Extract:
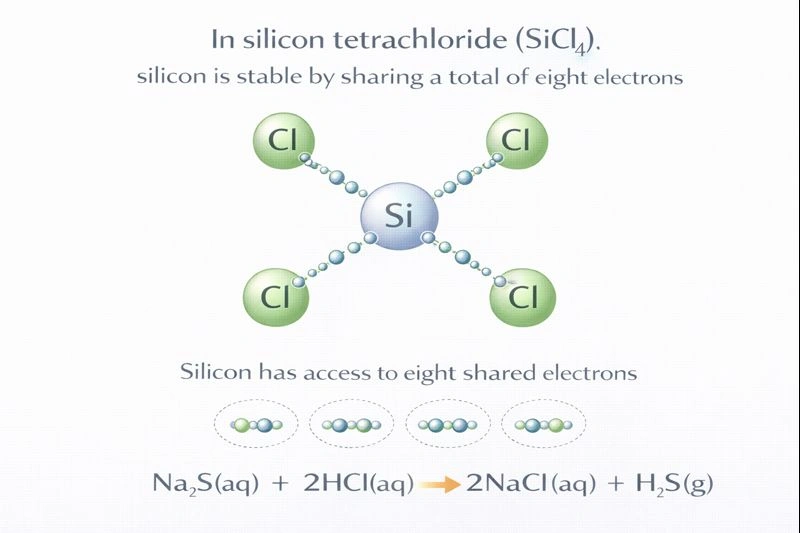
In the compound silicon tetrachloride (SiCl₄) shown below, how is silicon stable?
-
A
Silicon shares a total of four electrons.
-
B
Silicon shares a total of eight electrons.
-
C
Silicon gives eight electrons to surrounding chlorine atoms.
-
D
Silicon removes four electrons from surrounding chlorine atoms.
In silicon tetrachloride (SiCl₄), silicon achieves stability by sharing a total of eight electrons.
Silicon (Si) is in group 14 of the periodic table and has four valence electrons. Chlorine (Cl) is in group 17 and has seven valence electrons. To achieve a stable electron configuration, atoms often follow the octet rule, seeking eight electrons in their valence shell. In SiCl₄, silicon forms four single covalent bonds with four chlorine atoms. In each Si-Cl bond, two electrons are shared—one contributed by silicon and one by chlorine. Therefore, silicon is involved in four bonding pairs, which means it is surrounded by and shares a total of eight electrons (4 bonds × 2 electrons/bond), satisfying the octet rule.
A) Silicon shares a total of four electrons.
This statement undercounts the shared electrons. In covalent bonding, a "shared" pair consists of two electrons. If silicon shared only four electrons total, that would imply only two shared pairs (four electrons), or an average of one electron per bond. In reality, each bond involves a pair of shared electrons, so four bonds involve eight shared electrons from silicon's perspective (it "sees" eight electrons in its bonding sphere).
B) Silicon shares a total of eight electrons.
This is accurate. Each of the four covalent bonds utilizes one of silicon's four valence electrons. Through these bonds, silicon gains shared access to one electron from each of the four chlorine atoms. This gives silicon access to 4 (its own) + 4 (from Cl) = 8 electrons associated with its valence shell, fulfilling the octet.
C) Silicon gives eight electrons to surrounding chlorine atoms.
This describes an ionic transfer of electrons, not covalent sharing. Silicon does not have a strong enough tendency to lose four electrons (which would require considerable energy to form Si⁴⁺), nor would chlorine atoms accept two electrons each to form Cl²⁻. Silicon tetrachloride is a covalent molecular compound, not an ionic one.
D) Silicon removes four electrons from surrounding chlorine atoms.
This also describes an ionic process but in reverse, with silicon acting as an electron acceptor. Silicon is not electronegative enough to remove electrons from chlorine; in fact, chlorine is more electronegative (3.16 vs. Si's 1.90). In the Si-Cl bond, the shared electrons are pulled closer to the chlorine atoms, making the bonds polar covalent with a partial positive charge on silicon and partial negative charges on chlorine.
Conclusion:
Silicon tetrachloride is a covalent compound where silicon achieves a stable octet by forming four single covalent bonds. In each bond, two electrons are shared. Consequently, silicon is at the center of four bonding pairs, which corresponds to it being surrounded by and sharing a total of eight electrons. This satisfies the octet rule and provides electronic stability.
Topic Flashcards
Click to FlipHow many valence electrons does a silicon atom have, and how many more does it need to satisfy the octet rule?
Silicon has 4 valence electrons and needs 4 more to complete an octet of 8.
In a molecule of SiCl₄, how many single covalent bonds does the silicon atom form, and with which element?
Silicon forms four single covalent bonds, each with a chlorine atom.
What is the total number of electrons that silicon is "surrounded by" or effectively "sees" in its valence shell in SiCl₄?
Eight electrons (four bonding pairs).
Explain why the bonding in SiCl₄ is covalent rather than ionic, referencing the elements' electronegativities
The electronegativity difference between Si (~1.90) and Cl (~3.16) is not large enough for a complete electron transfer; instead, they share electrons.
What is the molecular geometry (shape) of the SiCl₄ molecule, and how does this relate to the number of electron pairs around silicon?
Tetrahedral geometry, because four bonding pairs of electrons repel each other equally.