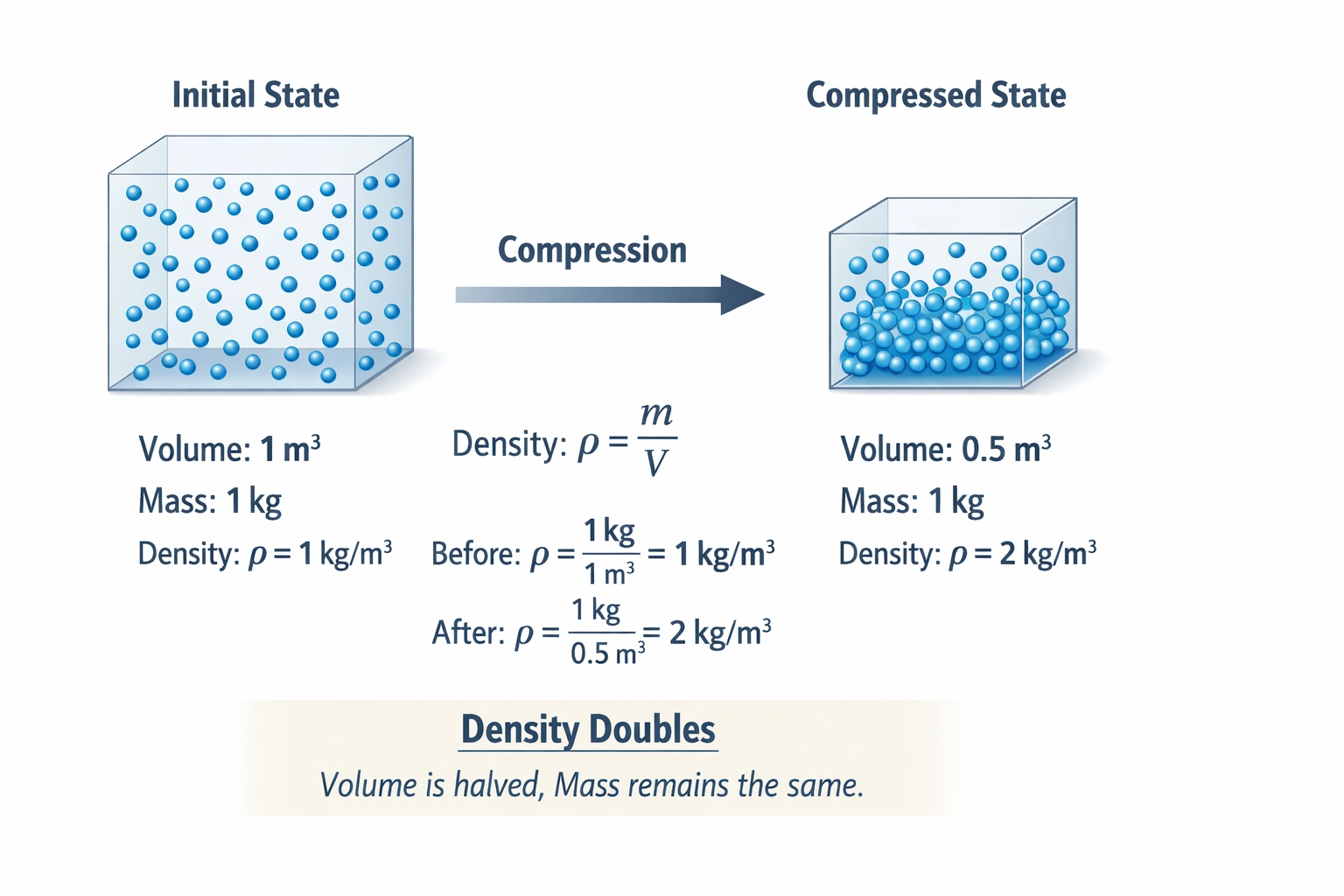
A kilogram of air is compressed from 1 m3 to 0.5 m3. Which statement is true?
-
A
The density is doubled.
-
B
The density is halved.
-
C
The mass is doubled.
-
D
The mass is halved.
Compressing a kilogram of air from 1 m³ to 0.5 m³ doubles its density while leaving mass unchanged.
Density, defined as mass per unit volume (ρ = m/V), responds inversely to volume when mass remains constant, halving the volume concentrates the same kilogram of air into half the space, precisely doubling the mass-to-volume ratio from 1 kg/m³ to 2 kg/m³.
A) The density is doubled.
Density equals mass divided by volume. Initially, density = 1 kg / 1 m³ = 1 kg/m³. After compression, density = 1 kg / 0.5 m³ = 2 kg/m³. The density precisely doubles because volume is halved while mass remains unchanged at one kilogram. This inverse relationship between density and volume under constant mass conditions makes this statement correct.
B) The density is halved.
Halving density would require either halving the mass or doubling the volume, neither occurs during compression. Since volume decreases rather than increases, density must increase rather than decrease. This option contradicts the fundamental definition of density and the physical process described.
C) The mass is doubled.
Mass represents the quantity of matter present and remains conserved during mechanical compression processes that don't involve nuclear reactions or matter addition/removal. The problem explicitly states "a kilogram of air" undergoes compression, confirming mass remains fixed at 1 kg throughout the process. No mechanism exists in simple compression to create additional matter.
D) The mass is halved.
Mass conservation is a fundamental principle in classical physics. Compression rearranges existing molecules into a smaller volume but doesn't eliminate matter. The air sample retains all its original molecules, only their spatial distribution changes. Halving mass would require physically removing half the air, which the problem doesn't describe.
Conclusion
Density varies inversely with volume when mass remains constant. Compressing a fixed mass of gas to half its original volume necessarily doubles its density while leaving mass unchanged. This principle underlies gas behavior in engines, atmospheric science, and respiratory physiology, where volume constraints directly impact material density without altering total mass content.
Topic Flashcards
Click to FlipThe density of a substance is defined as the ratio of two fundamental properties. Write the defining equation and state what each variable represents.
ρ=m/v, where ρ ρ (rho) is density, m m is mass, and V V is volume.
A solid metal block has a mass of 500 grams and a volume of 100 cm³. It is then melted and recast into a new shape with a volume of 50 cm³. What is the new density of the metal, and how does it compare to the original?
The new density is 10 g/cm³. It is exactly the same as the original density. (Density is an intensive property of a material; it does not change with the shape or amount as long as the substance is pure and at the same conditions. Original: 500g/100cm³=5 g/cm³. New mass is still 500g, new volume is 50cm³, so density is 500/50=10 g/cm³.
A sealed container holds a fixed mass of gas. If the volume of the container is reduced to one-third of its original size while temperature is held constant, what is the effect on the gas density?
The density triples. (Mass is constant, so ρ ∝ 1 / V ρ∝1/V. If V n e w = V o r i g i n a l / 3 V new =V original /3, then ρ n e w = 3 × ρ o r i g i n a l ρ new =3×ρ original ).
What is the key principle in classical physics that ensures the mass of a closed system remains constant during physical processes like compression or expansion?
The Law of Conservation of Mass.
If the density of an object increases, what are the two possible changes to its mass and volume that could cause this, assuming only one property changes at a time?
1. Mass increases while volume stays constant. 2. Volume decreases while mass stays constant.