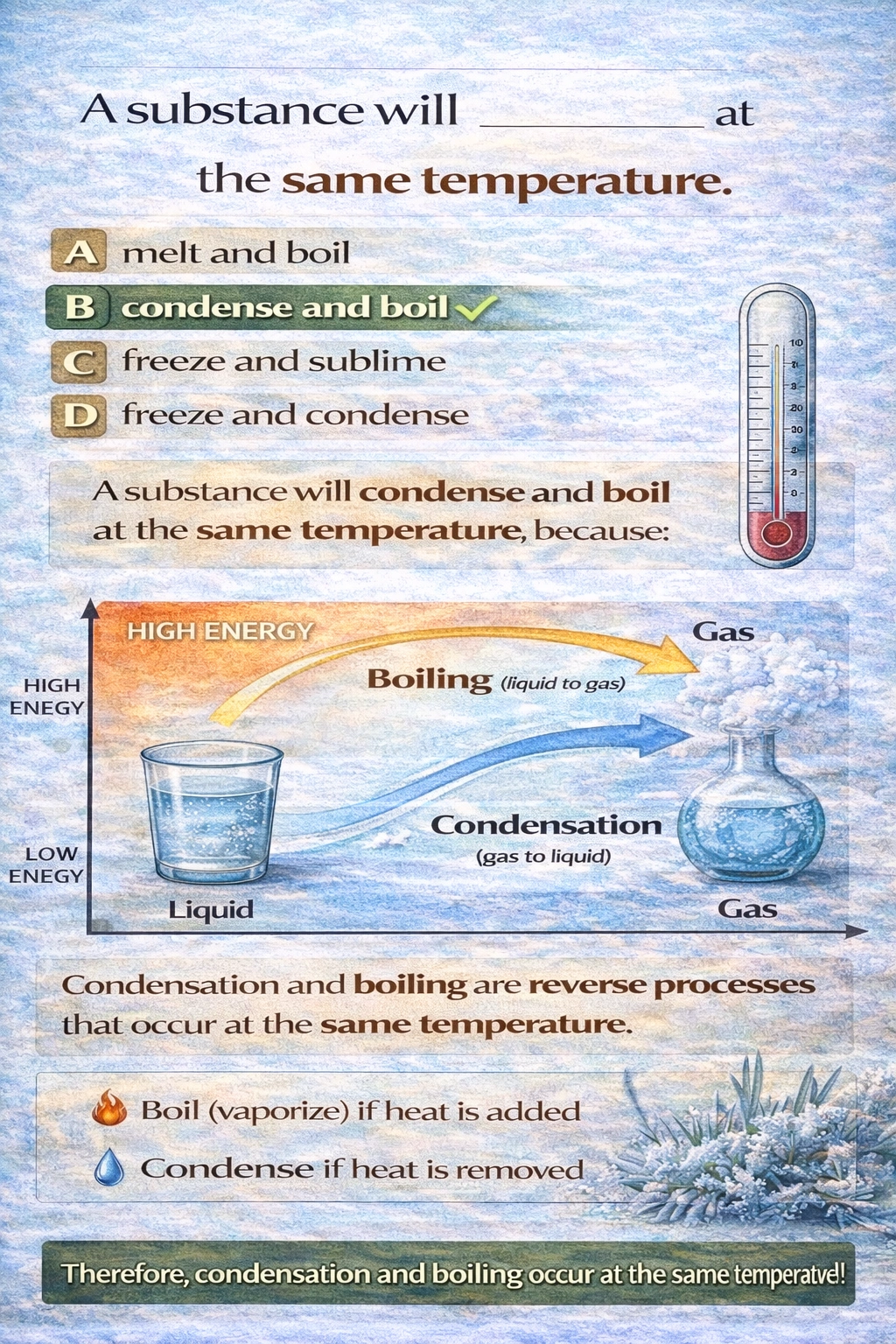
A substance will ______ at the same temperature.
-
A
melt and boil
-
B
condense and boil
-
C
freeze and sublime
-
D
freeze and condense
A substance will condense and boil at the same temperature.
For a pure substance at a given pressure, the transition between two specific phases occurs at a single, defined equilibrium temperature. The forward and reverse processes of the same phase change occur at this identical temperature under equilibrium conditions.
A) Melt and boil
Melting (solid to liquid) and boiling (liquid to gas) are two distinct phase changes that occur at characteristic temperatures: the melting point and the boiling point. These temperatures are not the same under standard conditions for any substance. For example, water melts at 0°C and boils at 100°C at standard pressure.
B) Condense and boil
Condensation (gas to liquid) and boiling (liquid to gas) are reverse processes of the same liquid-gas phase transition. At a specific pressure, the temperature at which a liquid boils is exactly the same as the temperature at which its vapor condenses. This is the substance's boiling point (or condensation point). In a closed system at equilibrium, boiling and condensation occur simultaneously at this temperature.
C) Freeze and sublime
Freezing (liquid to solid) occurs at the freezing point, which is the same as the melting point. Sublimation (solid to gas) occurs under conditions of temperature and pressure below the substance's triple point, where the solid phase is in equilibrium with the gas phase. The sublimation temperature is generally different from the freezing point. For instance, at atmospheric pressure, carbon dioxide sublimates at -78.5°C but does not have a liquid phase, so its freezing point isn't relevant under those conditions.
D) Freeze and condense
Freezing is the liquid-solid transition, and condensation is the gas-liquid transition. These are different phase changes with their own distinct equilibrium temperatures (freezing point and boiling/condensation point). They do not occur at the same temperature under standard conditions.
Conclusion:
The temperature for a phase transition is defined by the equilibrium between two phases. The process of boiling and its reverse, condensation, describe the dynamic equilibrium between the liquid and gas phases at a specific temperature and pressure. Therefore, a substance condenses and boils at the same temperature.
Topic Flashcards
Click to FlipWhat are the two reverse processes that occur at the same temperature for a pure substance?
Boiling (vaporization) and condensation. They describe the equilibrium between liquid and gas phases.
What is the name of the specific temperature at which a liquid boils and its vapor condenses?
The boiling point (which is also the condensation point at a given pressure).
Why is it incorrect to say a substance melts and boils at the same temperature?
Melting (solid-liquid) and boiling (liquid-gas) are two different phase transitions with distinct, separate equilibrium temperatures.
In a closed system at equilibrium (like water in a sealed pot), what two processes are happening simultaneously at the boiling point?
Liquid molecules are vaporizing (boiling) AND gas molecules are condensing back into liquid at equal rates.
For a given pressure, does the temperature of a phase change depend on whether it's the forward or reverse process (e.g., boiling vs. condensing)?
No. The equilibrium temperature between two phases is the same for the forward and reverse process.