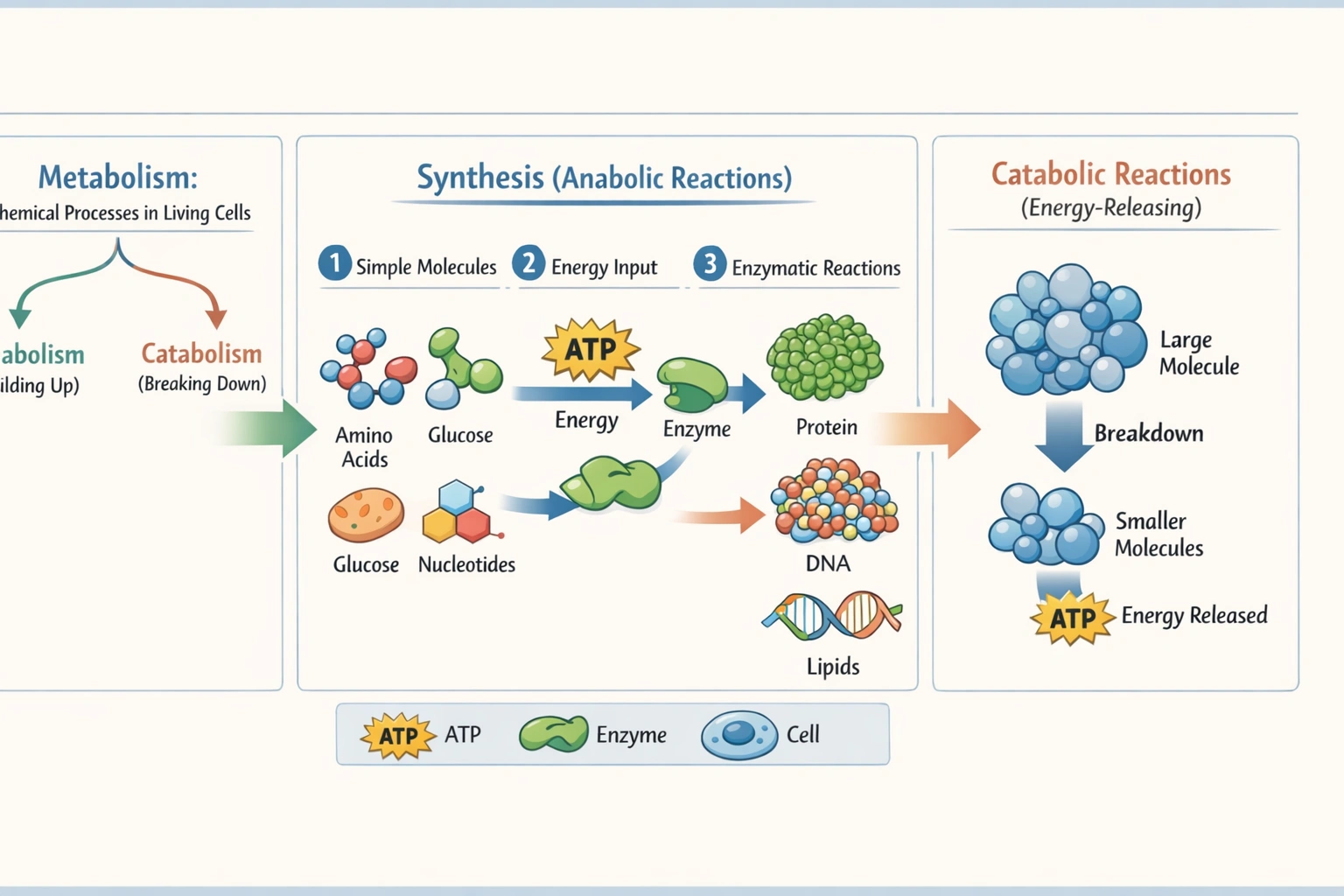
__________ is defined as the production of a substance by the coming together of chemical elements, groups, or simpler compounds or by the degradation of a complex compound where simple molecules are built larger and more complex.
A.
Excretion
B. Regulation
C. Respiration
D. Synthesis
Rationale
Synthesis is the biochemical process defined by the assembly of complex substances from simpler components.
This anabolic process involves the energy-requiring combination of chemical elements, functional groups, or simple compounds to form larger, more complex molecules. It is the constructive core of metabolism, encompassing essential functions like protein formation, DNA replication, and polysaccharide production.
A) Excretion
Excretion is the physiological process of eliminating metabolic waste products, such as urea, carbon dioxide, and excess salts, from an organism. It is a catabolic and eliminative function that removes simple breakdown products, which is the direct opposite of the constructive, building process described.
B) Regulation
Regulation refers to the control systems that maintain internal balance and coordinate bodily functions. While regulation governs the rate and timing of synthetic processes, the term itself describes a control mechanism, not the specific chemical act of building complex molecules from simpler parts.
C) Respiration
Cellular respiration is a catabolic pathway that breaks down complex organic molecules, like glucose, into simpler products to release energy. It is a degradative process that produces carbon dioxide and water, which contrasts with the anabolic, constructive process outlined in the question.
D) Synthesis
Synthesis precisely matches the given definition: it is the production of a complex compound through the combination of simpler chemical units. This includes all anabolic reactions in the body, such as forming proteins from amino acids or synthesizing glycogen from glucose molecules.
Conclusion:
The description of building larger, more complex molecules from simpler components is the definitive characteristic of synthesis, distinguishing it from excretory, regulatory, or energy-releasing processes.
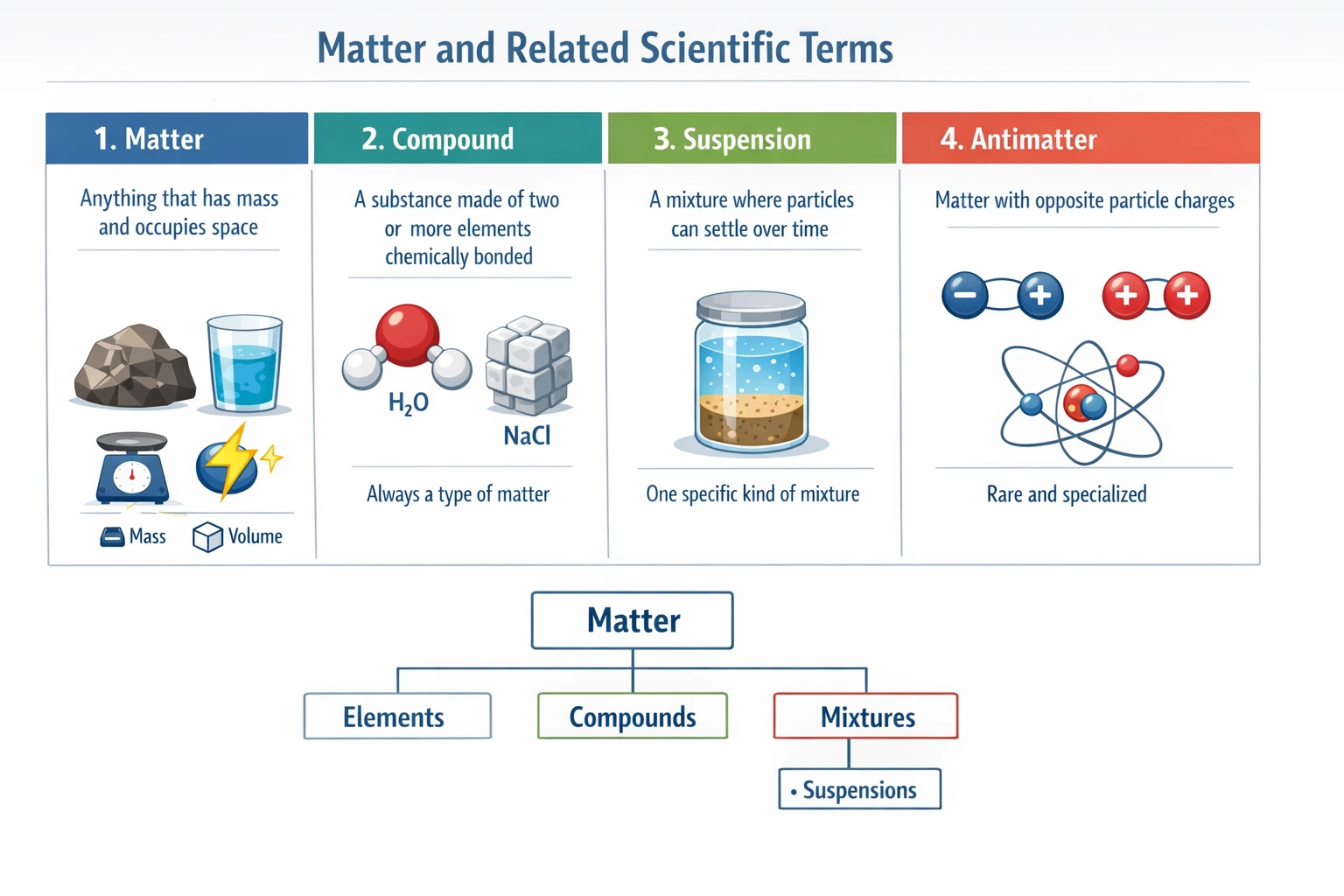
Anything that has weight and occupies space is considered (a)
A.
antimatter
B. compound
C. matter
D. suspension
Rationale
Matter is defined as anything that possesses mass and occupies space.
This classical definition encompasses all tangible objects and materials, regardless of their state, composition, or properties. Mass gives matter weight in a gravitational field, while volume refers to the three-dimensional space it occupies.
A) Antimatter
Antimatter refers to a specific and rare category of material composed of particles that have properties opposite to those of ordinary matter, such as positrons instead of electrons. While antimatter particles do possess mass and volume, the term describes a specialized subset within the broader classification of matter. In both everyday language and foundational science, the general term for any substance with mass and volume is "matter." Therefore, using "antimatter" as the defining answer would be incorrect, as it does not represent the universal concept described in the question.
B) Compound
A compound is a specific type of pure substance formed when two or more different chemical elements are chemically bonded together in a fixed proportion, such as water (Hâ‚‚O) or carbon dioxide (COâ‚‚). Although all compounds are indeed matter, the reverse is not true: matter includes a vast array of substances that are not compounds, including pure elements (like oxygen gas or gold) and various mixtures (like air or soil). The definition provided in the question is the universal definition of matter itself, not the narrower category of chemical compounds.
C) Matter
Matter is the all-encompassing scientific term for any physical substance that has mass and volume. It includes everything from atoms and subatomic particles to stars and galaxies, and it exists in various states (solid, liquid, gas, plasma) and forms (elements, compounds, mixtures). This definition is foundational in physics and chemistry, as it establishes the basic "stuff" that makes up the observable universe and undergoes physical and chemical changes.
D) Suspension
A suspension is a specific type of heterogeneous mixture in which solid particles are dispersed in a liquid but are large enough to eventually settle out due to gravity. Examples include sand in water or muddy river water. While a suspension is one particular form that matter can take, it represents only a tiny fraction of possible material substances. Most matter including pure elements, homogeneous solutions, and gases does not fit the definition of a suspension. Thus, this term is far too narrow to describe the universal concept presented in the question.
Conclusion:
The question provides the classical definition of the most fundamental category in physical science. While antimatter, compounds, and suspensions are all specific examples or subtypes of material substances, only the term "matter" universally and accurately describes anything that has mass and occupies space. Therefore, the correct answer is matter.
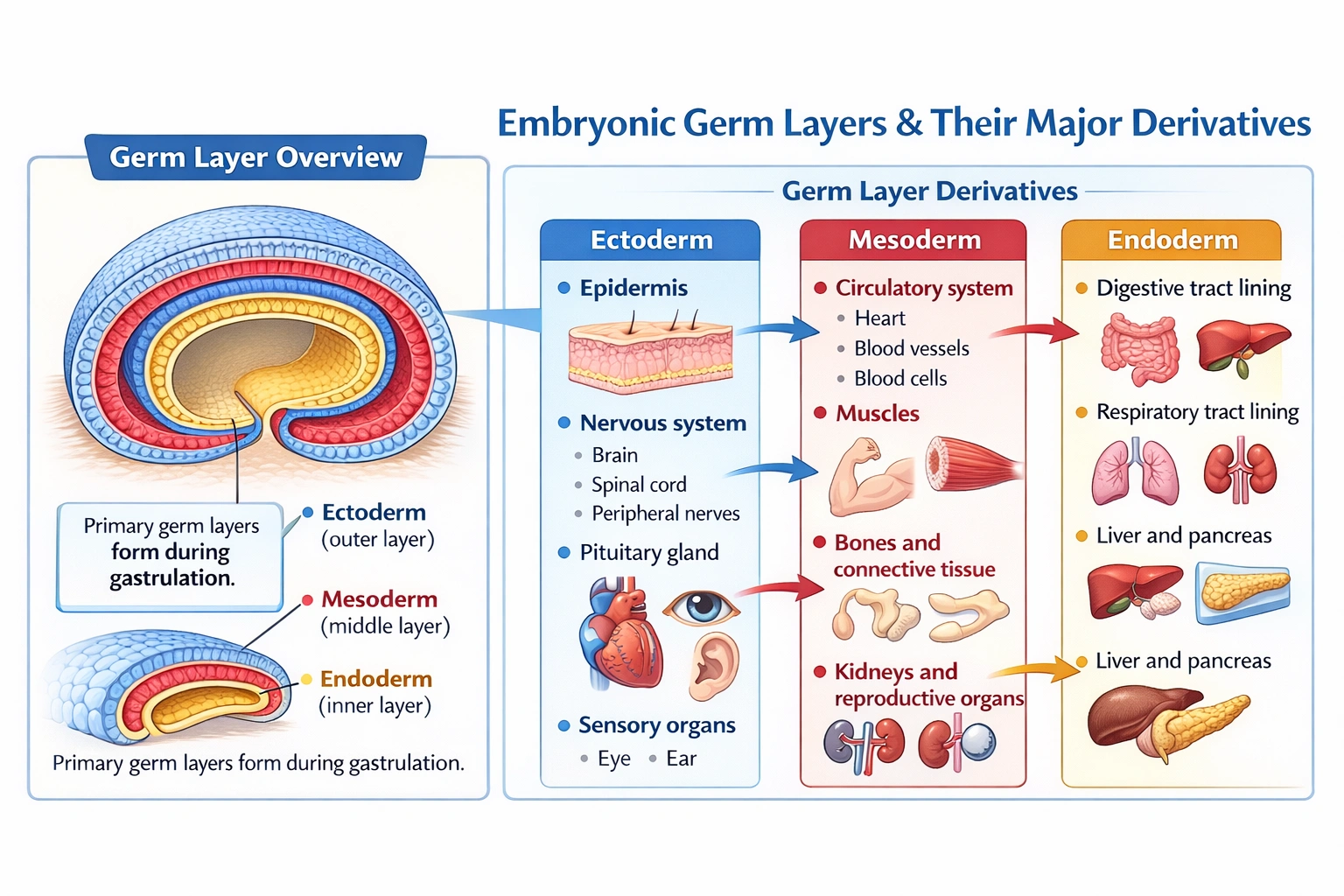
During embryonic/fetal development, all of the following are derived from the ectoderm EXCEPT the
A.
circulatory system
B. epidermis
C. nervous system
D. pituitary gland
Rationale
The circulatory system is not derived from the ectoderm.
It originates from the mesoderm. The other structures listedthe epidermis, nervous system, and pituitary gland are all classic derivatives of the ectodermal germ layer.
A) Circulatory system
The circulatory system, including the heart, blood vessels, and blood cells, is derived from the mesoderm, specifically the mesodermal layer that forms during gastrulation. The heart develops from the splanchnic lateral plate mesoderm, forming the primitive heart tube. Blood vessels develop from mesodermal cells called angioblasts, and blood cells originate from mesoderm-derived hemangioblasts. Therefore, the circulatory system is mesodermal in origin and is the correct exception in this list of otherwise ectodermal structures.
B) Epidermis
The epidermis, which is the outermost layer of the skin, along with its appendages (such as hair follicles, nails, sweat glands, and mammary glands), is unequivocally derived from the surface ectoderm. The ectoderm is the outermost of the three primary germ layers, and it gives rise to the entire external epithelial covering of the body.
C) Nervous system
The entire nervous system, including the brain, spinal cord (central nervous system), peripheral nerves, and sensory organs like the retina, originates from the neuroectoderm. The neuroectoderm is a specialized region of the ectoderm that thickens to form the neural plate. This plate folds to create the neural tube, which becomes the central nervous system. Additionally, neural crest cells, which are also ectodermal, migrate throughout the embryo to form many components of the peripheral nervous system, among other structures.
D) Pituitary gland
The pituitary gland, while having two distinct parts, is entirely ectodermal in origin. The anterior pituitary (adenohypophysis) develops from an upward outpouching of the oral ectoderm called Rathke's pouch. The posterior pituitary (neurohypophysis) develops from a downward extension of the neuroectoderm of the diencephalon (part of the developing brain). Thus, both components are derivatives of the ectoderm.
Conclusion:
In embryonic development, germ layers give rise to specific tissue lineages. The ectoderm forms the skin's epidermis and the nervous system, including specialized glands like the pituitary. The circulatory system, however, arises from the mesoderm, making it the correct exception.
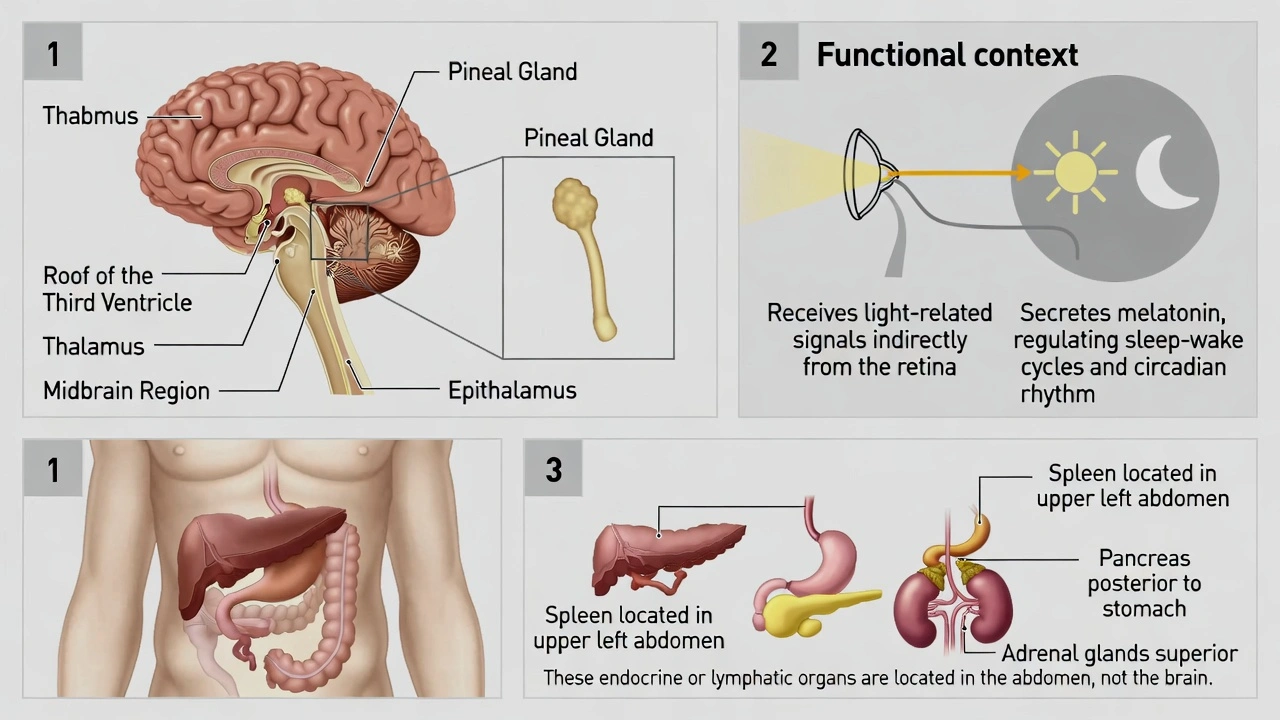
The pineal gland is located
A.
anterior to the spleen.
B. behind the stomach in the posterior abdomen.
C. in the midbrain on the roof of the third ventricle, attached to the thalamus.
D. on the upper surface of the kidneys.
Rationale
The pineal gland is located in the midbrain on the roof of the third ventricle, attached to the thalamus.
The pineal gland is a small, pinecone‑shaped endocrine gland found in the brain. Its precise anatomical position is within the epithalamus, situated posterior to the third cerebral ventricle and attached by a stalk to the habenular commissure and posterior commissure. This central location allows it to receive light‑related signals from the retina via a complex neural pathway, enabling its primary function of secreting melatonin to regulate circadian rhythms.
A. anterior to the spleen.
The spleen is an organ located in the upper left abdomen. A structure described as anterior to it would be positioned within the abdominal cavity, potentially near the stomach or colon. The pineal gland, however, is situated within the cranial cavity (the skull) and has no anatomical relationship with abdominal organs.
B. behind the stomach in the posterior abdomen.
This describes a retroperitoneal abdominal location, such as the position of the pancreas, abdominal aorta, and parts of the kidneys. These are all structures found in the abdominal cavity, which is separated from the brain by the diaphragm and thoracic cavity. The pineal gland is a structure of the central nervous system and is not located in the abdomen.
C. in the midbrain on the roof of the third ventricle, attached to the thalamus.
This is the accurate neuroanatomical description. The pineal gland is a midline structure that develops from the diencephalon. It sits posterior to the third ventricle and is attached to the epithalamus, which is part of the thalamus. This location is essential for its role in circadian rhythm regulation.
D. on the upper surface of the kidneys.
This location is specific to the adrenal (suprarenal) glands. These are separate endocrine organs that sit atop each kidney and are responsible for hormones like cortisol and adrenaline. The pineal gland is located in the brain, making this description incorrect.
Conclusion:
The pineal gland is a neuroendocrine organ found exclusively within the brain. Its correct location is in the epithalamus, on the roof of the third ventricle. The other options describe the positions of various abdominal organs the spleen, pancreas, and adrenal glands which are not related to the pineal gland's intracranial placement.
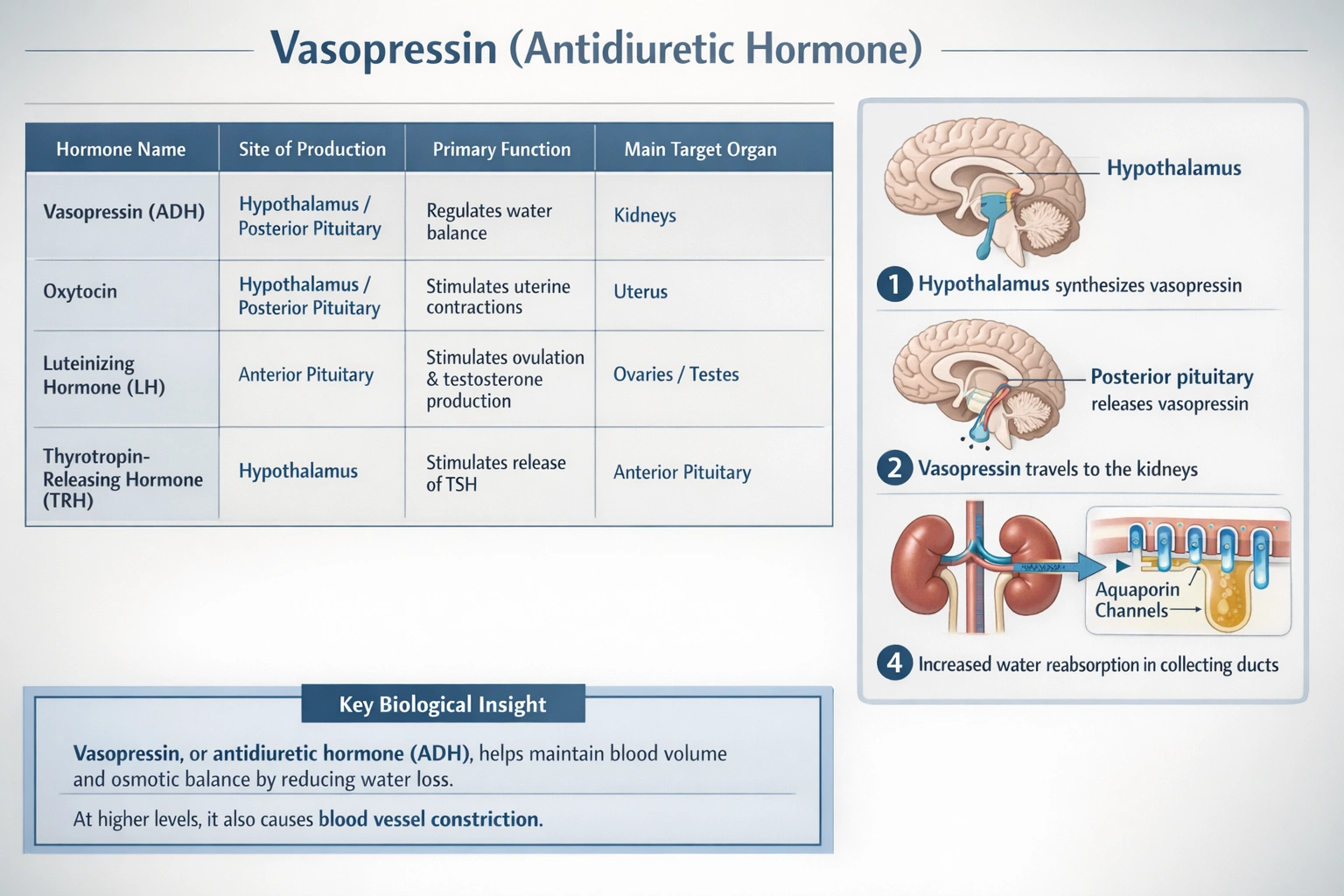
Vasopressin is also known as the
A.
antidiuretic hormone (ADH)
B. corticotrophin-releasing hormone (TRH)
C. luteinizing hormone (LH)
D. mammary hormone (MH)
Rationale
Vasopressin is also universally known as antidiuretic hormone (ADH).
It is a peptide hormone produced in the hypothalamus, stored in the posterior pituitary gland, and released into the bloodstream. Its primary function is to regulate the body's water balance by increasing water reabsorption in the kidneys, thereby concentrating urine and reducing urine output (an antidiuretic effect). The name "vasopressin" refers to its secondary, higher-dose effect of increasing blood pressure through the constriction of blood vessels.
A. antidiuretic hormone (ADH)
Vasopressin and antidiuretic hormone are two names for the same peptide hormone. It is synthesized in the hypothalamus, stored in the posterior pituitary gland, and released into the bloodstream. Its primary physiological role is to regulate water balance by acting on the kidneys. It binds to V2 receptors in the collecting ducts, triggering the insertion of aquaporin water channels into cell membranes. This greatly increases water reabsorption from the urine back into the bloodstream, thereby concentrating urine, reducing urine volume, and preserving body fluid. The name "antidiuretic" directly describes this function preventing excessive water loss. The name "vasopressin" refers to its secondary, pharmacological effect of raising blood pressure through vasoconstriction at higher concentrations.
B. corticotrophin-releasing hormone (TRH)
This option contains a compound error. The correct abbreviation for the hormone that stimulates adrenocorticotropic hormone (ACTH) release is CRH (corticotropin-releasing hormone). TRH stands for thyrotropin-releasing hormone, which stimulates thyroid-stimulating hormone (TSH) secretion. Both are distinct hypothalamic releasing hormones. While vasopressin can weakly potentiate CRH's action on ACTH release under stress, it is not synonymous with either CRH or TRH. These are separate hormones with different primary structures, primary functions, and target cells.
C. luteinizing hormone (LH)
Luteinizing hormone is a glycoprotein gonadotropin secreted by the anterior pituitary gland. It plays a central role in the reproductive system: in females, it triggers ovulation and promotes progesterone production; in males, it stimulates testosterone synthesis in Leydig cells of the testes. LH is chemically distinct from vasopressin it is a large glycoprotein, whereas vasopressin is a small peptide. Their sites of production, target tissues, and fundamental physiological roles (reproduction vs. fluid homeostasis) are completely different.
D. mammary hormone (MH)
There is no standard hormone in human endocrinology abbreviated as "MH." The primary hormones governing mammary gland function are prolactin (which promotes milk production) and oxytocin (which stimulates milk ejection). Oxytocin, like vasopressin, is produced in the hypothalamus and stored in the posterior pituitary, but it has a different structure and function. The term "mammary hormone" is non-scientific and misleading; it does not represent a synonym for vasopressin.
Conclusion:
The hormone vasopressin is identically known as antidiuretic hormone (ADH), a key regulator of renal water conservation. The other options refer to unrelated hormones involved in stress regulation (CRH), reproduction (LH), or lactation (prolactin/oxytocin), or constitute a non-standard term.
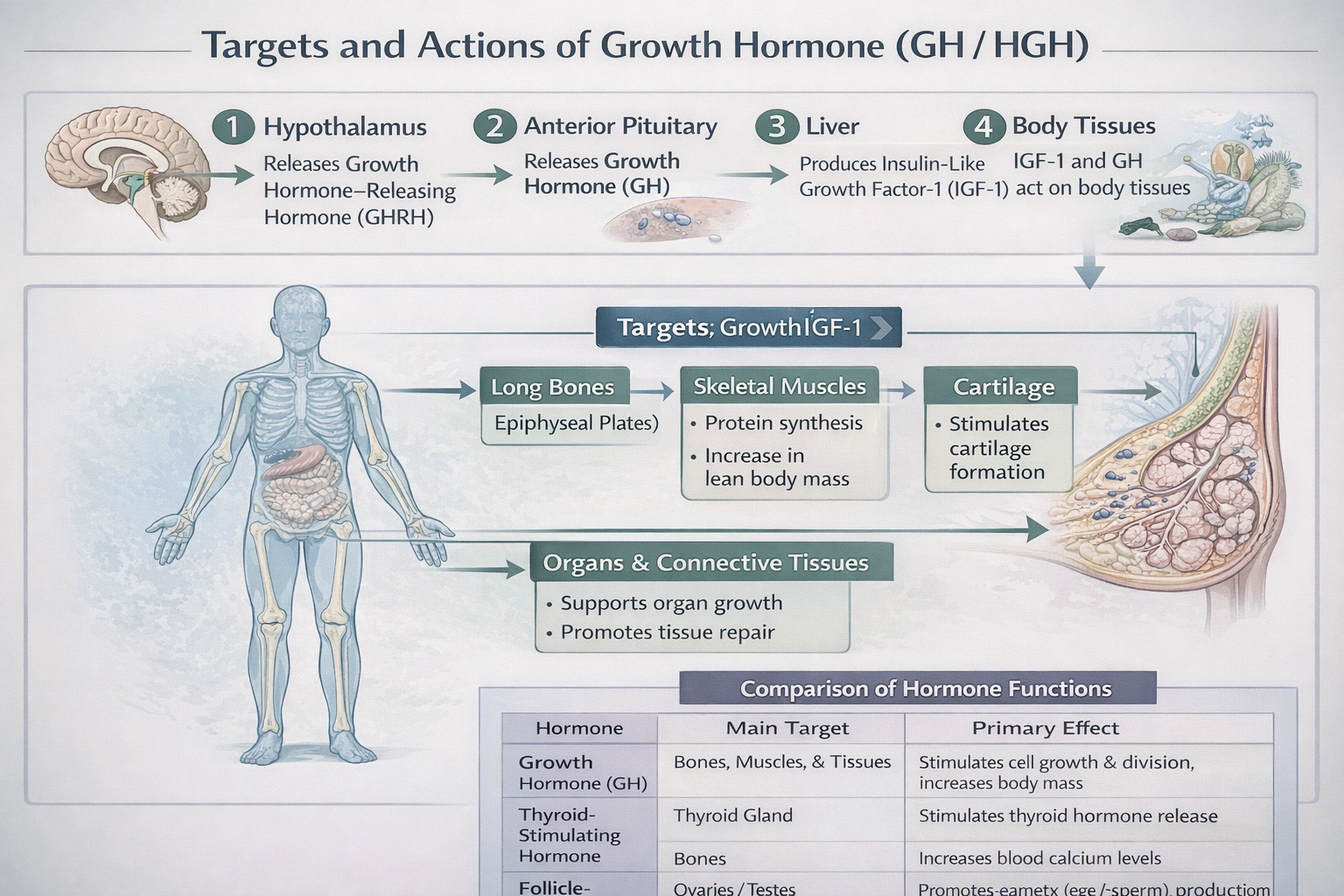
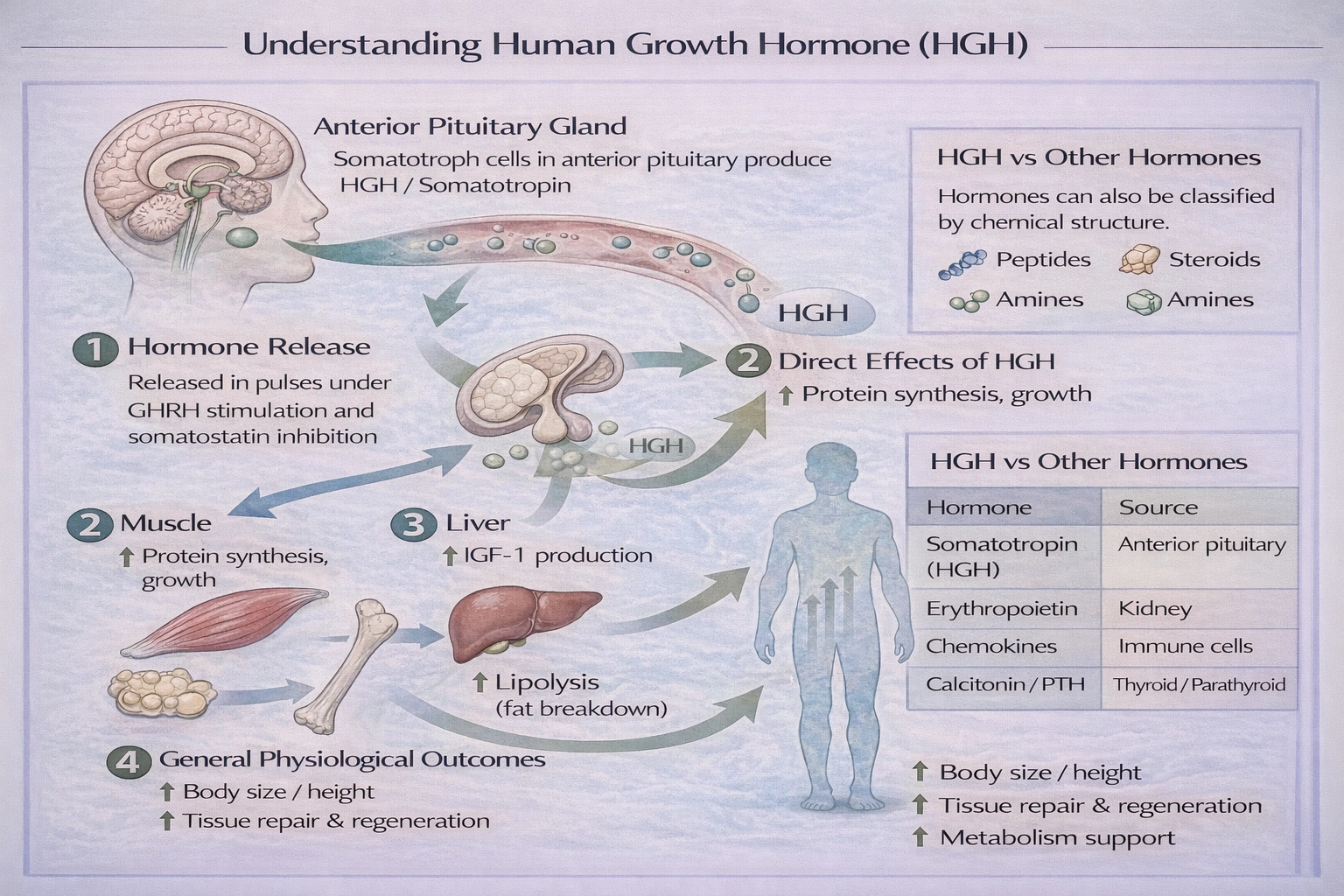
The growth hormone (HGH) targets/stimulates
A.
endocrine hormones.
B. mineral balance.
C. spermatogenesis.
D. tissues, bones, and muscles of the body.
Rationale
Growth Hormone (HGH), or somatotropin, targets and stimulates the tissues, bones, and muscles of the body.
It is a peptide hormone secreted by somatotroph cells in the anterior pituitary that exerts widespread anabolic and metabolic effects, promoting longitudinal growth in children and regulating body composition and tissue repair in adults. Its actions are mediated both directly and indirectly via the induction of Insulin-like Growth Factor-1 (IGF-1) primarily from the liver.
A. endocrine hormones.
While GH influences other endocrine axes it can modestly stimulate insulin secretion, enhance adrenal androgen production, and affect thyroid function these are secondary or permissive effects. GH is not a classic tropic hormone; it does not primarily target other endocrine glands for the sole purpose of regulating their secretion. True endocrine-regulating hormones are the pituitary tropic hormones: TSH (thyroid), ACTH (adrenal cortex), FSH/LH (gonads).
B. mineral balance.
Mineral homeostasis, particularly of calcium and phosphate, is precisely regulated by parathyroid hormone (PTH), calcitonin, and calcitriol (active vitamin D). GH and IGF-1 promote the active transport of calcium and phosphate into the bone matrix, contributing to bone growth and density. However, this is a slow, anabolic process of accretion, not the minute-to-minute homeostatic balance of serum ion concentrations, which is the domain of the parathyroid-renal axis.
C. spermatogenesis.
Spermatogenesis is critically dependent on Follicle-Stimulating Hormone (FSH), which acts on Sertoli cells, and on high intratesticular testosterone concentrations, maintained by Luteinizing Hormone (LH). GH may have supportive roles in testicular development and function, and GH deficiency can delay puberty, but it is not a direct mitogen or differentiating factor for germ cells. Isolated GH deficiency does not cause infertility, whereas deficiencies in FSH or LH do.
D. tissues, bones, and muscles of the body.
GH has direct effects on many tissues, promoting lipolysis in adipose tissue and gluconeogenesis in the liver. Its most prominent growth-promoting effects are often mediated by IGF-1. In bone, it stimulates chondrocyte proliferation in the epiphyseal plates, causing longitudinal growth. In muscle, it promotes amino acid uptake and protein synthesis, increasing lean mass. This systemic anabolic action on somatic tissues is its defining physiological role.
Conclusion:
Growth Hormone is a systemic anabolic agent whose primary targets are the body's somatic tissues specifically bone, cartilage, muscle, and organs. Its role is to promote overall growth during development and maintain tissue integrity in adulthood. The other options describe more specialized functions of other hormonal systems.
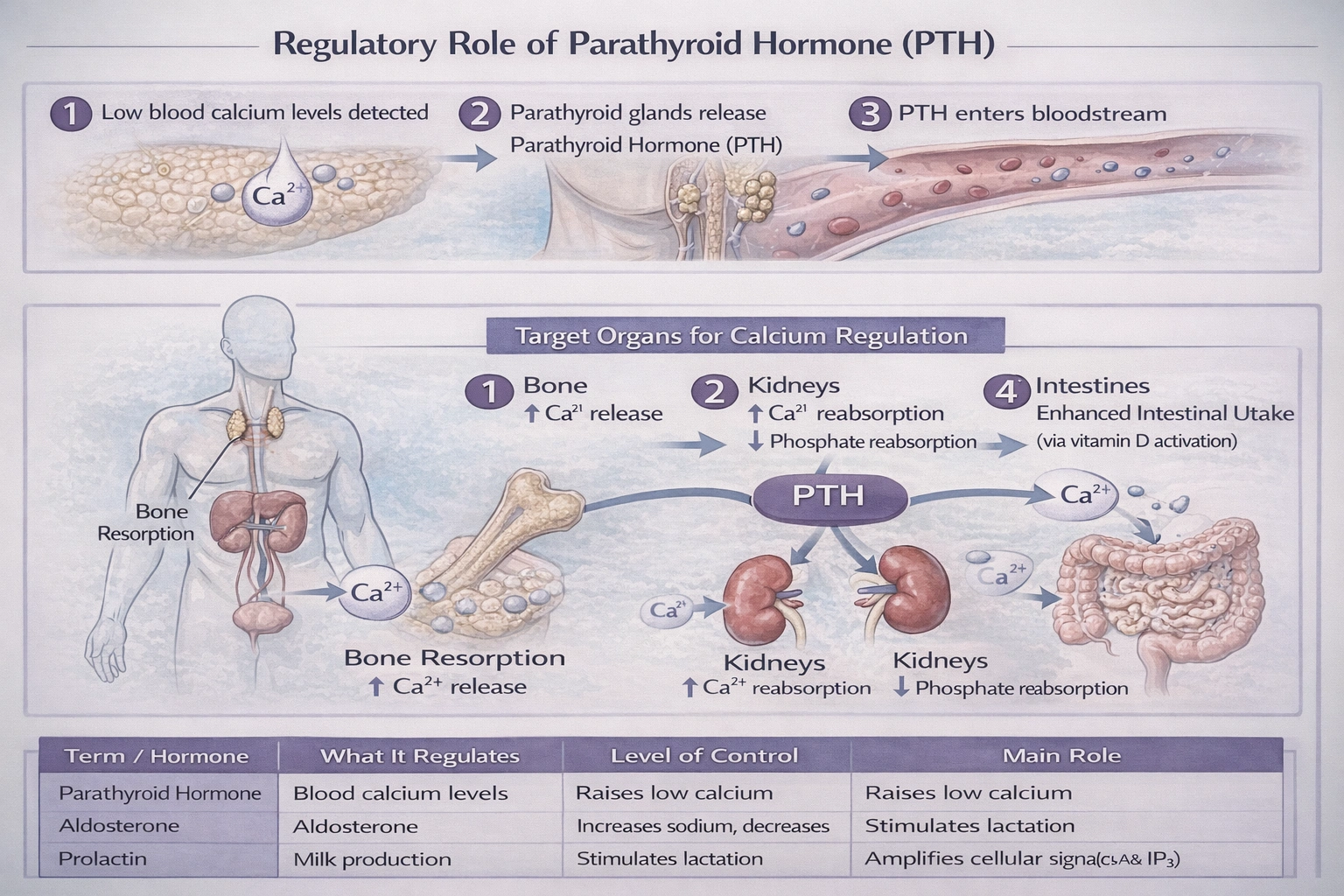
The parathyroid hormone regulates
A.
calcium.
B. mineral balance.
C. prolactin.
D. phospholipase C.
Rationale
Parathyroid hormone (PTH) is the primary regulator of blood calcium levels. Produced by the parathyroid glands, PTH acts directly on bone and kidneys, and indirectly on the intestines, to increase serum calcium concentration. This precise regulation is critical for neuromuscular function, bone integrity, and cellular signaling.
A. calcium.
PTH directly targets calcium homeostasis. In response to low blood calcium, PTH stimulates osteoclast activity to release calcium from bone, increases renal reabsorption of calcium in the distal tubules, and enhances intestinal calcium absorption by activating vitamin D. Its entire physiological purpose is to elevate and maintain ionized calcium levels in the blood.
B. mineral balance.
While PTH significantly affects calcium and phosphate balance, the term "mineral balance" is too broad. Other minerals like sodium, potassium, and magnesium are regulated by separate systems (e.g., aldosterone for sodium). PTH's role is specific to calcium-phosphate homeostasis, not the overall mineral economy of the body.
C. prolactin.
Prolactin secretion from the anterior pituitary is regulated by hypothalamic dopamine (inhibitory) and thyrotropin-releasing hormone (stimulatory). There is no physiological link between PTH and prolactin regulation. These hormones belong to entirely separate systems calcium metabolism versus lactation and reproduction.
D. phospholipase C.
Phospholipase C is an intracellular enzyme involved in the signal transduction pathway of PTH and many other hormones. When PTH binds its receptor, it activates phospholipase C to generate second messengers. However, PTH does not regulate this enzyme as an endpoint; enzyme activation is a mechanistic step in achieving its physiological goal of raising calcium levels.
Conclusion:
Parathyroid hormone's principal and specific function is the tight regulation of blood calcium concentration. Its actions on bone, kidney, and intestine are all coordinated toward this single end, distinguishing it from hormones that regulate broader mineral balance or unrelated physiological processes.
Most hormone manufacturing is regulated by the
A.
antidiuretic hormone (ADH).
B. negative feedback mechanisms.
C. parasympathetic nervous system.
D. response modifiers.
Rationale
The synthesis and secretion of most hormones in the endocrine system are primarily regulated by negative feedback mechanisms.
This fundamental physiological principle maintains homeostasis by ensuring that once a hormone reaches its desired level or effect, it suppresses further stimulation of its own production, preventing over-secretion and keeping bodily processes in balance.
A. antidiuretic hormone (ADH).
Antidiuretic hormone (ADH), also called vasopressin, is itself a hormone whose release is regulated by negative feedback involving blood osmolality and volume. ADH acts on the kidneys to conserve water, but it does not function as a master regulator governing the manufacture of other hormones. While it can modestly influence the release of ACTH in certain stress-related pathways, its role is specific to fluid and electrolyte balance, not the broad regulation of endocrine synthesis.
B. negative feedback mechanisms.
Negative feedback is the predominant and essential control system for hormone production. In this process, the end product or effect of a hormonal pathway inhibits earlier steps in the pathway. For example, high levels of thyroid hormones suppress the hypothalamus and pituitary, reducing secretion of TRH and TSH; elevated cortisol inhibits CRH and ACTH; and increased sex steroids inhibit GnRH and gonadotropins. This self-limiting loop maintains stable hormone concentrations within a narrow, optimal range and is the cornerstone of endocrine regulation.
C. parasympathetic nervous system.
The parasympathetic nervous system is involved in "rest and digest" functions and exerts direct neural control over some glandular secretions, such as stimulating salivary and gastric glands. However, the majority of hormone-producing endocrine glands (like the thyroid, adrenal cortex, gonads, and endocrine pancreas) are not primarily regulated by parasympathetic input. Their baseline manufacturing and release are controlled by hormonal signals and feedback loops, not by cholinergic neural activity.
D. response modifiers.
"Response modifiers" is not a standard term in endocrine physiology. It could loosely refer to agents that alter tissue sensitivity to a hormone (e.g., receptor up-regulation or down-regulation), but these do not constitute the primary regulatory mechanism for hormone synthesis. The rate of hormone manufacturing is governed by well-defined feedback systems, not by a broad class of modifiers a term more commonly associated with pharmacological interventions than with innate physiological control.
Conclusion:
Negative feedback inhibition is the central, ubiquitous principle regulating the production and release of most hormones in the body. This mechanism ensures stability and prevents extremes in hormone levels, distinguishing it from specific hormones like ADH, neural control systems, or non-specific modifiers.
Human growth hormone is also known as
A.
chemiosin
B. erythropoietin
C. osteoclastin
D. somatotropin
Rationale
Human growth hormone is also known as somatotropin.
This peptide hormone, produced by somatotroph cells in the anterior pituitary gland, is the primary endocrine regulator of postnatal growth, protein synthesis, and cellular regeneration. The name "somatotropin" derives from Greek roots: "soma" (body) and "trope" (turning or influencing), literally meaning "body influencer," which reflects its broad anabolic effects on tissues.
A. chemiosin
"Chemiosin" is not a recognized hormone in human physiology. It may be confused with "chemokines," which are a large family of small cytokines secreted by cells to induce chemotaxis in nearby responsive cells, primarily in immune and inflammatory processes. Chemokines are local signaling molecules, not circulating hormones, and they play no role in systemic growth regulation.
B. erythropoietin
Erythropoietin (EPO) is a glycoprotein hormone produced predominantly by peritubular interstitial cells in the kidney in response to hypoxia. Its exclusive function is to stimulate the bone marrow to increase the production of red blood cells (erythropoiesis). While vital for oxygen transport, EPO does not promote general somatic growth and is not an alternative name for growth hormone.
C. osteoclastin
There is no hormone named "osteoclastin." Bone remodeling involves osteoclasts (bone-resorbing cells) whose differentiation and activity are regulated by systemic hormones like parathyroid hormone (PTH) and calcitonin, and by local cytokines such as RANK ligand (RANKL). The term is a non-standard construct and does not refer to a genuine growth-promoting hormone.
D. somatotropin
Somatotropin is the precise scientific synonym for human growth hormone. It is a 191-amino acid polypeptide secreted in a pulsatile manner, largely under the control of hypothalamic growth hormone-releasing hormone (GHRH) and inhibited by somatostatin. Its growth-promoting actions are mediated both directly and indirectly via insulin-like growth factor 1 (IGF-1). The name is consistently used in endocrinology textbooks and clinical literature.
Conclusion:
Somatotropin is the correct alternative name for human growth hormone, a designation that highlights its role in stimulating overall body growth. This distinguishes it from hormones involved in hematopoiesis, immune cell trafficking, or bone-specific regulation.
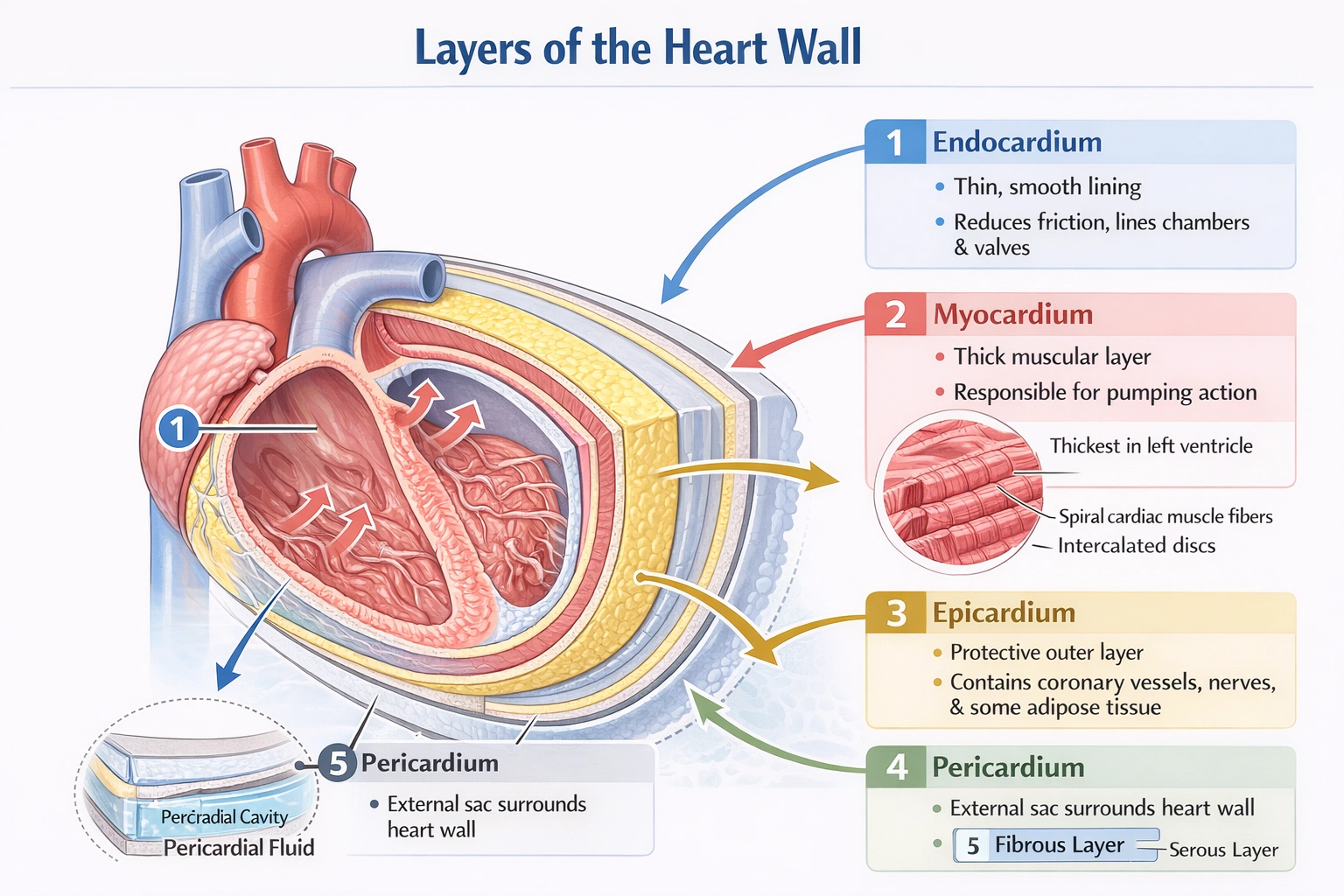
The middle layer of the heart wall is the
A.
endocardium
B. epicardium
C. pericardium
D. myocardium
Rationale
The middle layer of the heart wall is the myocardium.
This thick, muscular layer is composed of cardiac muscle tissue (cardiomyocytes) and is responsible for the heart's pumping action. It lies between the inner endocardial lining and the outer epicardial layer, and its thickness varies according to the chamber, being most substantial in the left ventricle due to the higher pressure needed to pump blood into the systemic circulation.
A. endocardium
The endocardium is the innermost layer of the heart wall, consisting of a thin, smooth endothelium and underlying connective tissue. It lines the atria, ventricles, and covers the heart valves, providing a friction‑reducing surface for blood flow and housing parts of the conduction system (e.g., Purkinje fibers). While crucial for internal integrity and electrical conduction, it is not the muscular middle layer.
B. epicardium
The epicardium, also called the visceral layer of the serous pericardium, forms the outermost layer of the heart wall. It is a thin serous membrane of mesothelial cells and connective tissue that adheres directly to the myocardium. It often contains adipose tissue, coronary blood vessels, and nerves, serving as a protective outer coating and a site for vascular supply. It is external to the myocardium, not the middle layer.
C. pericardium
The pericardium is not a layer of the heart wall itself. It is a double‑walled sac that encloses the entire heart and proximal great vessels. Its outer fibrous pericardium anchors the heart in the mediastinum, and its inner serous pericardium (parietal and visceral layers) produces pericardial fluid that reduces friction during cardiac contraction. The pericardium surrounds the heart but is separate from the three layers (endocardium, myocardium, epicardium) that constitute the heart wall proper.
D. myocardium
The myocardium is the thick, contractile middle layer of the heart wall, composed of specialized cardiac muscle cells arranged in spiral bundles. These cells are interconnected by intercalated discs that allow rapid electrical and mechanical coupling. The myocardium generates the force of contraction, ejecting blood from the chambers. Its thickness correlates with workload: the left ventricular myocardium is the thickest, followed by the right ventricle, then the atria.
Conclusion:
The heart wall is anatomically organized into three distinct layers: the inner endocardium, the middle myocardium (the muscular pump), and the outer epicardium. The pericardium is an external protective sac. Only the myocardium occupies the central, muscular portion of the wall, making it the correct answer.
Select the true statement.
A.
↑Cardiac output = ↑blood pressure.
B. ↑Cardiac output =↓blood pressure.
C. ↓Cardiac output = ↑blood pressure.
D. The relationship between cardiac output and blood pressure depends upon individual physiology and is not a constant or predictable factor.
Rationale
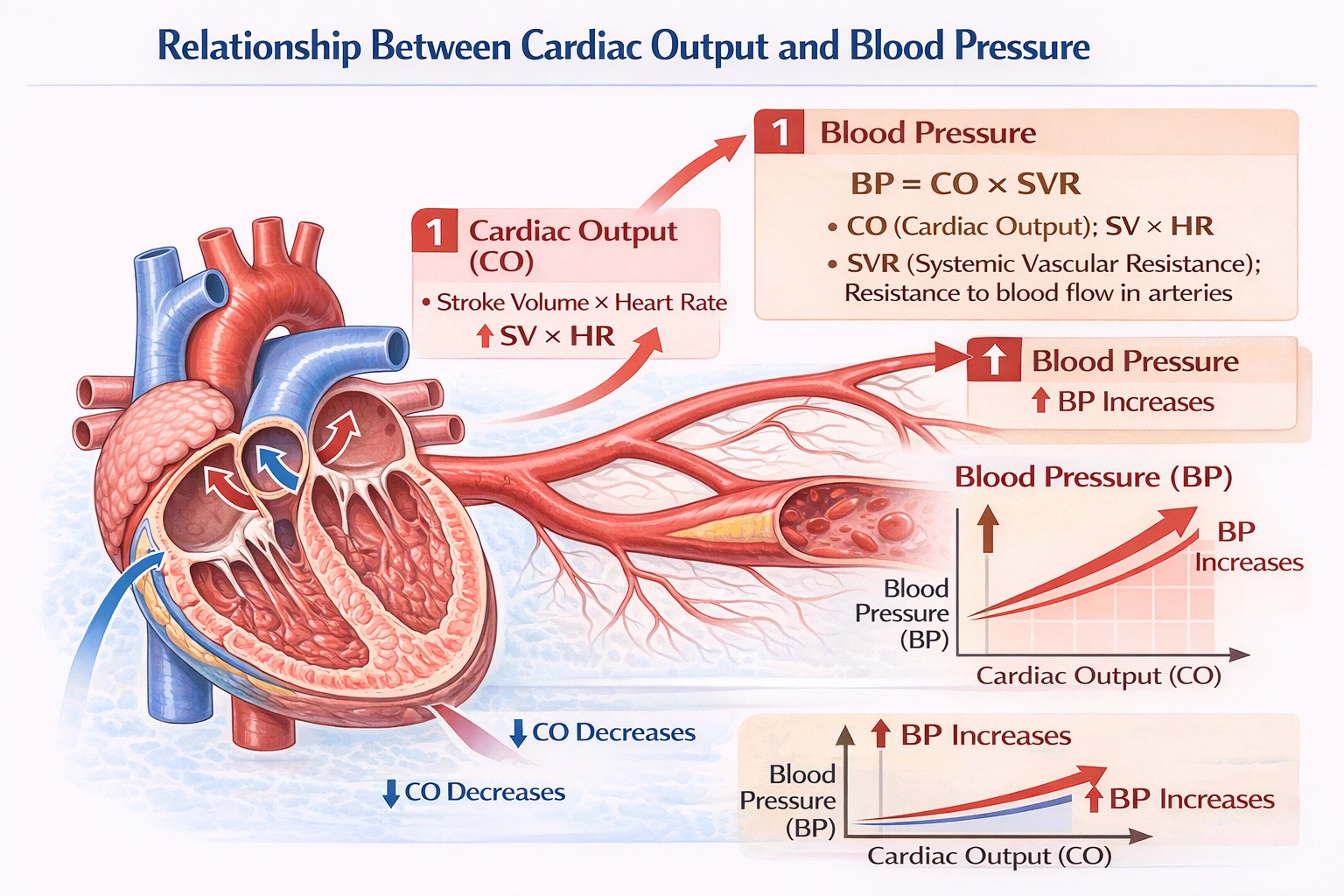
The true statement is: ↑Cardiac output = ↑blood pressure.
The relationship between cardiac output (CO) and blood pressure (BP) is defined by the fundamental hemodynamic equation:
BP = CO × Systemic Vascular Resistance (SVR).
Assuming systemic vascular resistance remains constant, an increase in cardiac output directly increases arterial blood pressure. This direct, proportional relationship is a cornerstone of cardiovascular physiology and is consistently observed in both normal regulation (e.g., during exercise) and clinical conditions.
A. ↑Cardiac output = ↑blood pressure.
This statement is correct and reflects a core principle of cardiovascular physiology. Cardiac output is the product of heart rate and stroke volume. When the heart pumps more blood into the arterial tree each minute, and if the arterioles do not dilate to compensate, the pressure within those arteries must increase. This relationship is consistently observed in physiological regulation and clinical scenarios, such as during exercise when increased cardiac output contributes to a rise in systolic blood pressure.
B. ↑Cardiac output =↓blood pressure.
This inverse relationship is not physiologically accurate under normal, steady-state conditions. A primary increase in cardiac output, without a concurrent and dominant decrease in systemic vascular resistance, will raise blood pressure. Situations where increased output coincides with falling pressure (e.g., distributive shock like sepsis) occur because peripheral vasodilation drastically lowers resistance a separate, overriding variable not because of a direct inverse link between output and pressure.
C.↓Cardiac output = ↑blood pressure.
This is generally false. A decrease in cardiac output (e.g., due to hemorrhage, heart failure, or bradycardia) typically leads to a fall in arterial pressure if vascular resistance remains unchanged. The body may attempt to compensate by increasing systemic vascular resistance via sympathetic activation, but this is a compensatory response to the drop in output, not a direct consequence of it. The initial direct effect of reduced cardiac output is hypotension.
D. The relationship between cardiac output and blood pressure depends upon individual physiology and is not a constant or predictable factor.
While individual variations (such as baseline vascular tone, blood volume, and autonomic responsiveness) influence the magnitude of the blood pressure change, the fundamental physical relationship between flow, resistance, and pressure is constant and predictable. The equation BP = CO × SVR is a deterministic model that reliably describes this interaction. Clinicians use this relationship to predict and manage hemodynamic states; it is not an unpredictable variable.
Conclusion:
Cardiac output and blood pressure are directly proportional when systemic vascular resistance is held constant. This predictable relationship is a cornerstone of cardiovascular physiology and clinical hemodynamic management.
In regulating heart function, the sympathetic nervous system releases __________ when stimulated.
A.
baroreceptors
B. chyme
C. enzymes
D. norepinephrine
Rationale
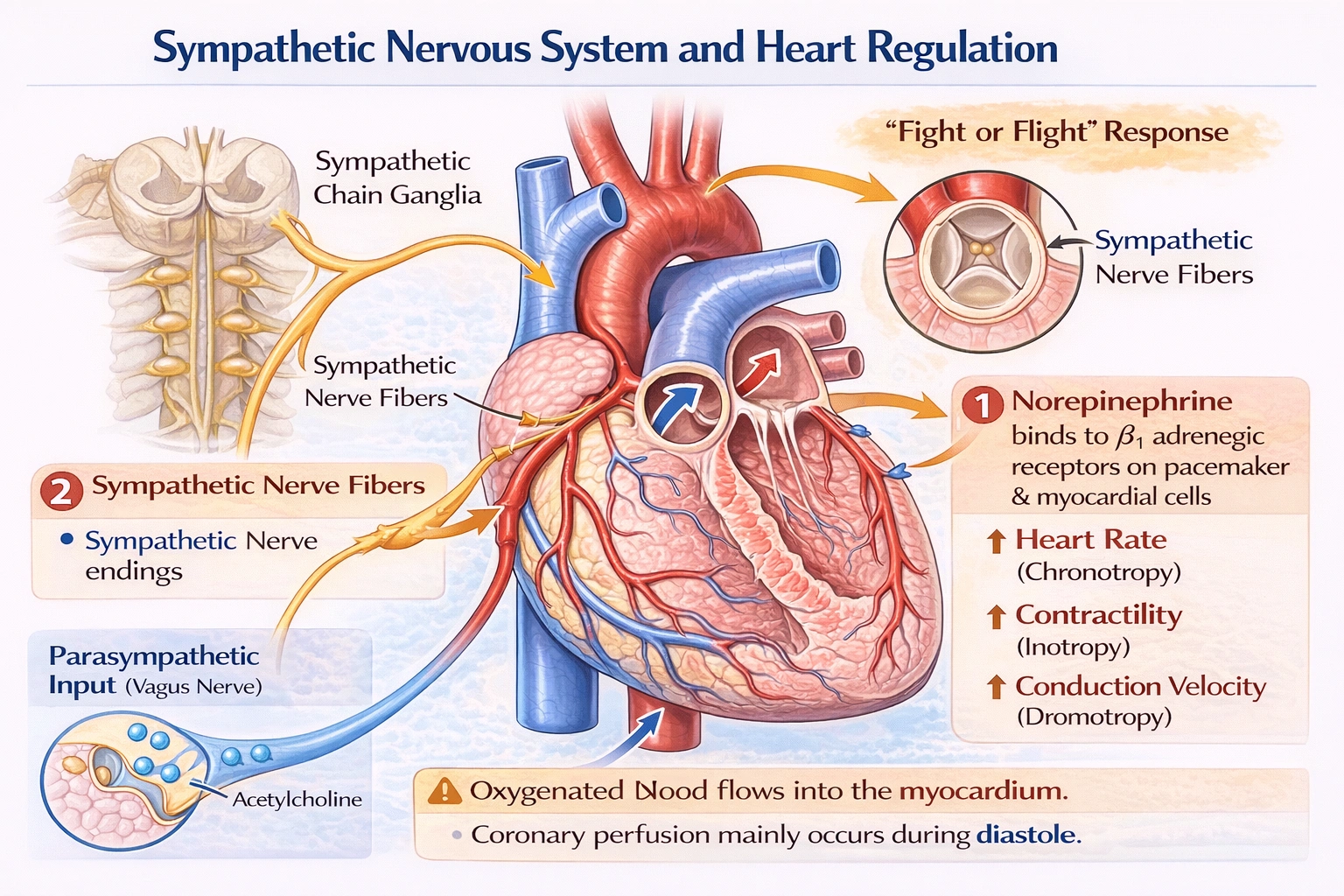
In regulating heart function, the sympathetic nervous system releases norepinephrine when stimulated.
The sympathetic nervous system increases heart rate, contractility, and conduction velocity as part of the "fight‑or‑flight" response. This is achieved through the release of the neurotransmitter norepinephrine from sympathetic nerve endings that innervate the heart. Norepinephrine binds primarily to beta‑1 adrenergic receptors on cardiac cells, leading to enhanced automaticity, faster conduction through the atrioventricular node, and stronger myocardial contractions.
A. baroreceptors
Baroreceptors are sensory nerve endings located in the carotid sinuses and aortic arch. They are mechanoreceptors that detect changes in arterial pressure and relay this information to the brainstem. They are not substances released by sympathetic nerves; rather, they are part of the afferent limb of autonomic reflexes that can trigger sympathetic or parasympathetic responses.
B. chyme
Chyme is the semi-fluid, partially digested food mixture found in the stomach and duodenum. It is a product of gastrointestinal secretion and mechanical digestion, with no role in neural cardiac regulation.
C. enzymes
Enzymes are protein catalysts for biochemical reactions. While sympathetic activation can influence enzymatic activity inside cells (e.g., activating adenylate cyclase via G-proteins), enzymes themselves are not the signaling molecules released from sympathetic nerve terminals to act on the heart. The direct agent of synaptic transmission is norepinephrine.
D. norepinephrine
Norepinephrine is the primary neurotransmitter of the sympathetic postganglionic neurons innervating the heart. It is stored in synaptic vesicles and released upon neural stimulation. It binds predominantly to beta-1 adrenergic receptors on cardiac myocytes and pacemaker cells, leading to increased heart rate (positive chronotropy), increased contractile force (positive inotropy), and faster conduction through the atrioventricular node (positive dromotropy).
Conclusion:
The sympathetic nervous system directly increases cardiac performance through the targeted release of norepinephrine onto the heart. Other options refer to sensory receptors, digestive contents, or intracellular molecules that are not the primary sympathetic neurotransmitters.
___________ can be defined as a complex system of capillary-like vessels, ducts, nodes, and organs that assist in maintaining the fluid environment of the body.
A.
Active transport
B. Circulatory pathways
C. Hormonal regulation
D. Lymphatic system
Rationale
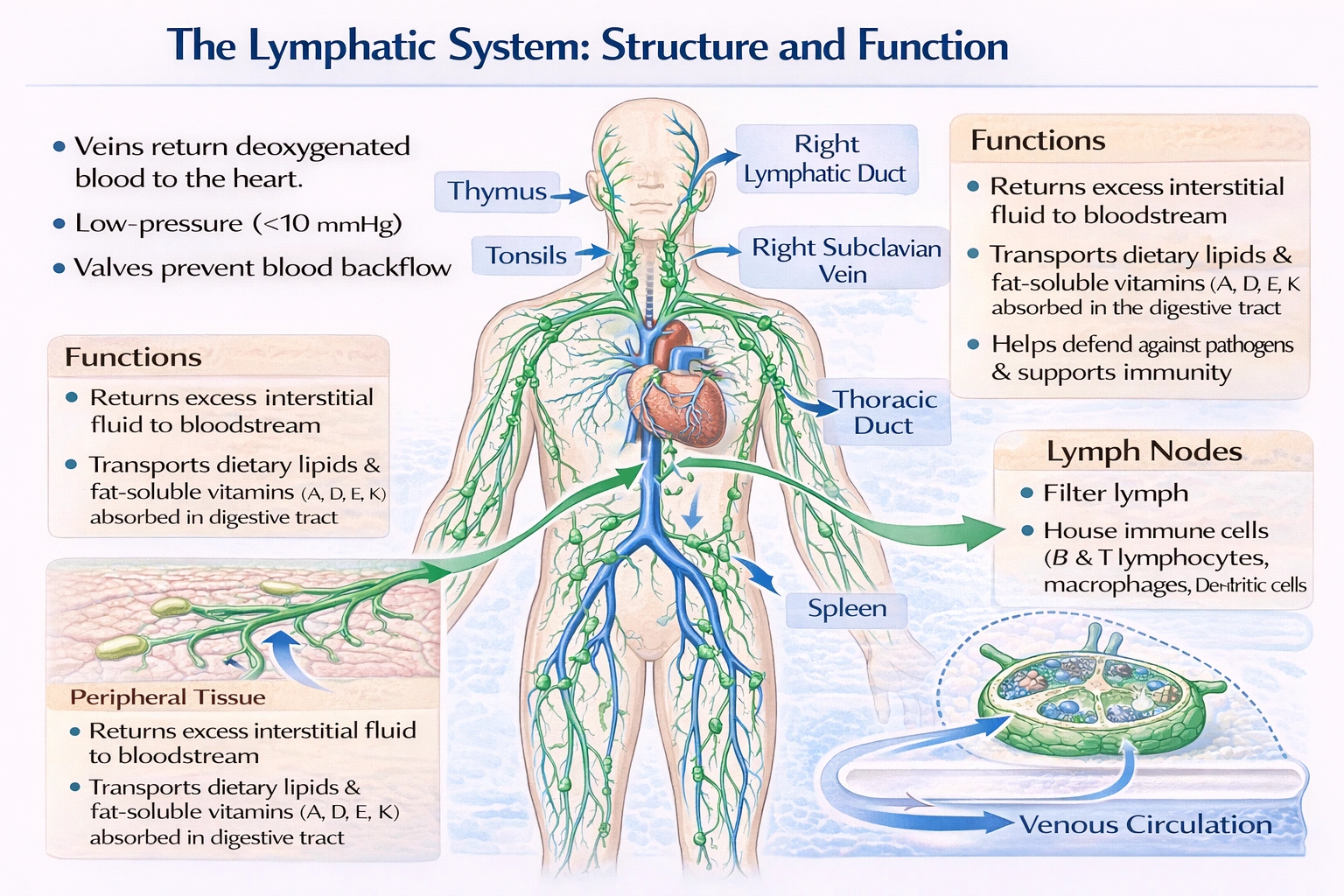
Lymphatic system can be defined as a complex system of capillary-like vessels, ducts, nodes, and organs that assist in maintaining the fluid environment of the body.
The lymphatic system is a network of delicate vessels (lymphatic capillaries), collecting ducts, lymph nodes, and lymphoid organs (such as the spleen, tonsils, and thymus). Its primary roles include returning excess interstitial fluid to the bloodstream, filtering lymph through nodes to remove pathogens and debris, transporting dietary lipids, and supporting immune surveillance all of which are essential for maintaining the body's fluid balance and internal environment.
A. Active transport
Active transport is a cellular-level process involving the movement of molecules across a membrane against their concentration gradient, requiring energy (ATP). It is not a macroscopic system of vessels and organs. While active transport occurs in some lymphatic endothelial cells, it is not the system itself.
B. Circulatory pathways
This is an ambiguous, non-technical term that could refer to routes of blood flow. The blood vascular system does include capillaries, but it does not centrally feature lymph nodes or have as its primary function the maintenance of the interstitial fluid environment in the way described. The description specifically matches the lymphatic system's structure and role.
C. Hormonal regulation
Hormonal regulation involves the endocrine system glands such as the pituitary, thyroid, and adrenals that secrete hormones into the blood to regulate various functions. While hormones like aldosterone and ADH do influence fluid balance, the endocrine system is not composed of vessels, ducts, and nodes that physically carry and filter fluid.
D. Lymphatic system
The lymphatic system begins with blind-ended lymphatic capillaries in peripheral tissues that absorb interstitial fluid (forming lymph). These capillaries merge into collecting vessels, which pass through lymph nodes organs that filter lymph and house immune cells. The lymph then travels via larger lymphatic trunks and ducts (e.g., the thoracic duct) to empty into the subclavian veins. This system directly maintains fluid balance, supports immunity, and facilitates fat absorption from the gut.
Conclusion:
The detailed description in the question stem capillary-like vessels, ducts, nodes, and a role in fluid homeostasis is a precise definition of the lymphatic system, distinguishing it from cellular transport mechanisms, the blood vasculature, or endocrine regulation.
The diastolic phase of the heart is when the
A.
atria and ventricles contract simultaneously.
B. atria contract and ventricles relax.
C. heart is resting and filling with blood.
D. ventricles contract and atria relax.
Rationale
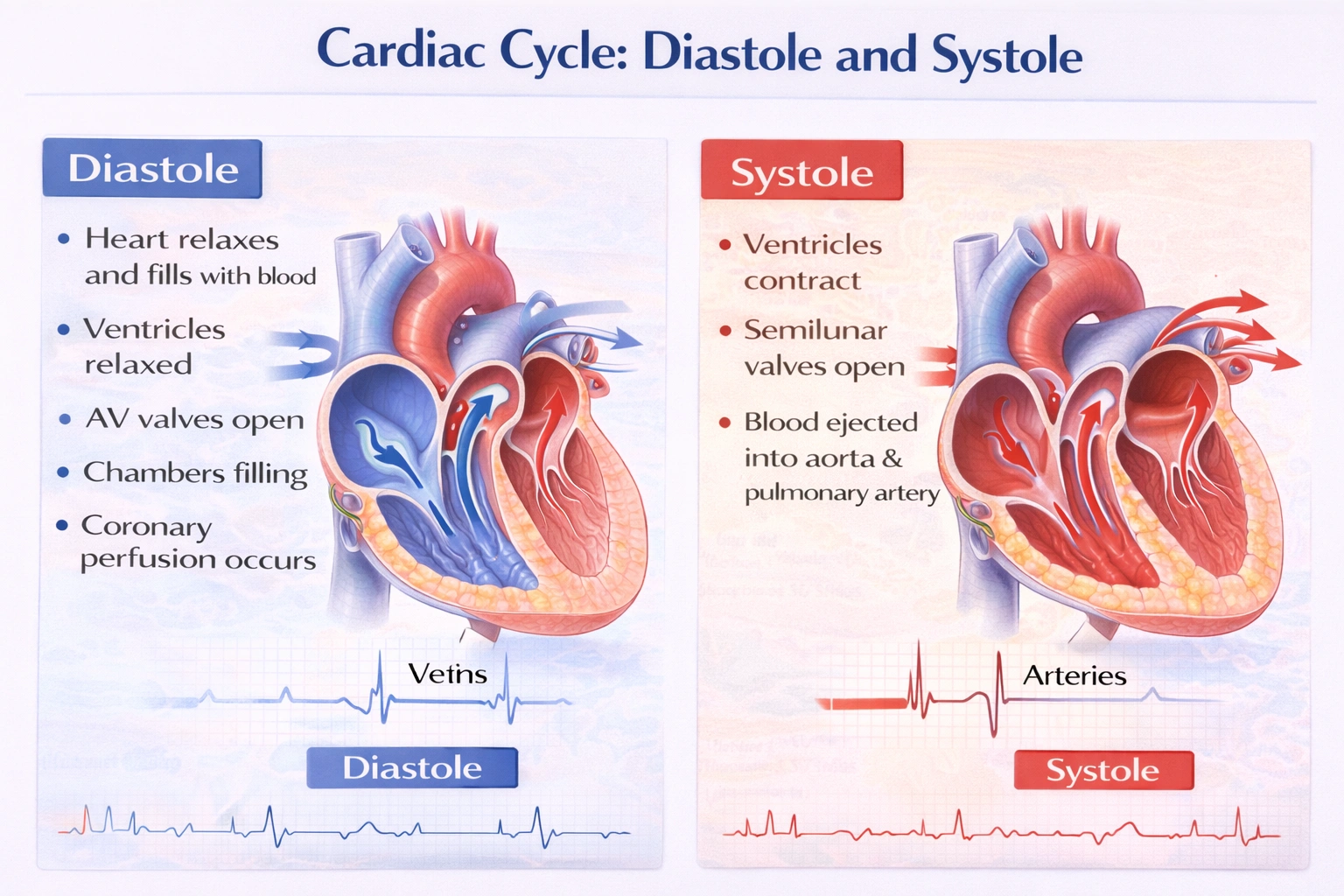
The diastolic phase of the heart is when the heart is resting and filling with blood.
Diastole represents the period of cardiac relaxation during which the chambers expand and receive blood returning from the veins, preparing for the next contraction.
A. atria and ventricles contract simultaneously.
Simultaneous contraction of all chambers does not occur in a normal cardiac cycle. The heart's conduction system ensures sequential contraction: the atria contract first (atrial systole), followed by a brief delay at the atrioventricular (AV) node, and then ventricular contraction (ventricular systole). This sequence is essential for efficient, unidirectional blood flow.
B. atria contract and ventricles relax.
This describes atrial systole, which occurs at the very end of diastole. During this brief event (sometimes called "atrial kick"), the atria contract to provide a final 20--30% boost of blood into the ventricles. While the ventricles are relaxed at this moment, this is only a component of the overall diastolic period, not its full definition.
C. heart is resting and filling with blood.
Diastole encompasses the period when the myocardium---especially the ventricles---is relaxed. The atrioventricular valves (tricuspid and mitral) are open, allowing passive filling from the atria. Coronary blood flow to the heart muscle is also greatest during diastole because the relaxed muscle no longer compresses the coronary arteries. This phase is crucial for cardiac recovery and preparation for the next systolic ejection.
D. ventricles contract and atria relax.
This defines ventricular systole, the active pumping phase. During systole, the ventricles contract to eject blood into the pulmonary artery and aorta, while the atria are relaxed and refilling. This is the opposite of diastole.
Conclusion:
Diastole is characterized by cardiac relaxation, chamber filling, and myocardial perfusion. While atrial systole contributes to late diastolic filling, the core of diastole is the period of rest and passive filling that occupies the majority of the cardiac cycle between ventricular contractions.
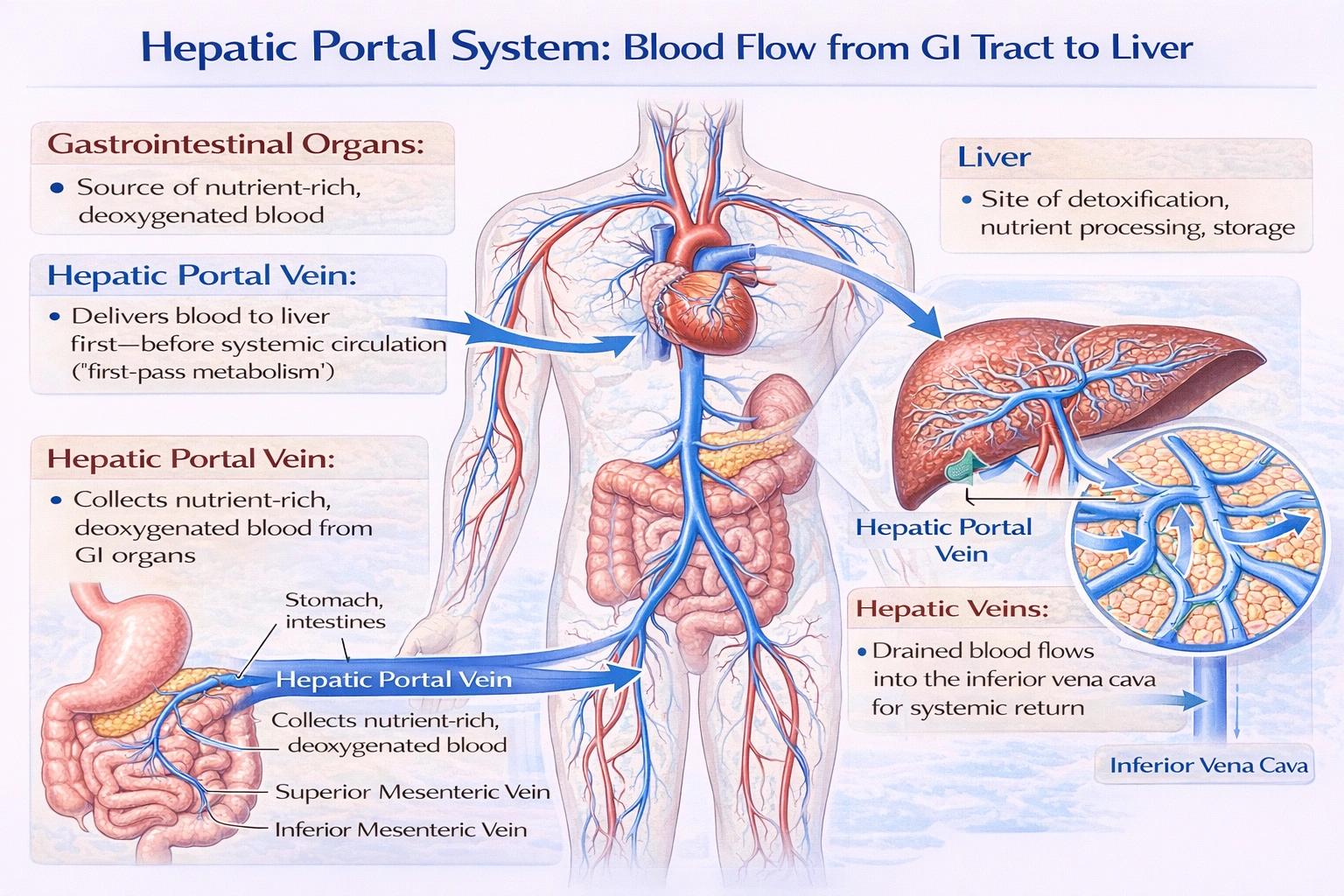
The hepatic portal system carries blood from the __________ to the __________.
A.
aorta, liver
B. gastrointestinal tract, liver
C. liver, heart
D. liver, inferior vena cava
Rationale
The hepatic portal system carries blood from the gastrointestinal tract to the liver.
The hepatic portal system is a specialized venous network that transports blood from the capillaries of the digestive organs including the stomach, intestines, pancreas, and spleen directly to the liver. This pathway allows the liver to process absorbed nutrients, metabolize drugs and toxins, and regulate blood composition before the blood returns to the general circulation.
A. aorta, liver
The aorta delivers oxygenated blood to the liver via the hepatic artery as part of the standard arterial supply. This is not the portal system; the portal system is a venous network carrying nutrient-rich but deoxygenated blood from the digestive organs to the liver.
B. gastrointestinal tract, liver
This is correct. The hepatic portal vein is formed by the union of the superior mesenteric vein (draining most of the small intestine and part of the colon) and the splenic vein (draining the spleen, pancreas, and parts of the stomach). It delivers blood directly to the liver sinusoids, enabling "first-pass" metabolism of nutrients, toxins, and other absorbed substances before they enter the systemic circulation.
C. liver, heart
Blood flows from the liver back to the heart via the hepatic veins, which empty into the inferior vena cava and then to the right atrium. This is the venous outflow from the liver, not the portal inflow.
D. liver, inferior vena cava
The hepatic veins carry blood from the liver to the inferior vena cava. This represents the post-hepatic venous drainage, which occurs after the liver has processed the blood received from the portal vein and hepatic artery.
Conclusion:
The hepatic portal system is uniquely defined as the venous route that carries blood from the gastrointestinal organs to the liver. This arrangement supports the liver's critical roles in metabolism, detoxification, and nutrient regulation before blood rejoins the systemic circulation. The other options either describe arterial supply to the liver or the venous outflow from it, not the portal system's specialized function.
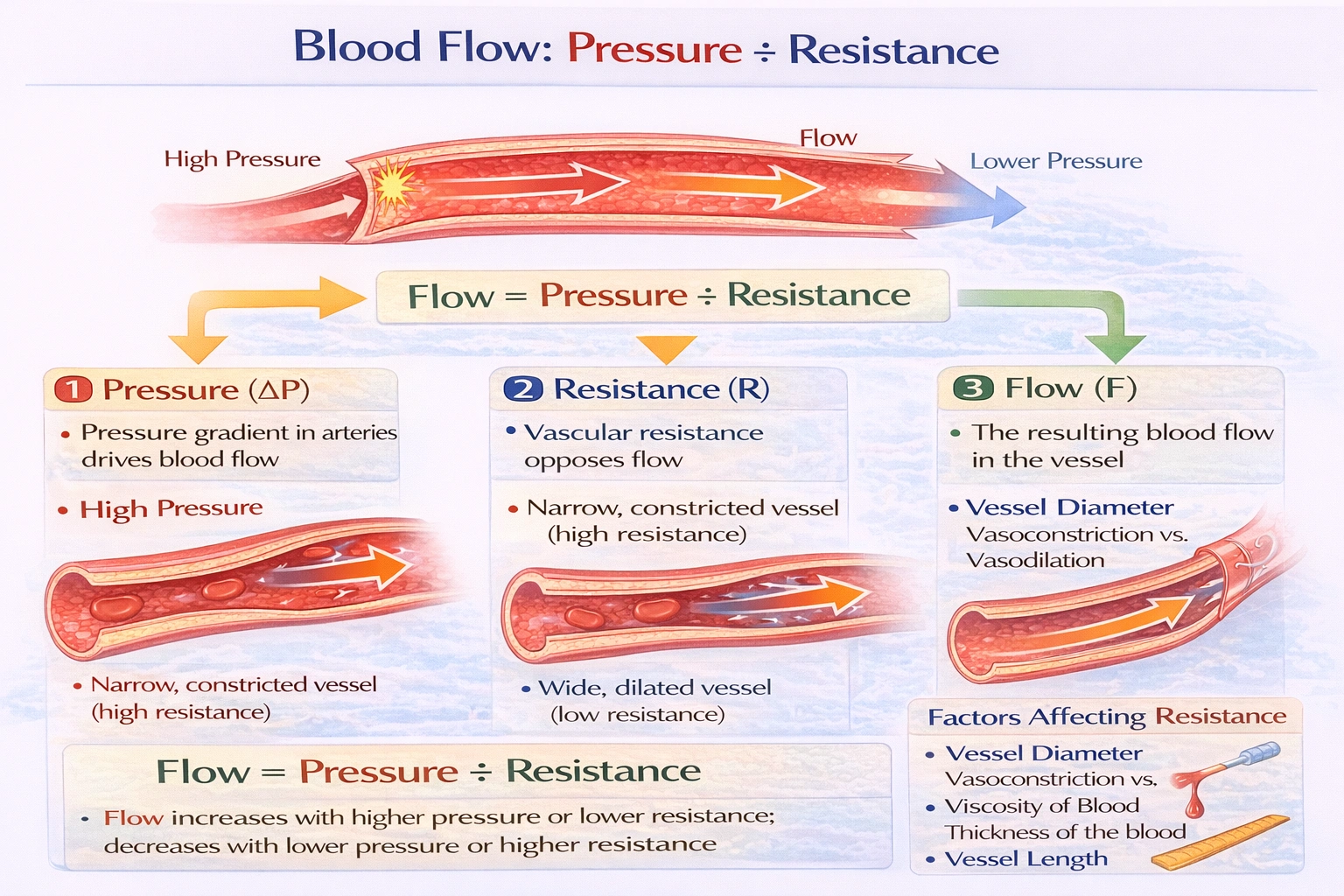
Blood flow equals
A.
pressure divided by resistance
B. pressure minus resistance
C. pressure plus resistance
D. resistance divided by pressure
Rationale
Blood flow equals pressure divided by resistance.
This relationship is derived from the fundamental hemodynamic principle analogous to Ohm's law in electrical circuits. Blood flow through any vascular segment is directly proportional to the pressure gradient (ÃŽâ€P) between two points and inversely proportional to the resistance (R) opposing that flow. The formula is expressed as: Flow = ÃŽâ€P / R.
In the circulatory system, increased pressure promotes flow, while increased resistance impedes it.
A. pressure divided by resistance
This is correct. The equation Flow = Pressure / Resistance defines the basic relationship in circulatory physiology. For example, if arterial pressure rises while resistance remains constant, blood flow increases. Conversely, if resistance increases (as in vasoconstriction), flow decreases unless pressure compensates.
B. pressure minus resistance
Pressure and resistance are not subtracted to determine flow. Resistance is a measure of hindrance to flow, not a value that is arithmetically deducted from pressure. This operation has no basis in hemodynamic principles.
C. pressure plus resistance
Adding resistance to pressure would incorrectly imply that greater resistance increases flow. In reality, resistance opposes flow; therefore, this relationship is physiologically backwards.
D. resistance divided by pressure
This inverts the correct relationship. Placing resistance in the numerator suggests that flow increases as resistance increases, which contradicts the established law that flow decreases when resistance rises.
Conclusion:
The fundamental law governing blood flow states that flow is directly proportional to the pressure gradient and inversely proportional to vascular resistance. Therefore, the correct mathematical representation is pressure divided by resistance.
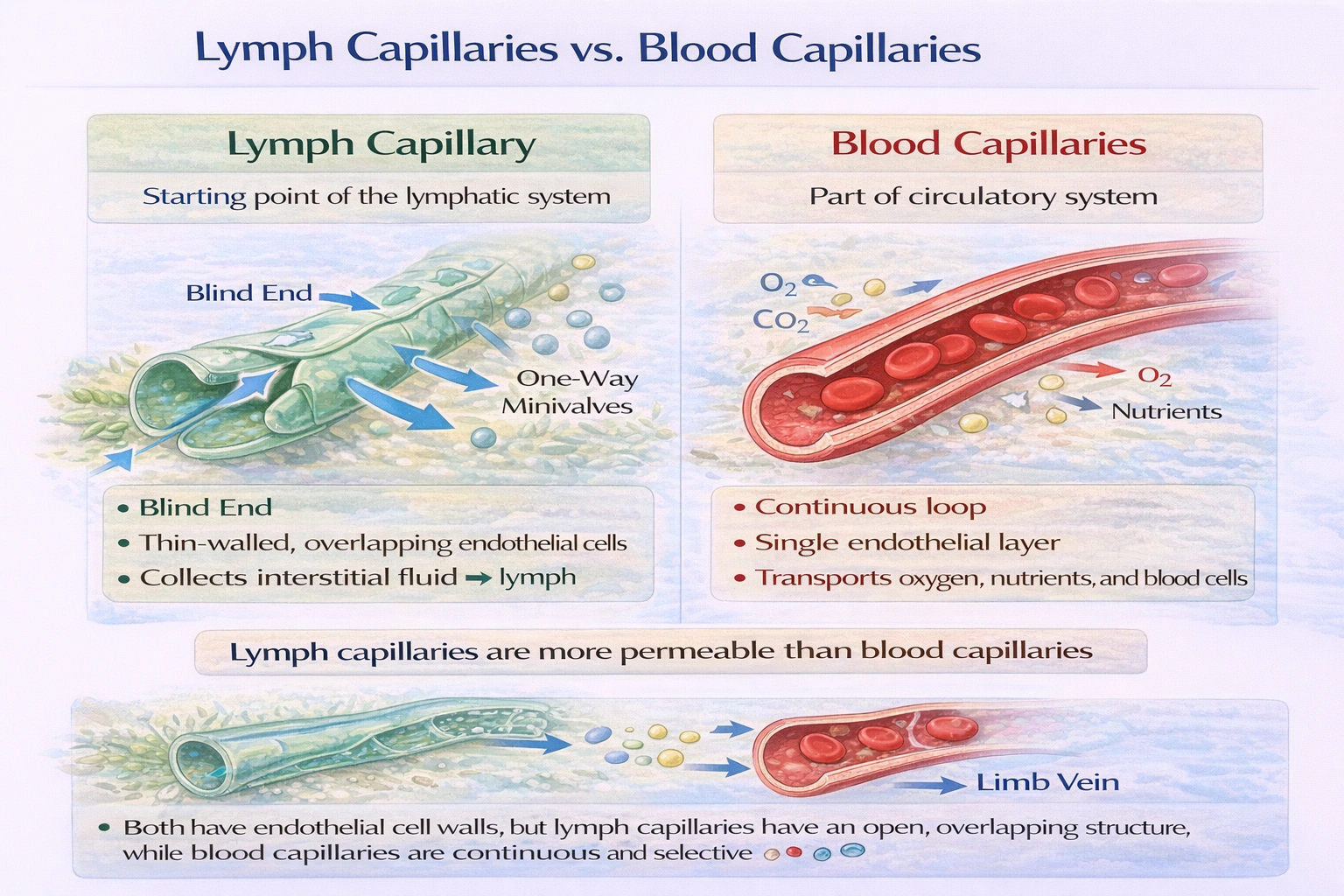
Lymph capillaries differ from blood capillaries in that they
A.
are less permeable.
B. are the starting point of the lymphatic system.
C. contain red blood cells.
D. have walls of endothelial cells.
Rationale
Lymph capillaries differ from blood capillaries in that they are the starting point of the lymphatic system.
Lymph capillaries originate blindly in peripheral tissues as microscopic, thin-walled vessels that collect excess interstitial fluid. This fluid becomes lymph, which then travels through larger lymphatic vessels, nodes, and ducts before returning to the venous bloodstream. Blood capillaries, in contrast, are part of a continuous, circular network that begins and ends with the heart.
A. are less permeable.
Lymph capillaries are more permeable than blood capillaries. Their endothelial cells overlap, forming one-way minivalves that allow large molecules, proteins, and even cells to enter, while preventing backflow. Blood capillaries have tighter junctions and a basement membrane for selective filtration.
B. are the starting point of the lymphatic system.
This is correct. Lymph capillaries represent the initial, open-ended vessels of the lymphatic network. They absorb interstitial fluid that has leaked from blood capillaries, initiating lymph formation. Blood capillaries are part of the closed cardiovascular circuit.
C. contain red blood cells.
Under normal conditions, lymph does not contain red blood cells. Lymph is derived from interstitial fluid, which is essentially plasma filtrate without formed elements. Blood capillaries carry whole blood, which includes red blood cells.
D. have walls of endothelial cells.
Both lymph and blood capillaries have walls composed of a single layer of endothelial cells. This is a structural similarity, not a distinguishing feature. The key difference lies in the organization and permeability of these endothelial cells.
Conclusion:
A primary distinction between lymph and blood capillaries is their role in their respective systems: lymph capillaries begin the open-ended lymphatic pathway, while blood capillaries are components of the continuous circulatory loop. This functional difference underlies their structural adaptations and permeabilities.
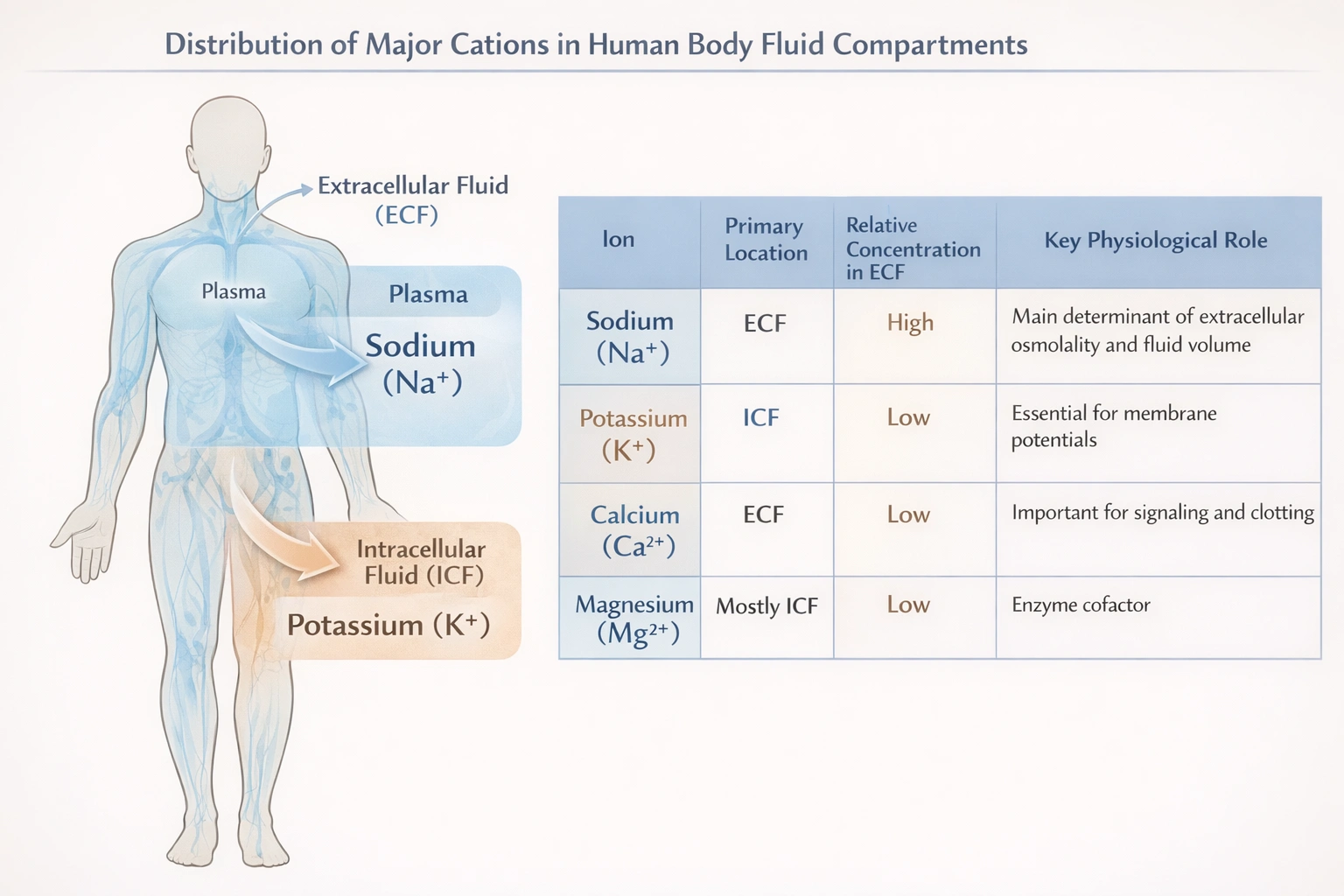
The major cation in the ECF is
A.
calcium.
B. magnesium.
C. potassium.
D. sodium.
Rationale
The major cation in the extracellular fluid (ECF) is sodium.
Cations are positively charged ions. The extracellular fluid compartment includes plasma and interstitial fluid. Sodium (Na+) is the most abundant cation in the ECF, with a normal concentration ranging from 135 to 145 millimoles per liter. It is the primary solute contributing to the osmolality of the ECF, meaning it is the principal determinant of extracellular fluid volume because water follows sodium osmotically. The regulation of sodium balance is therefore closely linked to the regulation of blood pressure and overall fluid homeostasis
A. calcium
Calcium is a vital divalent cation in the ECF, necessary for neuromuscular function, blood clotting, and cell signaling. However, its concentration is much lower (approximately 2.2 to 2.6 mmol/L) and tightly regulated. It is not the primary ion governing ECF volume or osmotic pressure.
B. magnesium
Magnesium is primarily an intracellular cation, with about 99% of the body's stores located within cells or bone. Its extracellular concentration is the lowest among major electrolytes (0.7 to 1.0 mmol/L). While important as a enzyme cofactor, it is a minor contributor to the ECF's cationic pool.
C. potassium
Potassium is the dominant cation inside cells, with an intracellular concentration of about 150 mmol/L. Its extracellular concentration is kept low (3.5 to 5.0 mmol/L) to maintain the resting membrane potential essential for nerve and muscle function. It is not a major cation in the ECF.
D. sodium
Sodium is present in the ECF at a concentration roughly 10 times higher than that of potassium and over 50 times higher than that of calcium. Its abundance and its role as the chief osmotic particle make it the major cation in the ECF. The body's mechanisms for regulating fluid balance, such as the renin-angiotensin-aldosterone system, primarily focus on sodium.
Conclusion
Sodium's high concentration and its central role in determining extracellular osmolality and fluid volume solidify its status as the major cation in the ECF. The other cations, while physiologically critical, are present in much smaller extracellular amounts or are predominantly intracellular.
Which of the following stimulates the hypothalamus to initiate the thirst sensation?
A.
decreased serum osmolality
B. intracellular dehydration
C. decrease in circulating blood volume
D. inhibition of ADH
Rationale
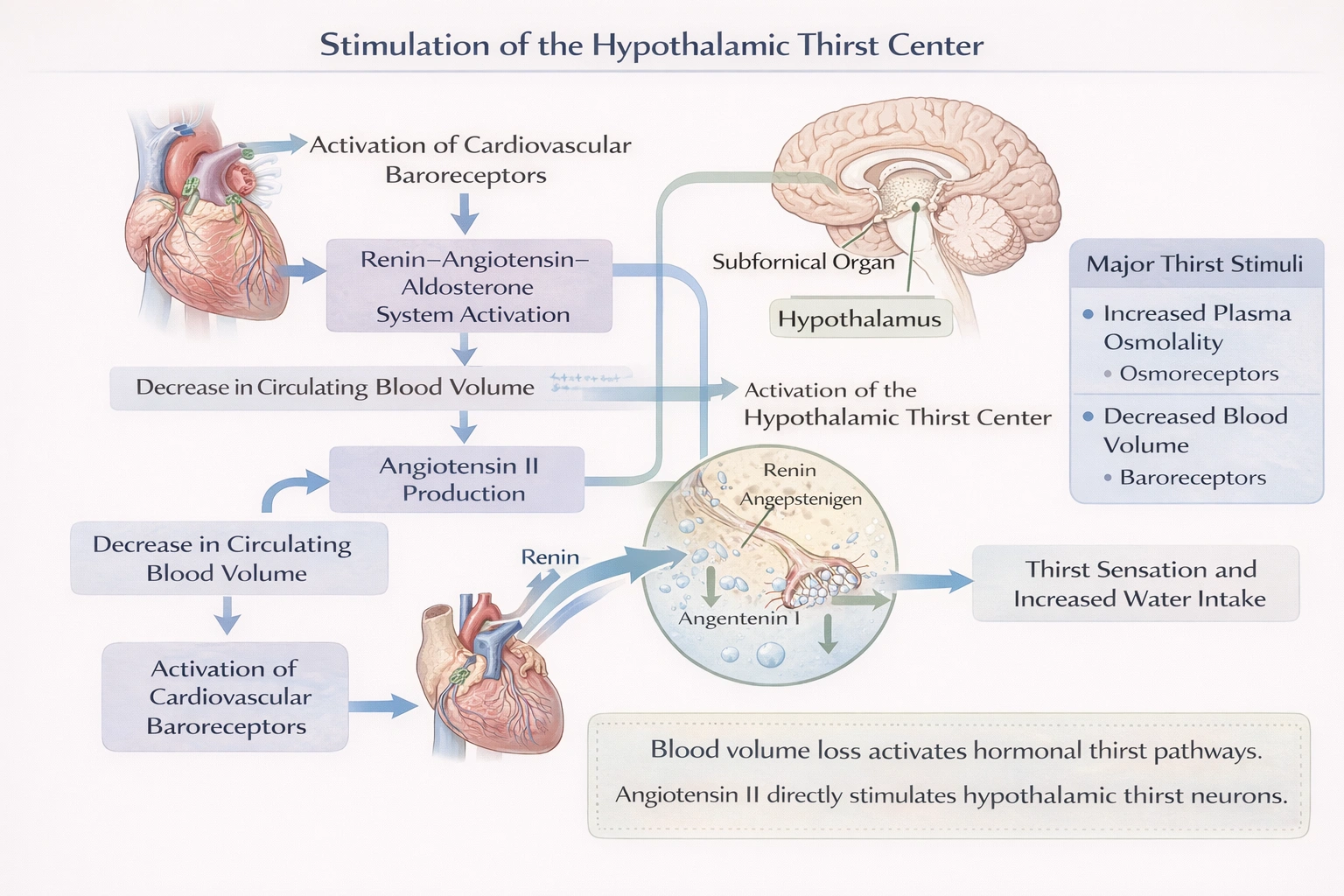
A decrease in circulating blood volume stimulates the hypothalamus to initiate the thirst sensation.
The hypothalamus activates thirst primarily in response to two physiological signals: increased plasma osmolality and decreased effective circulating volume, or hypovolemia. A significant drop in blood volume is sensed by baroreceptors in the great vessels and heart. This triggers the renin-angiotensin-aldosterone system (RAAS), leading to the production of angiotensin II. Angiotensin II acts on the subfornical organ and other circumventricular organs in the brain to directly stimulate neurons in the hypothalamic thirst center, driving conscious fluid-seeking behavior to restore plasma volume.
(A) decreased serum osmolality: A decrease in serum osmolality indicates a hypotonic extracellular environment. Hypothalamic osmoreceptors are exquisitely sensitive to increases in osmolality, which cause cellular dehydration and trigger thirst. A decrease in osmolality inhibits these osmoreceptors, suppressing the thirst center and reducing ADH release to promote water excretion.
(B) intracellular dehydration: Intracellular dehydration is the direct consequence of extracellular hypertonicity, where water leaves cells osmotically. This cellular shrinkage is the specific change detected by hypothalamic osmoreceptors, making it a potent primary stimulus for thirst. However, the question specifies a decrease in blood volume as the correct answer.
(C) decrease in circulating blood volume: This condition, hypovolemia, is detected by the cardiovascular baroreceptors and activates the powerful RAAS pathway, culminating in angiotensin II-mediated thirst stimulation, as detailed above.
(D) inhibition of ADH: Inhibition of antidiuretic hormone results in water diuresis and net fluid loss from the body. This can lead to subsequent dehydration, which increases osmolality or decreases volume, thereby secondarily activating thirst pathways. The inhibition itself, however, is a separate regulatory event and not the direct signal to the hypothalamic thirst center.
Conclusion
A decrease in circulating blood volume serves as a critical, independent trigger for thirst, utilizing the baroreceptor-RAAS-angiotensin II pathway to promote fluid intake and correct hypovolemia.
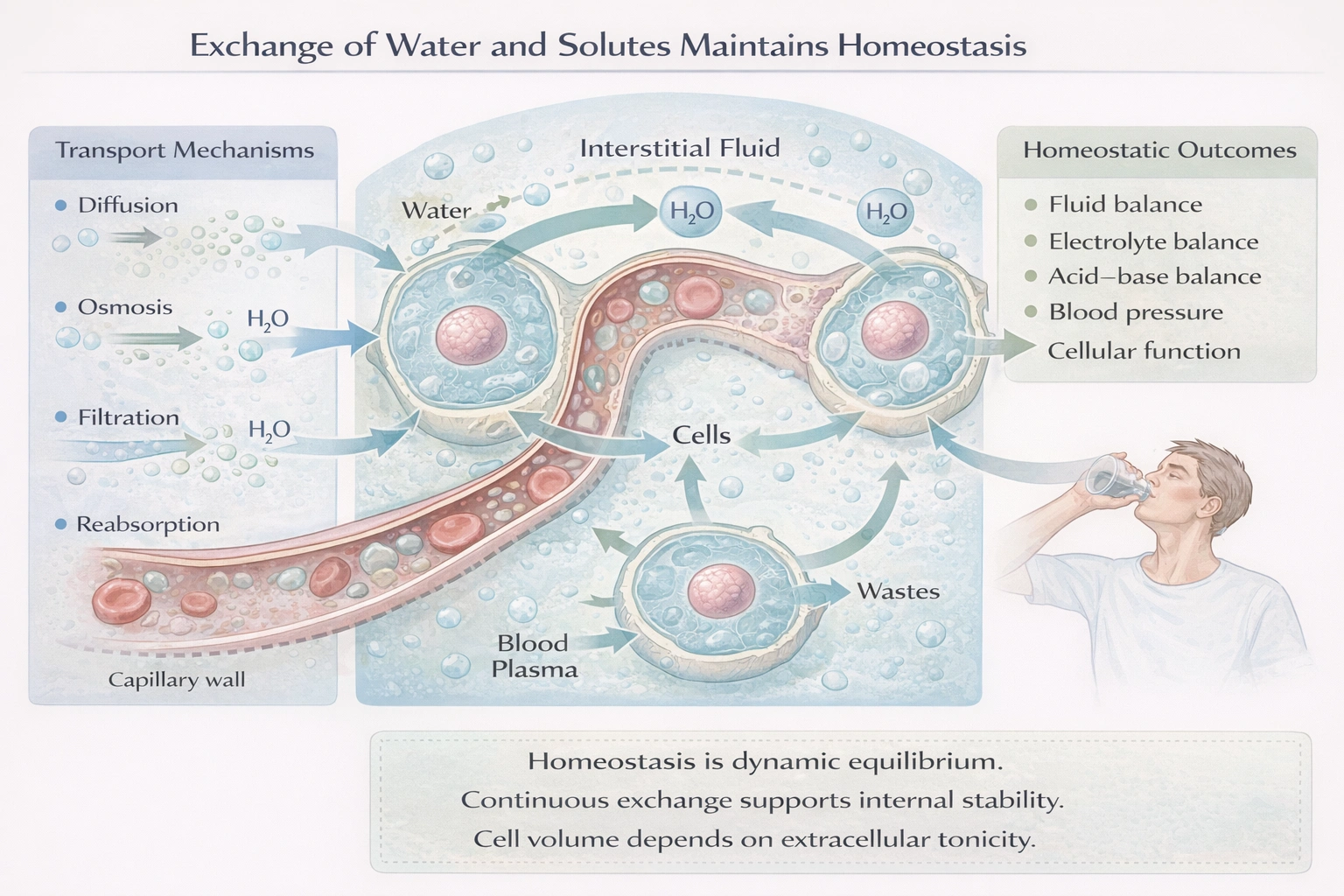
Water and solutes are in constant movement and exchange to assist in maintaining
A.
allocation of body fluids.
B. inhibition of acid-base balance.
C. interstitial compartments.
D. homeostasis of the body.
Rationale
Water and solutes are in constant movement and exchange to assist in maintaining homeostasis of the body.
Homeostasis is the state of dynamic equilibrium within the internal environment. The continuous, bidirectional movement of water, electrolytes, gases, nutrients, and wastes via processes like filtration, reabsorption, diffusion, and osmosis across capillary walls and cell membranes is the fundamental mechanism that allows for the regulation of all critical physiological variables.
(A) allocation of body fluids: The movement of fluids does result in their distribution among the intracellular, interstitial, and plasma compartments. However, this "allocation" is one of the outcomes of the exchange processes, serving the higher-purpose goal of systemic stability, not the ultimate goal itself.
(B) inhibition of acid-base balance: The constant movement of solutes, particularly hydrogen ions, bicarbonate, and carbon dioxide, is the very basis for establishing and regulating acid-base balance. These processes work to maintain balance, not to inhibit it.
(C) interstitial compartments: Maintaining the interstitial environment is a vital, localized function of fluid and solute exchange, ensuring nutrient delivery and waste removal for cells. This is one specific compartmental outcome of the broader exchange processes that sustain the whole organism.
(D) homeostasis of the body: This exchange is the foundational activity that enables the regulation of core parameters such as body temperature, blood pressure, pH, and fluid/electrolyte balance, which collectively constitute homeostasis.
Conclusion
The relentless exchange of materials is the essential physical process underpinning the body's ability to maintain homeostasis, the stable internal condition required for survival and function.
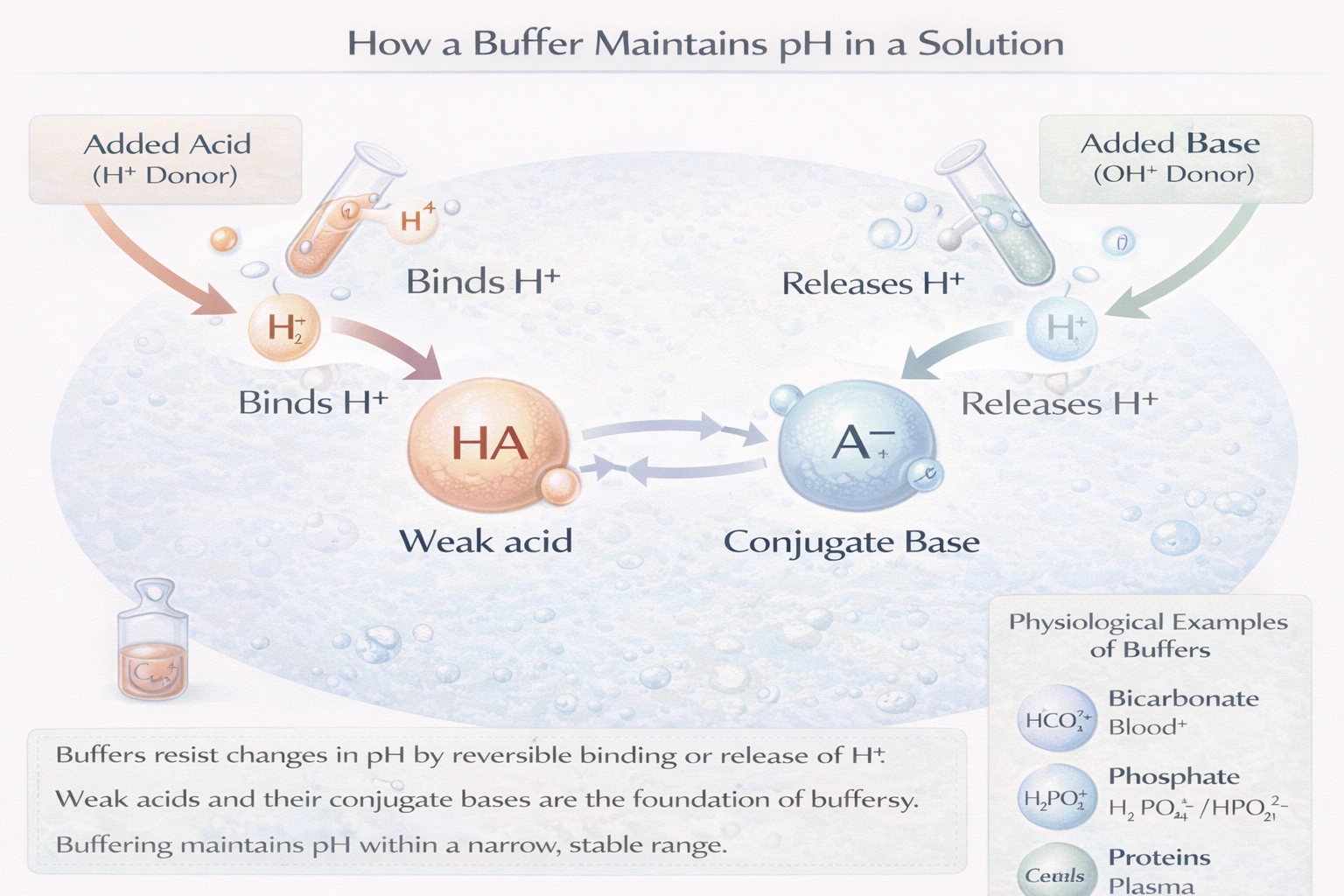
A (An) __________ is a molecule that tends to either bind or release hydrogen ions in order to maintain a particular pH.
A.
acid
B. base
C. buffer
D. nonelectrolyte
Rationale
A buffer is a molecule that tends to either bind or release hydrogen ions in order to maintain a particular pH.
A buffer is a substance or combination of substances that resists drastic changes in the pH of a solution when small amounts of acid (H⺠donors) or base (OH⻠donors) are added. It typically consists of a weak acid and its conjugate base (or a weak base and its conjugate acid). A buffer works by having components that can neutralize added ions: the base component can bind excess H⺠ions, while the acid component can release H⺠ions to neutralize added OH⻠ions. This dual action allows it to maintain the hydrogen ion concentration, and thus the pH, within a narrow, stable range.
(A) acid: An acid is a substance that donates hydrogen ions (Hâº) to a solution, thereby increasing the H⺠concentration and lowering the pH. Its defining action is to change the pH, not to stabilize or maintain it against changes.
(B) base: A base is a substance that accepts hydrogen ions (Hâº) or donates hydroxide ions (OHâ») to a solution, thereby decreasing the H⺠concentration and raising the pH. Similar to an acid, a base acts as an agent that alters the pH rather than resists changes to it.
(C) buffer: A buffer is specifically defined by its capacity to minimize pH fluctuations through its ability to alternately bind or release H⺠ions, as described in the explanation above.
(D) nonelectrolyte: A nonelectrolyte is a substance that dissolves in water but does not dissociate into ions (e.g., glucose, urea). Because it does not produce ions in solution, it does not interact with H⺠or OH⻠ions and has no capacity to influence the pH of a solution.
Conclusion
A buffer is the specific type of molecule or system designed to maintain pH stability by its reversible binding and release of hydrogen ions, making it the correct term for the described function.
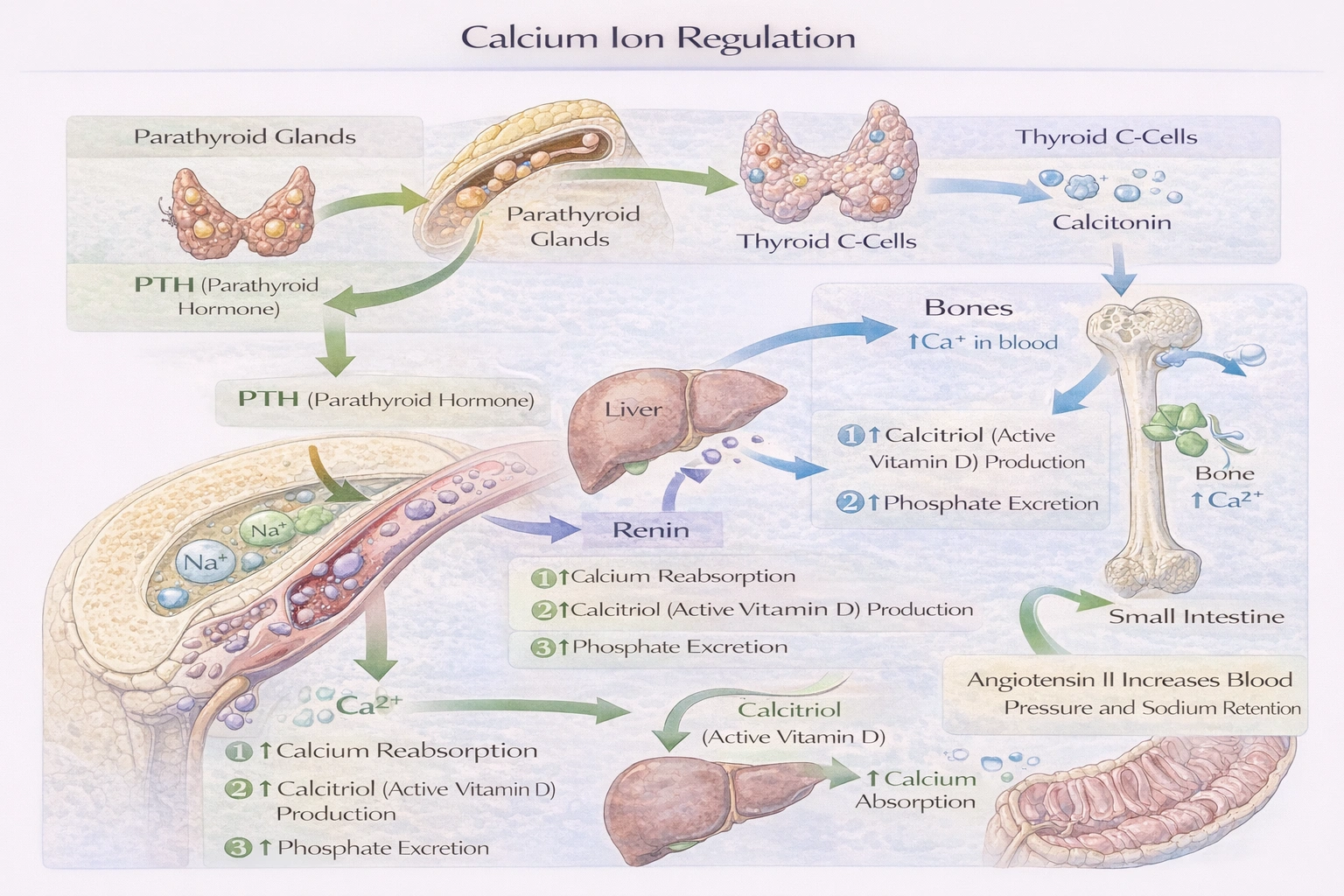
Calcium ions are regulated by hormones of the __________ gland.
A.
adrenal
B. parathyroid
C. pineal
D. pituitary
Rationale
Calcium ions are regulated by hormones of the parathyroid gland.
The maintenance of ionized calcium within a narrow physiological range (approximately 1.1-1.3 mmol/L) is critical for neuromuscular excitability, cardiac function, blood coagulation, and bone integrity. The parathyroid glands, typically four small glands embedded in the posterior capsule of the thyroid, are the central orchestrators of this balance through the secretion of parathyroid hormone. Chief cells within these glands sense extracellular calcium levels via a G-protein coupled calcium-sensing receptor. When ionized calcium levels drop, inhibition of this receptor is relieved, leading to increased synthesis and secretion of PTH, an 84-amino acid polypeptide. PTH acts directly on bone and kidney. On bone, it binds to receptors on osteoblasts, which in turn release factors (like RANKL) that stimulate osteoclast differentiation and activity, resulting in bone resorption and the release of calcium and phosphate into the bloodstream. In the kidney, PTH acts on the proximal convoluted tubule to inhibit phosphate reabsorption (promoting phosphaturia) and to stimulate the production of 1,25-dihydroxyvitamin D (calcitriol) by activating the enzyme 1-alpha-hydroxylase. In the distal tubule, PTH enhances calcium reabsorption. The calcitriol produced then acts on the small intestine to dramatically increase the absorption of dietary calcium and phosphate. This integrated three-organ response (bone, kidney, gut) effectively raises serum calcium. Conversely, rising calcium levels stimulate the CaSR, suppressing PTH release. While the thyroid C-cells secrete calcitonin, which can lower calcium by inhibiting osteoclasts, its role in human daily calcium homeostasis is minor compared to PTH.
(A) adrenal: The adrenal glands consist of two functionally distinct parts. The adrenal cortex produces glucocorticoids (e.g., cortisol from the zona fasciculata), mineralocorticoids (aldosterone from the zona glomerulosa), and adrenal androgens. Cortisol, in excess, can have anti-vitamin D effects and promote bone resorption, leading to osteoporosis, but this is a pathological catabolic effect, not a precise homeostatic one. Aldosterone regulates sodium and potassium, not calcium. The adrenal medulla produces catecholamines (epinephrine, norepinephrine), which have no direct role in calcium regulation. No adrenal hormone has the primary, physiological function of regulating blood calcium ion concentration.
(B) parathyroid: The parathyroid gland's singular focus is calcium homeostasis. The secretion of PTH is exquisitely sensitive to minute changes in blood calcium. The hormone's actions are rapid (renal effects within minutes) and sustained (bone effects over hours). Disorders like hypoparathyroidism (low PTH) lead to hypocalcemia, tetany, and seizures, while hyperparathyroidism (high PTH) causes hypercalcemia, kidney stones, and bone demineralization, underscoring its critical regulatory role.
(C) pineal: The pineal gland is a small, pine-cone shaped neuroendocrine structure in the epithalamus. Its primary and well-established product is melatonin. Melatonin synthesis is entrained to the light-dark cycle and regulates sleep-wake patterns, seasonal reproductive cycles in animals, and has antioxidant properties. It does not produce any hormone known to influence calcium metabolism, and there is no physiological link between pineal function and calcium ion regulation.
(D) pituitary: The pituitary gland, or hypophysis, is often called the "master gland" because its anterior lobe secretes tropic hormones (TSH, ACTH, FSH, LH) that control other endocrine glands, and growth hormone (GH) and prolactin (PRL) with direct effects. The posterior lobe stores and releases oxytocin and ADH. While GH can influence bone growth and calcium incorporation, and TSH can indirectly affect calcium through its control of thyroid hormone (which affects bone turnover), none of these hormones are the primary or direct regulators of minute-to-minute blood calcium levels. The parathyroid gland operates largely independently of pituitary control.
Conclusion: The parathyroid glands, via the secretion of parathyroid hormone, serve as the body's principal endocrine regulator of ionized calcium, directly modulating bone resorption, renal reabsorption, and intestinal absorption to maintain calcium homeostasis.
A commonly used diagnostic test for evaluating acid-base balance is the arterial blood gas (ABG) that
A.
assesses the efficiency of the renal system.
B. evaluates pulmonary gas exchange efficiency.
C. evaluates serum osmolality.
D. None of the above
Rationale
A commonly used diagnostic test for evaluating acid-base balance is the arterial blood gas (ABG) that, according to the provided options, is best described by "None of the above."
An arterial blood gas (ABG) analysis is a comprehensive diagnostic procedure whose primary and fundamental purpose is to evaluate a patient's acid-base homeostasis and respiratory status. It achieves this by directly measuring specific parameters from an arterial blood sample: pH (the negative logarithm of hydrogen ion concentration, indicating acidaemia or alkalaemia), the partial pressure of carbon dioxide (PaCOâ‚‚) (the respiratory component, reflecting alveolar ventilation), and the partial pressure of oxygen (PaOâ‚‚). Additionally, it either directly measures or allows for the calculation of key metabolic parameters: bicarbonate (HCO₃â») concentration (calculated via the Henderson-Hasselbalch equation from the measured pH and PaCOâ‚‚) and base excess/deficit (a calculated value representing the amount of strong acid or base needed to titrate blood to a normal pH at a standard PaCOâ‚‚, isolating the metabolic component). The interpretation of an ABG involves integrating these values to diagnose complex disorders, such as metabolic acidosis, respiratory alkalosis, or mixed acid-base disturbances. While it provides critical data on pulmonary oxygen exchange (via PaOâ‚‚) and the efficiency of COâ‚‚ elimination (via PaCOâ‚‚), its design and clinical application are fundamentally centered on providing a complete picture of acid-base balance by assessing both respiratory and metabolic components in tandem. No single option (A, B, or C) fully or accurately captures this comprehensive, dual-purpose nature of the test in the context of acid-base evaluation.
(A) assesses the efficiency of the renal system. While the kidneys are the ultimate long-term regulators of acid-base balance by reabsorbing filtered bicarbonate, generating new bicarbonate, and excreting titratable acids and ammonium ions, the ABG test itself is not a direct test of renal efficiency or function. The ABG offers a real-time snapshot of the blood's acid-base status, which is influenced by both respiratory and renal (metabolic) function. However, it does not measure glomerular filtration rate (GFR), renal blood flow, or tubular secretory capacity. A normal or abnormal bicarbonate level on an ABG may suggest a renal contribution to a metabolic disturbance, but confirming renal efficiency requires separate tests like serum creatinine, blood urea nitrogen (BUN), estimated GFR, urinalysis, or fractional excretion of electrolytes. For instance, a patient with chronic renal failure will have a metabolic acidosis visible on ABG, but the ABG result alone does not diagnose the renal failure; it only identifies the acidotic consequence.
(B) evaluates pulmonary gas exchange efficiency. This option identifies a major and critical function of the ABG but presents it in an isolated and incomplete manner relative to the question's focus on "acid-base balance." The ABG is the gold standard for assessing gas exchange: the PaOâ‚‚ directly indicates arterial oxygenation efficiency and can reveal hypoxemia, while the PaCOâ‚‚ is a precise measure of alveolar ventilation (high PaCOâ‚‚ indicates hypoventilation; low PaCOâ‚‚ indicates hyperventilation). However, the question specifically asks about evaluating "acid-base balance." PaCOâ‚‚ is the respiratory determinant of blood pH, making it an inseparable part of acid-base analysis. By stating "evaluates pulmonary gas exchange efficiency," this option reduces the ABG's role to a pulmonary function test, ignoring its simultaneous and integral role in quantifying the respiratory acid-base component and its interplay with metabolic factors (HCO₃â», base excess) to form a complete acid-base diagnosis.
(C) evaluates serum osmolality. Serum osmolality is a distinct laboratory value that quantifies the total concentration of osmotically active solutes (e.g., sodium, glucose, urea) per kilogram of plasma water. It is crucial for diagnosing disorders of water balance, such as diabetes insipidus, syndrome of inappropriate antidiuretic hormone secretion (SIADH), and toxic alcohol ingestions. Osmolality is either measured directly by freezing point depression osmometry or calculated from the formula: (2 x Na+) + (Glucose / 18) + (BUN / 2.8). A standard arterial blood gas panel does not measure or report serum osmolality. These are separate tests ordered for different clinical questions osmolality for fluid/tonicity disorders, ABG for acid-base and respiratory status. Confusing the two can lead to significant diagnostic error.
(D) None of the above. This is the correct selection because each preceding option is either incorrect or provides an incomplete description of the ABG's role in evaluating acid-base balance. Option A incorrectly assigns a direct renal assessment function to the ABG. Option B correctly identifies a key capability but frames it too narrowly, isolating pulmonary gas exchange from the holistic acid-base analysis the test is designed to perform. Option C is factually wrong, as osmolality is not part of an ABG analysis. The ABG's definitive purpose in clinical practice is the integrated assessment of acid-base status (through pH, PaCOâ‚‚, HCO₃â», and base excess) and respiratory function (through PaOâ‚‚ and PaCOâ‚‚). Therefore, none of the individual statements accurately and fully describe the test in the context of acid-base evaluation.
Conclusion: An arterial blood gas analysis is an indispensable diagnostic tool designed to provide a simultaneous, integrated evaluation of a patient's acid-base equilibrium (encompassing both respiratory and metabolic components) and respiratory gas exchange. Its scope and utility extend beyond the specific, limited functions described in options A, B, and C, making "None of the above" the only accurate choice for describing its primary use in evaluating acid-base balance.
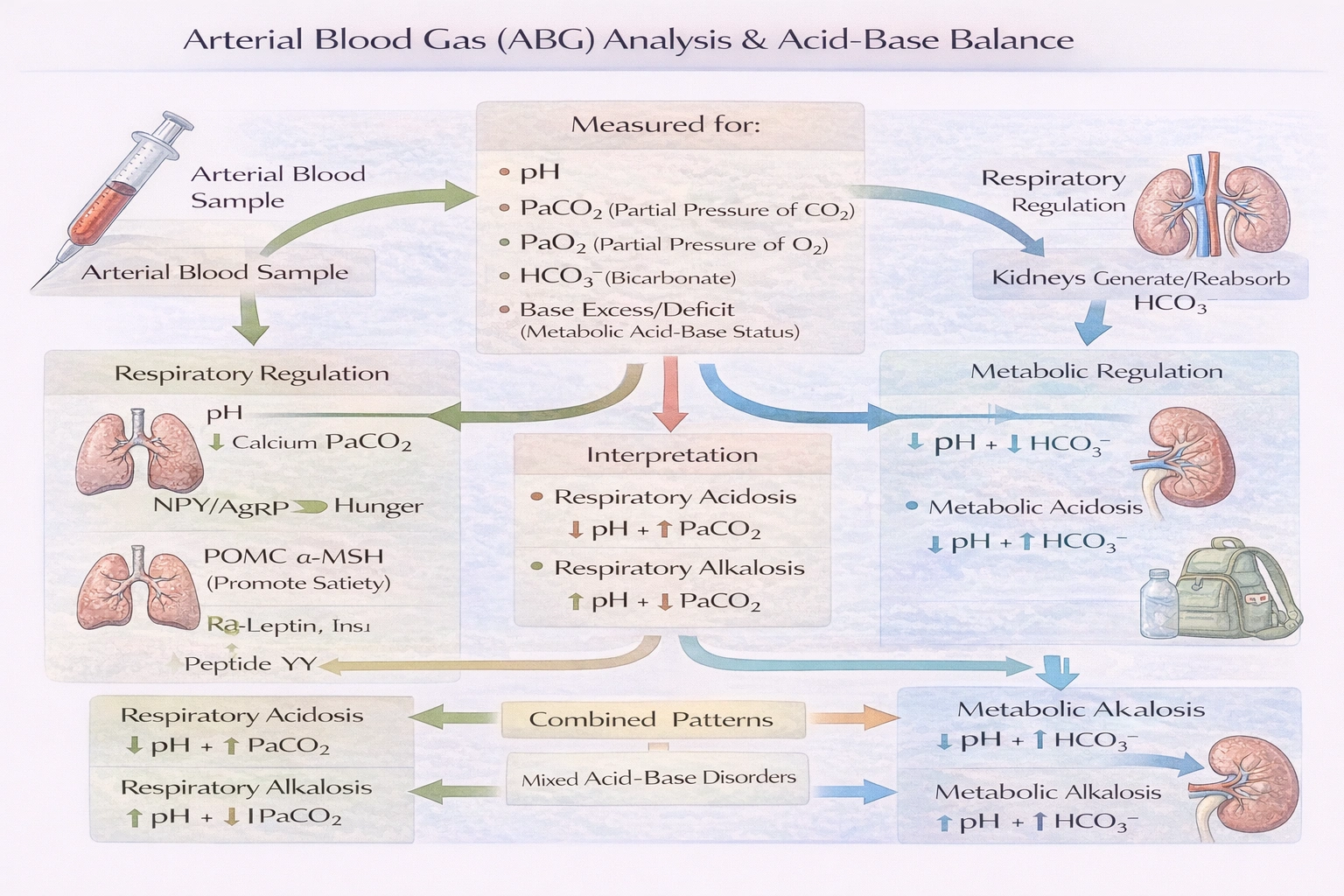
Accessory organs of digestion include all of the following EXCEPT
A.
esophagus.
B. gallbladder.
C. liver.
D. salivary glands.
Rationale
Accessory organs of digestion include all of the following EXCEPT the esophagus.
The accessory organs of digestion are defined as structures that are not part of the continuous alimentary canal (the tube from mouth to anus) but which secrete substances essential for the chemical breakdown of food. These organs develop as outgrowths from the embryonic gut and connect to the digestive tract via ducts. The esophagus, however, is a primary and integral component of the alimentary canal itself. It is a muscular tube lined with stratified squamous epithelium designed for protection during the rapid transit of the food bolus. Its function is purely mechanical transport via peristalsis; it secretes mucus for lubrication but produces no digestive enzymes, acids, buffers, or emulsifying agents. Therefore, while the gallbladder, liver, and salivary glands all secrete critical digestive substances (bile and saliva, respectively) and are anatomically external to the gut lumen, the esophagus is the conduit itself and does not meet the criteria of an accessory organ.
(A) esophagus. The esophagus is the muscular conduit of the upper gastrointestinal tract whose exclusive role is propulsion. It originates from the embryonic foregut and functions as a passageway, moving the bolus from the pharynx to the stomach through coordinated peristaltic contractions. Its mucosal and submucosal glands produce only mucus to reduce friction and protect the epithelial lining. It contains no acinar cells, chief cells, or other secretory cell types capable of producing substances that chemically digest macronutrients. As a fundamental segment of the alimentary canal through which food directly passes, it is categorically not an accessory organ.
(B) gallbladder. The gallbladder is a classic accessory digestive organ. It is a small, hollow, pear-shaped sac located on the inferior surface of the liver. Its primary function is to store and concentrate hepatic bile by actively absorbing water and ions. When stimulated by the hormone cholecystokinin (CCK) in response to dietary fats in the duodenum, the gallbladder contracts and ejects its concentrated bile via the cystic duct into the common bile duct, which empties into the duodenum. Bile is not an enzyme but contains bile salts that emulsify lipids, a crucial step in fat digestion and absorption. The gallbladder's role is purely secretory storage; food does not enter it.
(C) liver. The liver is the largest gland in the body and a vital accessory digestive organ. Beyond its hundreds of metabolic functions (including detoxification, protein synthesis, and glycogen storage), its primary digestive role is the continuous synthesis of bile. Hepatocytes secrete bile into bile canaliculi, which drain into progressively larger ducts, ultimately forming the common hepatic duct. Bile contains bile salts (for fat emulsification), cholesterol, phospholipids (like lecithin), and the waste product bilirubin. This secretion is essential for the digestion and absorption of dietary fats and fat-soluble vitamins (A, D, E, K). The liver's digestive function is exclusively secretory, qualifying it as a major accessory organ.
(D) salivary glands. The major salivary glands (parotid, submandibular, and sublingual) are accessory organs that produce and secrete saliva into the oral cavity via distinct ducts. Saliva is a complex fluid containing water, electrolytes, mucus, antibacterial compounds (like lysozyme and IgA), and the digestive enzymes salivary amylase (ptyalin) and lingual lipase. Salivary amylase initiates the hydrolysis of starch into maltose and dextrins, while lingual lipase begins triglyceride digestion. This enzymatic secretion is fundamental to the initial chemical digestion of food, and because the glands are external to the oral cavity and connect via ducts, they fit the definition of accessory organs.
Conclusion: Accessory digestive organs are defined by their secretion of substances that aid chemical digestion while being external to the alimentary canal; the esophagus is an intrinsic part of the canal responsible for transport, making it the clear exception.
Deglutition is the process of
A.
absorption of nutrients.
B. elimination of indigestible food.
C. swallowing partially digested food.
D. None of the above
Rationale
Deglutition is the process of, as defined by the provided options, "None of the above."
Deglutition is the precise physiological term for the complex neuromuscular act of swallowing. It is a coordinated sequence that transports a bolus from the oral cavity into the esophagus, bypassing the airway. The process is classically divided into three phases:
1) Oral (Voluntary) Phase: The tongue presses the bolus against the hard palate and propels it posteriorly into the oropharynx.
2) Pharyngeal (Involuntary) Phase: A reflex triggered by tactile stimulation of the oropharynx. This phase involves the sequential elevation of the soft palate (to close the nasopharynx), elevation and anterior movement of the hyoid bone and larynx, closure of the glottis by the epiglottis and vocal folds, and contraction of the pharyngeal constrictor muscles to propel the bolus through the upper esophageal sphincter.
3) Esophageal (Involuntary) Phase: Primary peristalsis (a coordinated wave of muscular contraction) moves the bolus down the esophagus to the stomach.
Critically, the material swallowed is a bolus a cohesive mass of masticated food mixed with saliva. It is not "partially digested food," which accurately describes chyme, the semi-fluid mixture of partially digested food, gastric secretions, and hydrochloric acid found in the stomach. Deglutition occurs before gastric digestion, so the bolus has undergone only minimal chemical alteration. Therefore, deglutition is specifically defined as swallowing, which is distinct from nutrient absorption, waste elimination, or the swallowing of chyme.
(A) absorption of nutrients. Nutrient absorption is the process by which the end products of digestion (monosaccharides, amino acids, fatty acids, monoglycerides, vitamins, minerals, and water) are transported across the intestinal epithelium into the bloodstream (portal circulation) or lymphatic system (lacteals). This occurs predominantly in the small intestine (duodenum, jejunum, ileum), facilitated by structural adaptations like villi, microvilli, and specific membrane transport proteins. Absorption is spatially, temporally, and functionally distinct from swallowing, which is an initial mechanical and neural event occurring in the upper GI tract.
(B) elimination of indigestible food. The elimination of indigestible residues, bacteria, sloughed epithelial cells, and other waste as feces is termed defecation. This process involves the rectum and anus and is governed by the defecation reflex, which integrates involuntary spinal reflexes with voluntary cortical control. Elimination represents the terminal end of the digestive process, occurring hours to days after ingestion and deglutition.
(C) swallowing partially digested food. This is a physiologically inaccurate description for deglutition. The substance swallowed is the bolus, which has undergone only initial mechanical breakdown and very limited chemical digestion (primarily from salivary amylase). True partial digestion, resulting in chyme, occurs in the stomach after the action of gastric acid, pepsin, and gastric lipase. Deglutition is completed before food reaches the stomach and is transformed into chyme. Therefore, swallowing precedes the creation of partially digested food in the gastric sense.
(D) None of the above. Since deglutition is defined specifically as the act of swallowing a bolus and does not correspond to absorption (A), elimination (B), or the swallowing of chyme (C), this is the correct selection. The provided options describe other, later stages of the digestive process or an incorrect characterization of the swallowed material.
Conclusion: Deglutition is a defined neuromuscular event---the swallowing of a food bolus which is not accurately described by any of the other offered phrases, making "None of the above" the appropriate choice.
Secretion of gastric juices is regulated by all of the following EXCEPT
A.
cholecystokinin, peptide, and secretin.
B. hormonal control.
C. the parasympathetic nervous system.
D. the sympathetic nervous system.
Rationale
Secretion of gastric juices is regulated by all of the following EXCEPT cholecystokinin, peptide, and secretin.
Gastric juice secretion (a mixture of hydrochloric acid, pepsinogen, intrinsic factor, and mucus) is meticulously controlled by an interplay of neural, hormonal, and paracrine mechanisms to optimize digestion and protect the gastric mucosa. The classic regulatory framework involves three phases: Cephalic (vagally mediated, triggered by the thought, sight, smell, or taste of food), Gastric (mediated by distension and the chemical nature of stomach contents via local reflexes and gastrin), and Intestinal (inhibitory feedback from the duodenum).
Key stimulatory regulators include: 1) Parasympathetic nervous system (Vagus nerve): Releases acetylcholine, which directly stimulates parietal cells and also triggers the release of gastrin-releasing peptide (GRP) to promote gastrin secretion. 2) Hormonal control (Gastrin): Released from G-cells in the gastric antrum, gastrin is a potent stimulator of acid secretion via direct and indirect (histamine-mediated) pathways. 3) Histamine (Paracrine): Released from enterochromaffin-like (ECL) cells, it acts on H2 receptors of parietal cells to powerfully potentiate acid secretion. The sympathetic nervous system generally inhibits gastric secretion during stress. In contrast, when the duodenum receives acidic, hyperosmotic, or fatty chyme, it releases hormones known as enterogastrones that inhibit gastric secretion and motility to slow gastric emptying. Cholecystokinin (CCK) and Secretin are the prototypical enterogastrones. CCK (from I-cells) stimulates pancreatic enzyme secretion and gallbladder contraction but inhibits gastric acid secretion and gastric motility. Secretin (from S-cells) stimulates pancreatic bicarbonate secretion to neutralize acid and also inhibits gastric acid production. The term "peptide" in the option is ambiguous but likely refers to Gastric Inhibitory Peptide (GIP), which is released in response to fats and glucose and also suppresses gastric acid secretion. Thus, these substances are regulatory but act as inhibitors, not stimulators, of gastric juice production.
(A) cholecystokinin, peptide, and secretin. This group consists of duodenal hormones that function as enterogastrones. Their primary role in gastric regulation is to provide negative feedback. CCK and secretin are released in response to fatty acids and low duodenal pH, respectively, and both act to reduce gastric acid secretion and motility, allowing the duodenum adequate time to neutralize and process the incoming acidic chyme. If "peptide" refers to GIP (also known as glucose-dependent insulinotropic peptide), it is released in response to fats and glucose and also inhibits acid secretion. Therefore, these are exceptions among a list that otherwise implies factors involved in promoting or controlling secretion in a stimulatory context.
(B) hormonal control. Hormonal control is a fundamental and broad regulatory mechanism for gastric secretion. The prototypical hormone is gastrin, which powerfully stimulates acid release. Other hormones, like ghrelin (the "hunger hormone"), also have stimulatory effects. This broad option correctly identifies that hormones play a major role in regulation, though the term encompasses both stimulatory (gastrin) and inhibitory (e.g., somatostatin) hormones.
(C) the parasympathetic nervous system. The parasympathetic division, via the vagus nerve (Cranial Nerve X), is the principal neural stimulator of gastric secretion. During the cephalic and gastric phases, vagal activation leads to direct cholinergic stimulation of parietal cells and indirect stimulation via GRP-induced gastrin release. This system is crucial for the anticipatory "feed-forward" and initial digestive response to food.
(D) the sympathetic nervous system. The sympathetic nervous system exerts a predominantly inhibitory influence on gastric secretion as part of the systemic "fight-or-flight" response. Norepinephrine from sympathetic postganglionic fibers and circulating epinephrine reduce blood flow to the gastric mucosa, decrease peristaltic activity, and inhibit the secretory activity of gastric glands via direct and indirect mechanisms. While it suppresses secretion, it is still a regulatory factor within the autonomic nervous system's control over gastric function.
Conclusion: Gastric juice secretion is promoted by hormonal (gastrin), parasympathetic, and local paracrine factors, and is inhibited by sympathetic input and specific intestinal hormones. Cholecystokinin and secretin are intestinal hormones that function as inhibitors of gastric secretion, making them the exception in a list that, in context, implies regulatory factors typically involved in stimulation.
HESI A2 Exams
Biology Quizzes
3 Practice Tests
Biology Quizzes
3 Practice Tests
Chemistry Quizzes
3 Practice Tests
Chemistry Quizzes
3 Practice Tests
Reading Quizzes
4 Practice Tests
Reading Quizzes
4 Practice Tests
Grammar Quizzes
3 Practice Tests
Grammar Quizzes
3 Practice Tests
Vocabulary Quizzes
3 Practice Tests
Vocabulary Quizzes
3 Practice Tests
Physics Quizzes
3 Practice Tests
Physics Quizzes
3 Practice Tests
HESI Quizzes
3 Practice Tests
HESI Quizzes
3 Practice Tests