The amount of heat that has been removed from the substance allows the particles to draw closer together, and the material changes from a liquid to a solid. Which of the following is being described?
-
A
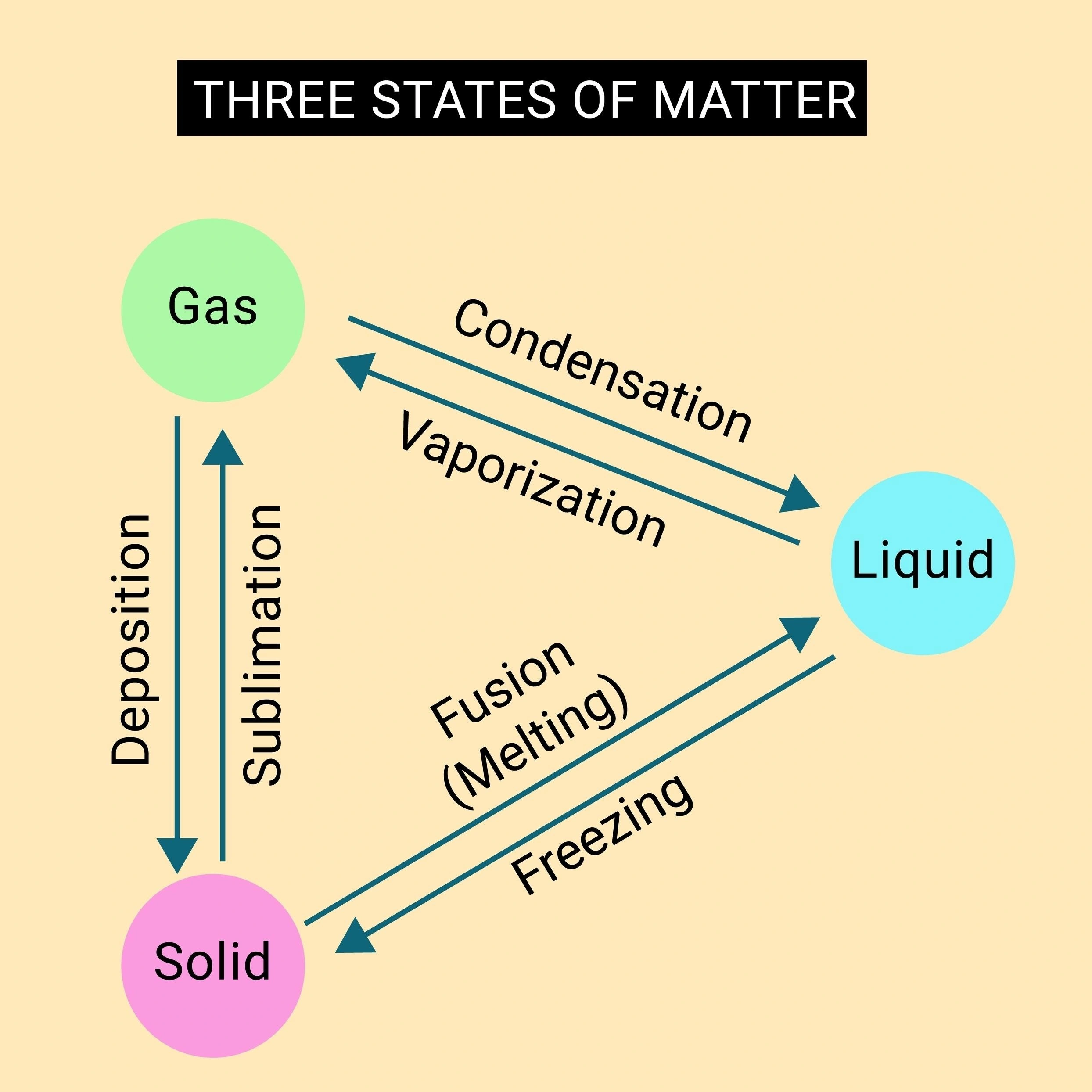
Condensation
-
B
Deposition
-
C
Freezing
-
D
Sublimation
The process being described is freezing.
The description explicitly states the starting phase (liquid), the ending phase (solid), and notes the removal of heat (an exothermic process). This matches the definition of freezing or solidification.
A) Condensation
Condensation is the phase change from a gas to a liquid. Heat is removed during condensation, and particles come closer together as they transition from a dispersed gas to a denser liquid. However, the description specifies the final state is a solid, not a liquid. Therefore, this does not match.
B) Deposition
Deposition is the direct change from a gas to a solid. Heat is released, and particles come together to form a solid. However, the starting state for deposition is a gas, not a liquid. The description clearly begins with a liquid, so this is incorrect.
C) Freezing
Freezing, or solidification, is the phase change from a liquid to a solid. When heat is removed from a liquid, the particles lose kinetic energy. This reduction in motion allows the attractive intermolecular forces to pull the particles into a fixed, ordered arrangement characteristic of a solid. The particles draw closer together into a more rigid structure, and the substance releases latent heat. This description matches freezing perfectly.
D) Sublimation
Sublimation is the change from a solid directly to a gas. This process requires the addition of heat (endothermic) to provide particles with enough energy to break free from the solid lattice and become a dispersed gas. The particles move far apart, and heat is absorbed, not removed. This is the opposite of the described process.
Conclusion:
The only phase change that fits all three criteria—starting as a liquid, ending as a solid, and involving the removal of heat (exothermic)—is freezing. Condensation ends as a liquid, deposition begins as a gas, and sublimation absorbs heat and ends as a gas.
Topic Flashcards
Click to FlipWhat is the name of the phase change where a substance transitions from a liquid to a solid, releasing heat in the process?
Freezing (or solidification).
During freezing, what happens to the motion and arrangement of the particles as heat is removed?
Particles lose kinetic energy, slow down, and are drawn by intermolecular forces into a fixed, ordered arrangement (a solid lattice).
How does the phase change "freezing" differ from "deposition"?
Freezing is liquid → solid. Deposition is gas → solid. Both release heat, but they start from different phases.
True or False: The process of freezing is an endothermic change.
False. Freezing is exothermic; it releases thermal energy (heat) into the surroundings.
What two factors in the description ("heat removed," "liquid to solid") definitively identify the process as freezing?
1. Removal of heat (exothermic). 2. Phase change from liquid to solid. Only freezing fits both.