Balance this equation: Zn + HCl ZnCl₂ + H₂
-
A
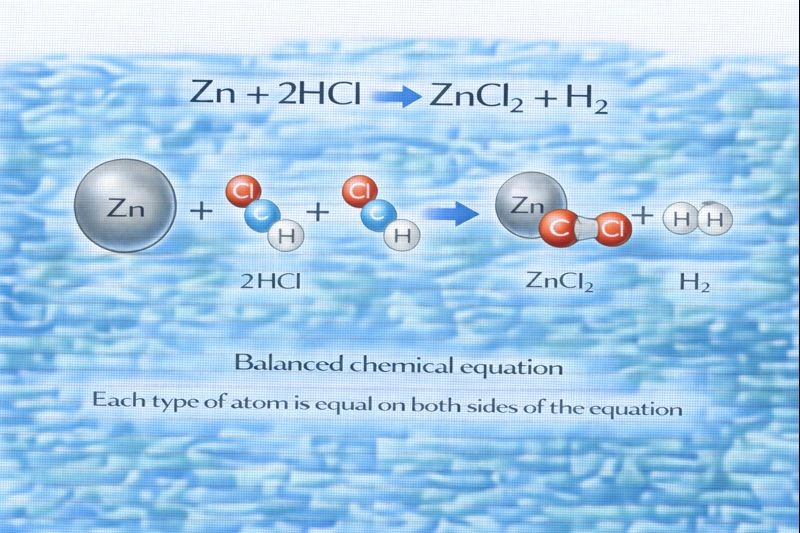
Zn + 2HCl ZnCl₂ + H₂
-
B
. Zn + HCl 2ZnCl₂ + H₂
-
C
2Zn + HCl 2ZnCl₂ + H₂
-
D
Zn + 4HCl ZnCl₂ + H₂
The balanced equation is Zn + 2HCl → ZnCl₂ + H₂.
Balancing a chemical equation requires that the number of each type of atom be the same on both sides of the equation, in accordance with the law of conservation of mass. In this reaction, zinc reacts with hydrochloric acid to produce zinc chloride and hydrogen gas. Careful counting of zinc (Zn), hydrogen (H), and chlorine (Cl) atoms on both sides shows that two molecules of HCl are needed to balance the atoms correctly.
A. Zn + 2HCl → ZnCl₂ + H₂
This option is correct. On the left side, there is one zinc atom, two hydrogen atoms, and two chlorine atoms (from 2HCl). On the right side, there is one zinc atom in ZnCl₂, two chlorine atoms in ZnCl₂, and two hydrogen atoms combined to form H₂ gas. Each element has the same number of atoms on both sides of the equation, making this a properly balanced chemical equation.
B. Zn + HCl → 2ZnCl₂ + H₂
On the right side, the coefficient 2 in front of ZnCl₂ means there are two zinc atoms and four chlorine atoms. However, on the left side there is only one zinc atom and one chlorine atom from a single HCl molecule. This creates an imbalance in both zinc and chlorine atoms.
C. 2Zn + HCl → 2ZnCl₂ + H₂
Although the number of zinc atoms appears balanced (two on each side), the hydrogen and chlorine atoms are not. There is only one hydrogen atom and one chlorine atom on the left side from HCl, but there are four chlorine atoms on the right side in 2ZnCl₂ and two hydrogen atoms forming H₂. The coefficients do not properly balance all elements.
D. Zn + 4HCl → ZnCl₂ + H₂
On the left side, four HCl molecules provide four hydrogen atoms and four chlorine atoms. On the right side, ZnCl₂ contains only two chlorine atoms, and H₂ contains only two hydrogen atoms. This results in excess hydrogen and chlorine atoms on the left side, so the equation is not balanced.
Conclusion
A balanced chemical equation must have equal numbers of each type of atom on both sides. Only the equation Zn + 2HCl → ZnCl₂ + H₂ satisfies this requirement by balancing zinc, hydrogen, and chlorine atoms correctly while using the smallest whole-number coefficients.
Topic Flashcards
Click to FlipWhen balancing the equation Zn + HCl → ZnCl₂ + H₂, how do you ensure the law of conservation of mass is obeyed?
The number of atoms of each element must be equal on both sides. For Zn: 1 on each side. For H: 2 from 2HCl on the left matches 2 in H₂ on the right. For Cl: 2 from 2HCl on the left matches 2 in ZnCl₂ on the right.
Why can't the coefficient for HCl in this reaction be 1? What imbalance would it cause?
If HCl had a coefficient of 1, the left side would have 1 Cl and 1 H, but the right side would have 2 Cl (in ZnCl₂) and 2 H (in H₂). This violates atom conservation.
Write the balanced ionic equation for the reaction between zinc and hydrochloric acid, assuming ZnCl₂ dissociates completely.
Zn(s) + 2H⁺(aq) → Zn²⁺(aq) + H₂(g). (Cl⁻ is a spectator ion).
What type of chemical reaction is Zn + 2HCl → ZnCl₂ + H₂, and what general pattern does it follow?
It is a single displacement (replacement) reaction. The pattern is: A + BC → AC + B, where Zn (A) displaces H (B) from HCl (BC).
If you started with 2 moles of HCl, how many moles of H₂ gas would be produced according to the balanced equation? How many grams of Zn would be required?
From Zn + 2HCl → ZnCl₂ + H₂, 2 moles of HCl produce 1 mole of H₂ and require 1 mole of Zn. 1 mole of Zn = 65.38 g.