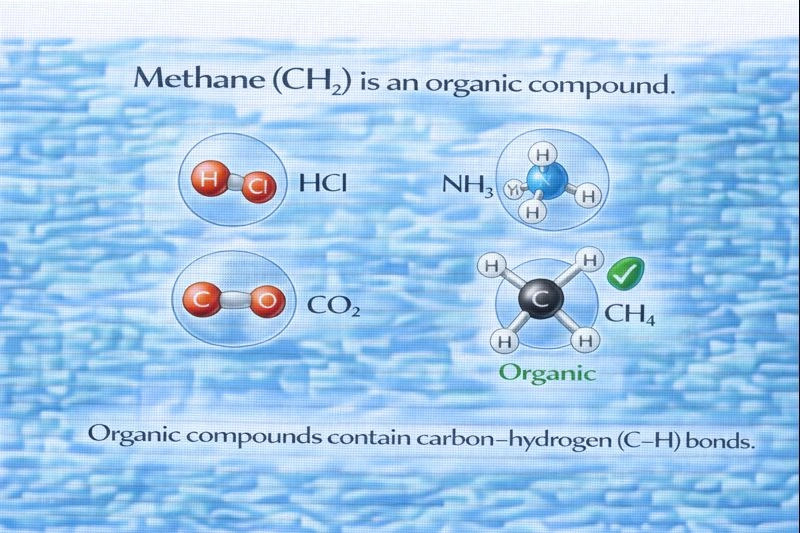
Which of these is an organic compound?
-
A
HCl
-
B
NH₃
-
C
CO₂
-
D
CH₄
The organic compound among the choices is methane (CH₄).
Organic compounds are generally defined as compounds that contain carbon–hydrogen (C–H) bonds. While not all carbon-containing substances are organic, the presence of C–H bonding is a key criterion used in basic chemistry to classify a compound as organic. Methane is the simplest hydrocarbon and serves as the foundational structure for all organic chemistry.
A. HCl
Hydrochloric acid consists of hydrogen and chlorine only and contains no carbon atoms. Because organic compounds must contain carbon—specifically carbon bonded to hydrogen—this substance is classified as inorganic. It is best known as a strong acid rather than an organic compound.
B. NH₃
Ammonia is composed of nitrogen and hydrogen and does not contain carbon. Since carbon is absent altogether, ammonia cannot be considered organic. It is classified as an inorganic compound commonly involved in acid–base chemistry and biological nitrogen metabolism.
C. CO₂
Carbon dioxide does contain carbon; however, it lacks any carbon–hydrogen or carbon–carbon bonds. It is classified as an inorganic oxide of carbon and behaves very differently from organic compounds. Substances such as CO₂, carbonates, and bicarbonates are standard exceptions that contain carbon but are still considered inorganic.
D. CH₄
Methane is composed of one carbon atom bonded to four hydrogen atoms, forming clear C–H bonds. This structure makes methane the simplest hydrocarbon and a classic example of an organic compound. It is the first member of the alkane family and forms the basis for more complex organic molecules.
Conclusion
Organic compounds are identified by the presence of carbon–hydrogen bonds. Among the options given, methane uniquely meets this criterion, while the others are classified as inorganic due to the absence of C–H bonding or carbon altogether.
Topic Flashcards
Click to FlipWhat is the most fundamental defining feature that distinguishes an organic compound from an inorganic one in modern chemistry?
The presence of at least one carbon-hydrogen (C-H) bond. (While most organic compounds contain carbon, the key is its bonding to hydrogen, with notable exceptions like urea).
Why is carbon dioxide (CO₂) classified as an inorganic compound, despite containing carbon?
It lacks carbon-hydrogen (C-H) bonds. Carbon oxides, carbonates, cyanides, and carbides are traditionally considered inorganic.
Urea (CO(NH₂)₂) is considered the first organic compound synthesized in a lab from inorganic precursors. Does it fit the C-H bond rule?
Yes, it contains C-H bonds (its formula is often written as H₂NCONH₂, showing C-H bonds in the -NH₂ groups).
Name the four major classes of large, complex organic molecules that are essential for life (biomolecules).
Carbohydrates, Lipids, Proteins, and Nucleic Acids.
Which of the following is not an organic compound: CH₃OH (methanol), Na₂CO₃ (sodium carbonate), C₆H₁₂O₆ (glucose), CH₃COOH (acetic acid)? Explain why.
Na₂CO₃ (sodium carbonate). It is a salt containing carbon but lacks C-H bonds, making it inorganic.