Compare the melting points of three metals: gold (1063°C), lead (328°C) and mercury (-38.9°C). Which of the following statements is true regarding intermolecular forces of these metals?
-
A
The intermolecular forces are strongest between atoms of gold.
-
B
The intermolecular forces are strongest between atoms of mercury.
-
C
The intermolecular forces of these metals are stronger after they melt.
-
D
The intermolecular forces of these metals are stronger after they melt.
The intermolecular forces are strongest between atoms of gold.
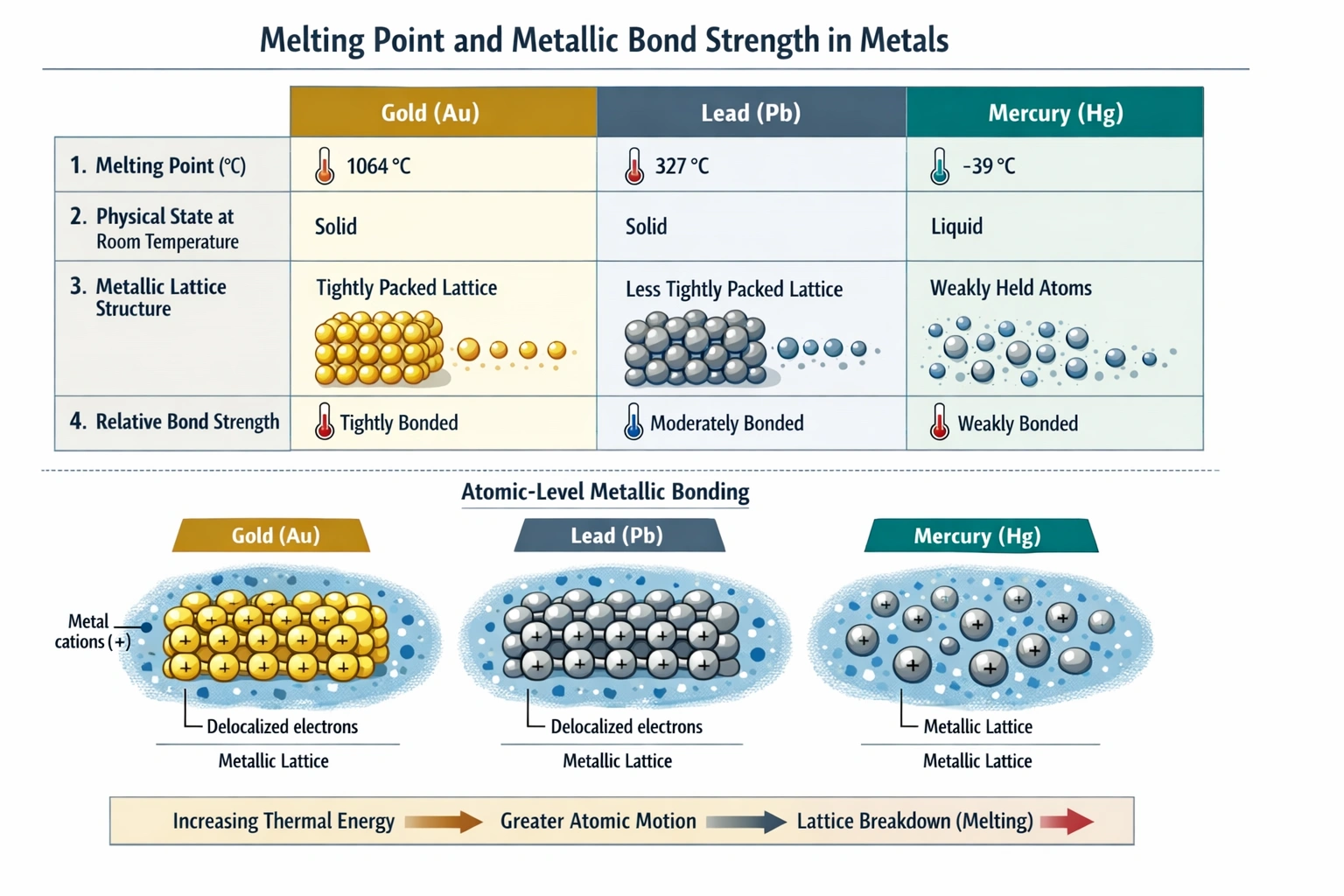
For metallic elements, the primary "intermolecular" force is the metallic bond a strong attraction between a lattice of positive metal ions and a delocalized "sea" of valence electrons. The strength of these metallic bonds is directly reflected in physical properties like melting point. A higher melting point indicates that more thermal energy is required to overcome these attractive forces and break the orderly metallic lattice.
A) The intermolecular forces are strongest between atoms of gold.
Gold has the highest melting point (1063°C) among the three, meaning it requires the most energy input to transition from solid to liquid. This high energy requirement is a direct consequence of the exceptionally strong metallic bonding in gold. Factors contributing to this strength include the number of delocalized electrons per atom, the charge density of the ions, and the lattice structure.
B) The intermolecular forces are strongest between atoms of mercury.
Mercury has the lowest melting point (-38.9°C), which is below room temperature, making it a liquid under standard conditions. This very low melting point indicates that the metallic bonds between mercury atoms are relatively weak. The weakness is often attributed to relativistic effects in its electron orbitals that make the 6s electrons less available for delocalized bonding. Stronger forces would require more energy to melt, which is not the case for mercury.
C) The intermolecular forces of these metals are stronger after they melt.
This is incorrect. Melting is the process of overcoming the intermolecular forces that hold the solid lattice together. When a metal melts, the rigid, long-range order of the solid is lost, and the ions gain mobility. While some metallic character persists in the liquid (which is why liquid metals conduct electricity), the cohesive forces are on average weaker than in the solid state. If the forces were stronger after melting, it would require energy to melt and then the liquid would be more stable, which contradicts the definition of melting as an endothermic process that destabilizes the solid structure.
D) The intermolecular forces of these metals get stronger as they are heated.
Intermolecular forces, such as metallic bond strength, are intrinsic properties of the substance based on electronic structure. They do not intensify with added heat. In fact, heating increases the kinetic energy of the particles, which increases the amplitude of their vibrations (in a solid) or their speed (in a liquid/gas). This increased motion works against the attractive forces, making it easier to separate particles. The forces themselves remain constant, but their ability to hold particles in an ordered arrangement diminishes as thermal energy increases.
 Conclusion:
Conclusion:
Melting point is a reliable comparative indicator of the strength of the forces holding particles together in the solid state. Since gold has the highest melting point, it logically follows that the metallic bonds (the intermolecular forces in this context) are strongest in gold.
Topic Flashcards
Click to FlipThe melting points of gold, lead, and mercury are 1063°C, 328°C, and -38.9°C respectively. Which metal has the strongest intermolecular (metallic) bonding forces?
Gold. The highest melting point indicates the strongest forces holding the metal lattice together.
What is the primary type of "intermolecular" force holding atoms together in a metallic solid like gold?
Metallic bonding (the attraction between positive metal ions and a delocalized sea of valence electrons).
Based on melting point alone, rank these metals in order of INCREASING strength of their metallic bonds: Mercury, Gold, Lead.
Mercury (weakest), Lead, Gold (strongest).
Mercury is a liquid at room temperature (25°C). What does this directly imply about the strength of its metallic bonds compared to other metals?
Its metallic bonds are relatively weak, as less thermal energy is needed to overcome them.
Why does gold require so much more heat energy to melt than lead or mercury?
Because the metallic bonds between gold atoms are significantly stronger and require more energy to break.