How many potassium and nitrogen ions are needed to form an ionic compound?
-
A
How many potassium and nitrogen ions are needed to form an ionic compound?
-
B
Three potassium ions and one nitrogen ion
-
C
One potassium ion and three nitrogen ions
-
D
One potassium ion and three nitrogen ions
Three potassium ions and one nitrogen ion are needed.
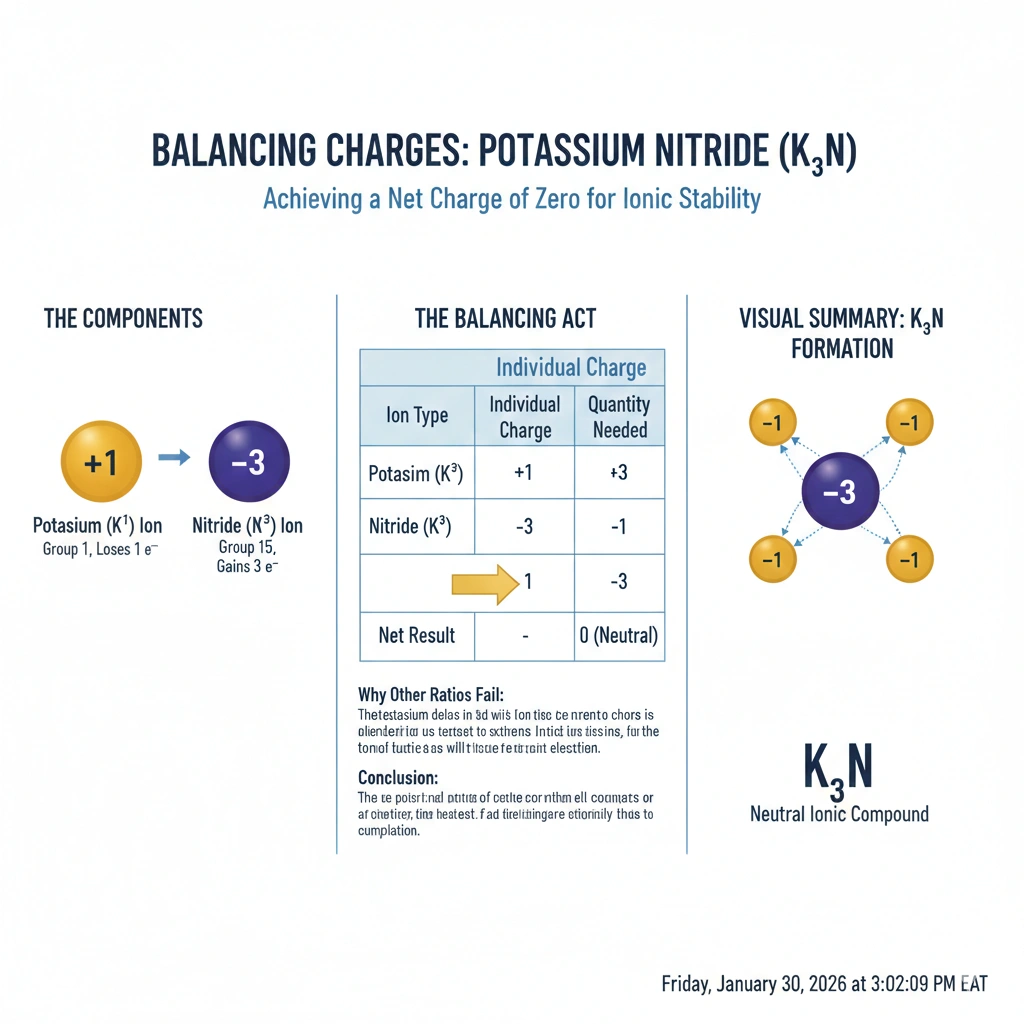
Ionic compounds are electrically neutral. Their chemical formulas are determined by balancing the positive charges of the cations with the negative charges of the anions. Potassium (K) typically forms a +1 ion (K⁺), while nitrogen (N) typically gains three electrons to form a nitride ion with a -3 charge (N³⁻).
A) One potassium ion and one nitrogen ion
One K⁺ ion carries a +1 charge. One N³⁻ ion carries a -3 charge. The net charge would be (+1) + (-3) = -2. This compound would not be electrically neutral and could not exist as a stable, bulk ionic solid. It would be a polyatomic ion with a -2 charge.
B) Three potassium ions and one nitrogen ion
Three K⁺ ions provide a total charge of +3 (3 * +1 = +3). One N³⁻ ion provides a charge of -3. The sum of the charges is (+3) + (-3) = 0. This results in a neutral ionic compound with the formula K₃N, known as potassium nitride.
C) One potassium ion and three nitrogen ions
One K⁺ ion provides a +1 charge. Three N³⁻ ions provide a total charge of -9 (3 * -3 = -9). The net charge would be (+1) + (-9) = -8. This large charge imbalance would prevent the formation of a stable neutral compound.
D) Three potassium ions and three nitrogen ions
Three K⁺ ions provide +3 charge. Three N³⁻ ions provide -9 charge. The net charge would be (+3) + (-9) = -6. Again, this is not electrically neutral and is incorrect.
Conclusion:
To achieve electrical neutrality in an ionic compound, the total positive charge must equal the total negative charge. Since potassium forms a +1 ion and nitrogen forms a -3 ion, the lowest ratio that balances charge is three +1 ions for every one -3 ion. Therefore, three potassium ions and one nitrogen ion are required.
Topic Flashcards
Click to FlipWhat are the charges of the ions formed by potassium (K) and nitrogen (N) when they react?
Potassium forms a +1 cation (K⁺). Nitrogen forms a -3 anion (the nitride ion, N³⁻).
What is the primary rule for determining the formula of an ionic compound?
The compound must be electrically neutral. The total positive charge must equal the total negative charge.
How do you calculate the simplest ratio of K⁺ to N³⁻ ions needed for a neutral compound?
The +1 charge of K⁺ and -3 charge of N³⁻ have a lowest common multiple of 3. Therefore, you need 3 K⁺ ions (+3 total) for every 1 N³⁻ ion (-3 total).
What is the name and chemical formula of the ionic compound formed between potassium and nitrogen?
Potassium nitride, with the formula K₃N.
Why would a compound with the formula "KN" (one K⁺ and one N³⁻) not be stable?
It would have a net charge of -2 [(+1) + (-3) = -2], violating the rule of electrical neutrality for ionic compounds.