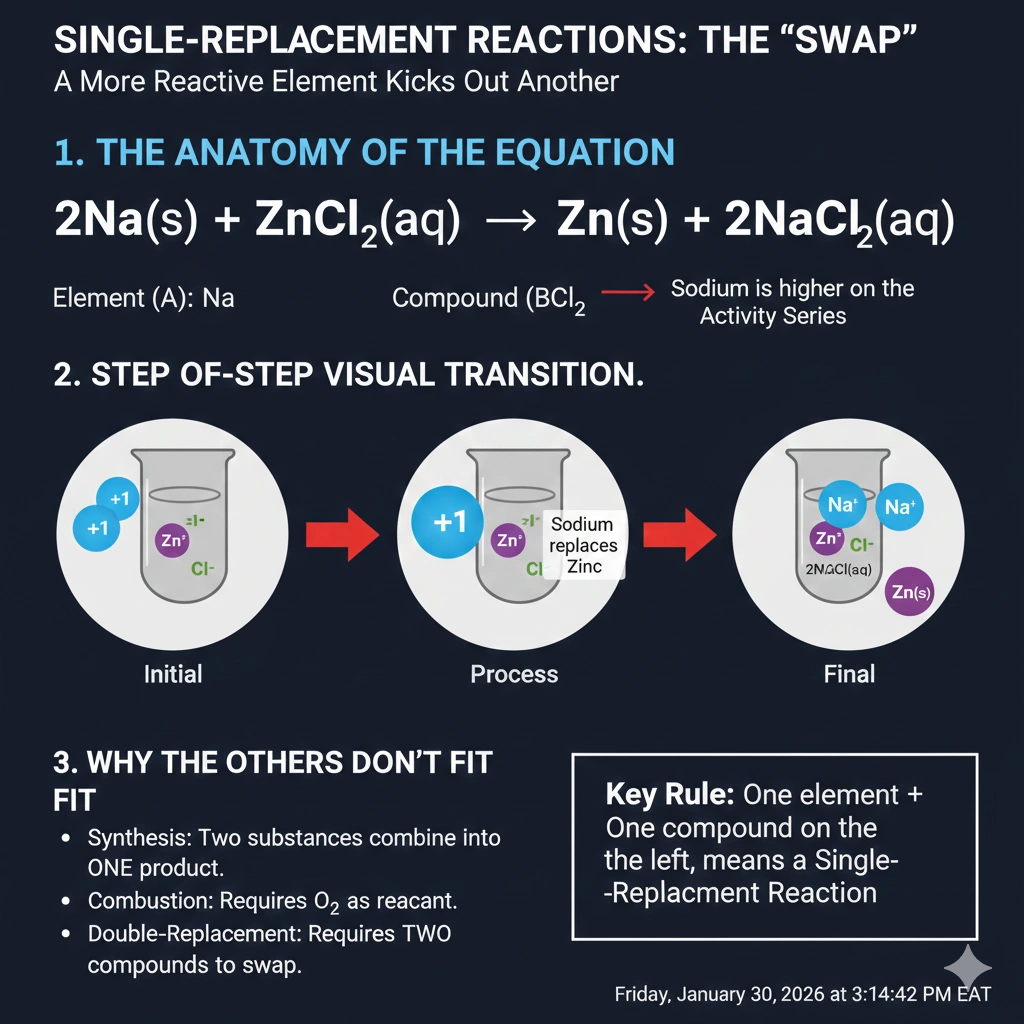
What type of reaction is described by the following equation? 2Na(s) + ZnCl₂(aq) → Zn(s) + 2NaCl(aq)
-
A
Synthesis
-
B
Combustion
-
C
Single-replacement
-
D
Double-replacement
The reaction is a single-replacement reaction.
Classifying chemical reactions involves recognizing patterns in how reactants rearrange to form products. Single-replacement reactions feature an element reacting with a compound, where the element displaces another element in that compound.
A) Synthesis
A synthesis (or combination) reaction involves two or more substances combining to form a single, more complex product (A + B → AB). This reaction has two reactants and two products, so it does not fit the pattern of synthesis. Synthesis reactions typically have fewer product substances than reactant substances.
B) Combustion
A combustion reaction involves a substance (often a hydrocarbon) reacting rapidly with oxygen gas (O₂) to produce energy, carbon dioxide, and water. Oxygen is not a reactant in this equation, and the products are a metal and a salt, not oxides. Therefore, it is not a combustion reaction.
C) Single-replacement
A single-replacement (or single-displacement) reaction occurs when one element replaces another element in a compound. The general form is A + BC → AC + B. In this equation, solid sodium (Na) replaces zinc (Zn) in the compound zinc chloride (ZnCl₂). Sodium, being more reactive than zinc, displaces it from the compound. The products are solid zinc metal and aqueous sodium chloride (NaCl). This fits the single-replacement pattern perfectly.
D) Double-replacement
A double-replacement reaction involves the exchange of ions between two compounds, typically in aqueous solution. The general form is AB + CD → AD + CB. In this reaction, one of the reactants (Na) is an elemental solid, not a compound. Therefore, an ion exchange between two compounds cannot occur, ruling out a double-replacement reaction.
Conclusion:
The reaction features an elemental reactant (Na) displacing the metal in a compound (ZnCl₂) to form a new compound and a new elemental solid. This is the hallmark of a single-replacement reaction.
Topic Flashcards
Click to FlipWhat is the general pattern (using letters A, B, and C) for a single-replacement reaction?
A + BC → AC + B, where element A replaces element B in the compound.
In the reaction 2Na(s) + ZnCl₂(aq) → Zn(s) + 2NaCl(aq), which element is being replaced and which is the replacing element?
Zinc (Zn) is the element being replaced. Sodium (Na) is the more active element doing the replacing.
Why can this reaction not be classified as a double-replacement reaction?
One of the reactants (Na) is an elemental solid, not an ionic compound. Double-replacement requires two compounds to exchange ions.
What happens to the Na atom and the Zn²⁺ ion in this reaction in terms of electron transfer?
Each Na atom loses one electron to become Na⁺. Each Zn²⁺ ion gains two electrons (from two Na atoms) to become Zn metal.
How does the reactivity series of metals help predict if this single-replacement reaction will occur?
Sodium (Na) is higher on the reactivity series than zinc (Zn). A more reactive metal can displace a less reactive metal from its compound.