Which of the following parts of an atom has the largest mass?
-
A
A proton
-
B
A neutron
-
C
The nucleus
-
D
The electron cloud
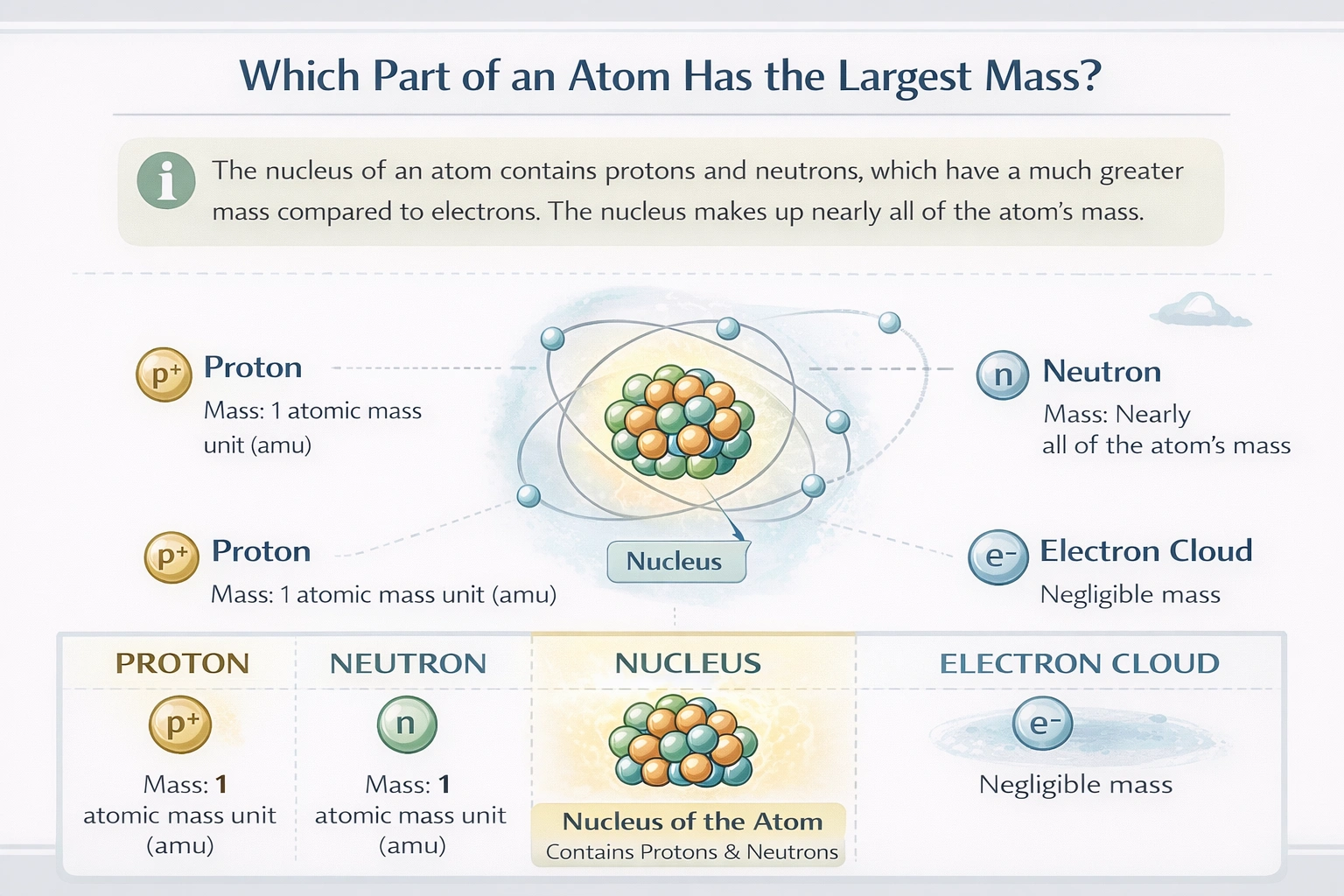
The nucleus of an atom contains the largest mass.
The mass of an atom is concentrated almost entirely in its nucleus, the dense, central core. The nucleus is composed of protons and neutrons, collectively called nucleons. Each nucleon has a mass approximately 1,836 times greater than that of an electron. Since the nucleus aggregates all of an atom's protons and neutrons, its collective mass represents over 99.9% of the atom's total mass, despite occupying a minuscule fraction of the atom's volume.
A) A proton
A single proton has a mass of about 1.6726 × 10⁻²⁷ kg, defined as roughly 1 atomic mass unit (amu). While it is a massive particle compared to an electron, it constitutes only one unit of the atom's total mass. An atom contains multiple protons, and their combined mass, along with that of the neutrons, is what gives the nucleus its dominant mass.
B) A neutron
A single neutron has a mass slightly greater than a proton, approximately 1.6749 × 10⁻²⁷ kg (~1 amu). Like the proton, it is a fundamental massive particle. However, as an individual component, its mass is less than the combined mass of all nucleons within the nucleus.
C) The nucleus
The nucleus is the structure that contains all the atom's protons and neutrons. For example, a carbon-12 atom has a nucleus with 6 protons and 6 neutrons. The combined mass of these 12 nucleons constitutes essentially the entire mass of the carbon atom. The nucleus, as the aggregate of these particles, therefore contains the greatest mass of any part of the atom.
D) The electron cloud
The electron cloud refers to the region surrounding the nucleus where electrons are probabilistically located. Electrons have a very small mass, about 9.109 × 10⁻³¹ kg, which is only about 1/1836 amu each. Although electrons occupy most of the atom's volume and there are as many electrons as protons in a neutral atom, their total combined mass is negligible compared to the mass of the nucleus.
Conclusion:
The distribution of mass within an atom is extremely asymmetrical. The relatively heavy protons and neutrons are packed into the tiny, dense nucleus, while the very light electrons are dispersed in the vast space around it. Consequently, when considering the component parts of an atom, the nucleus—as the collective repository of all nucleons—possesses the overwhelmingly largest portion of the atom's total mass.
Topic Flashcards
Click to FlipWhere is over 99.9% of an atom's total mass concentrated?
In the nucleus (which contains protons and neutrons).
Which is heavier: a single proton or a single electron, and by approximately what factor?
A proton is about 1,836 times heavier than an electron.
Why does the electron cloud contribute almost nothing to an atom's total mass, even though it occupies most of the atom's volume?
Because each electron's mass is extremely small (about 1/1836 the mass of a proton or neutron).
What two subatomic particles, found in the nucleus, are collectively called "nucleons"?
Protons and neutrons.
If you could weigh a nucleus and the surrounding electron cloud of the same atom separately, which would have the larger reading on the scale?
The nucleus would have a vastly larger reading (mass).