What is the net charge of the ionic compound barium iodide (BaI₂)?
-
A
-1
-
B
0
-
C
+1
-
D
+2
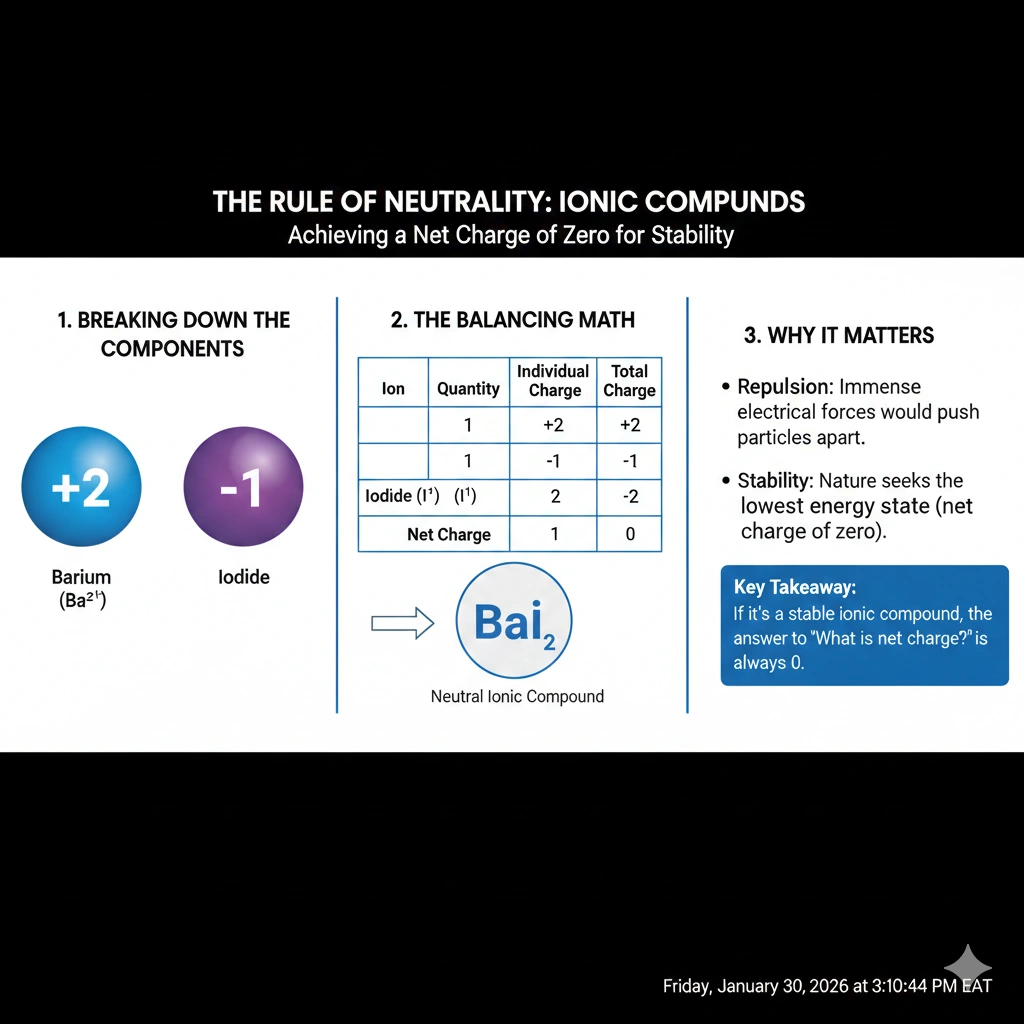
The net charge of barium iodide (BaI₂) is 0.
All stable, standalone ionic compounds must have a net electrical charge of zero. This fundamental rule arises from the need for electrical neutrality in bulk matter; otherwise, immense repulsive forces would prevent the formation of a stable crystal lattice.
A) -1, C) +1, D) +2
These options suggest the compound possesses a net positive or negative charge. A chemical species with a net charge is called an ion (simple or polyatomic). An ionic compound like BaI₂ is a neutral combination of ions. If it had a non-zero net charge, it would be an ion itself and would need to combine with a counterion to form a different neutral compound. For example, [BaI]⁺ would need to combine with an anion like Cl⁻ to form BaICl.
B) 0
Ionic compounds are formed via the electrostatic attraction between cations and anions. The formula subscript indicates the simplest ratio of ions that yields overall charge balance. In barium iodide, the barium ion is Ba²⁺. The iodide ion is I⁻. The subscript "2" after I indicates two iodide ions are present. Thus, the total positive charge is +2 (from one Ba²⁺) and the total negative charge is -2 (from two I⁻ ions: 2 * -1 = -2). The sum is (+2) + (-2) = 0. The compound is electrically neutral.
Conclusion:
The defining characteristic of any ionic compound is its electrical neutrality. The chemical formula is constructed to reflect the precise ratio of ions that cancels all charges. For BaI₂, one Ba²⁺ cation balances two I⁻ anions, resulting in a net charge of zero.
Topic Flashcards
Click to FlipWhat are the individual ion charges that make up barium iodide (BaI₂)?
The barium ion is Ba²⁺ (charge +2). Each iodide ion is I⁻ (charge -1).
What is the fundamental rule about the net charge of any stable, standalone ionic compound?
The net charge must be zero. Ionic compounds are electrically neutral.
How does the subscript "2" in BaI₂ relate to the charge of the Ba²⁺ ion?
The subscript indicates that two I⁻ ions (total charge -2) are needed to balance the +2 charge of one Ba²⁺ ion, achieving neutrality.
If an ionic compound had a net charge of +1, what would it be classified as instead?
It would be classified as a polyatomic ion (a charged molecule), not a neutral ionic compound.
Calculate the total positive and negative charge in one formula unit of BaI₂ to prove its neutrality.
Positive: 1 Ba²⁺ = +2. Negative: 2 I⁻ = 2 * (-1) = -2. Sum: (+2) + (-2) = 0.