The difference between hydrochloric acid and hydro sulphuric acid is _______ .
-
A
that only the first is binary
-
B
that only the first is ternary
-
C
the presence of hydrogen
-
D
the nonmetallic elements in each
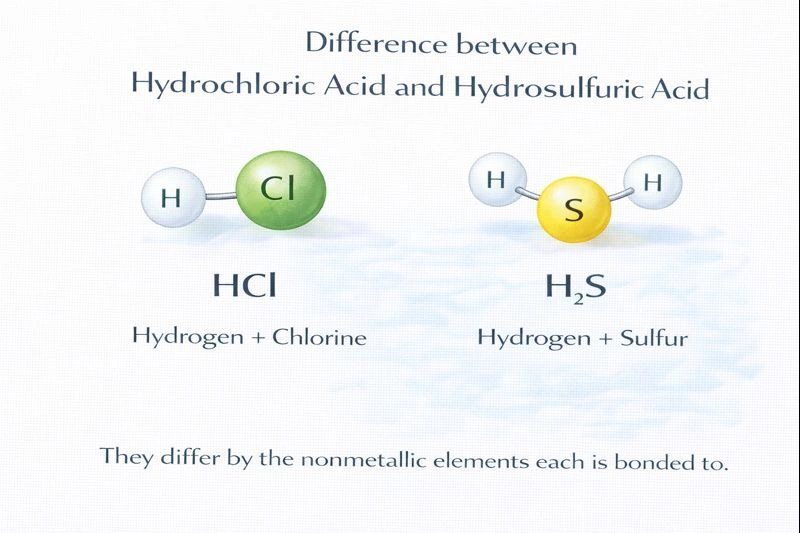
The difference between hydrochloric acid and hydro sulphuric acid is the nonmetallic elements in each.
Hydrochloric acid and hydro sulphuric acid are both binary acids, meaning they are composed of hydrogen and one other nonmetal. While they share this general classification, they differ in the specific nonmetal bonded to hydrogen. This difference affects their chemical behavior, strength, and properties.
A. That only the first is binary
Hydrochloric acid (HCl) consists of hydrogen and chlorine, making it a binary acid. Hydro sulphuric acid (H₂S) consists of hydrogen and sulphur and is also a binary acid. Since both contain only hydrogen and one other nonmetal, neither is unique in this respect.
B. That only the first is ternary
Ternary acids, also called oxyacids, contain hydrogen, oxygen, and another element. Examples include sulphuric acid (H₂SO₄) or nitric acid (HNO₃). Neither hydrochloric acid nor hydro sulphuric acid contains oxygen, so neither is ternary.
C. The presence of hydrogen
Hydrogen is present in both acids and is a defining feature of all acids under the Arrhenius definition. Therefore, hydrogen does not distinguish one from the other.
D. The nonmetallic elements in each
This is correct. Hydrochloric acid contains chlorine as its nonmetal component, whereas hydro sulphuric acid contains sulphur. The identity of the nonmetal determines the nature of the anion formed in solution (chloride versus sulfide) and leads to differences in acidity, odor, and chemical reactivity.
Conclusion
Both hydrochloric acid and hydro sulphuric acid are binary acids containing hydrogen, but they differ in the nonmetal element bonded to hydrogen. Therefore, the correct answer is D.
Topic Flashcards
Click to FlipWhat is the definition of a binary acid?
An acid composed of only two elements: hydrogen and one other nonmetal.
What are the full chemical formulas and corresponding nonmetal elements for hydrochloric acid and hydrosulphuric acid?
Hydrochloric acid: HCl (nonmetal = Chlorine, Cl). Hydrosulphuric acid: H₂S (nonmetal = Sulfur, S).
What is a ternary (or oxyacid), and why does this term NOT apply to either HCl or H₂S?
A ternary acid contains hydrogen, oxygen, and a third element. Neither HCl nor H₂S contains oxygen, so they are not ternary.
Why is option C ("the presence of hydrogen") an incorrect differentiator between HCl and H₂S?
Hydrogen is the acidic component present in all Arrhenius acids, including both HCl and H₂S, so it is not a distinguishing feature.
Using the naming rules for binary acids, how would you name an acid with the formula HBr(aq)?
Hydrobromic acid. Binary acids in aqueous solution use the prefix "hydro-" and the suffix "-ic."