Extract:
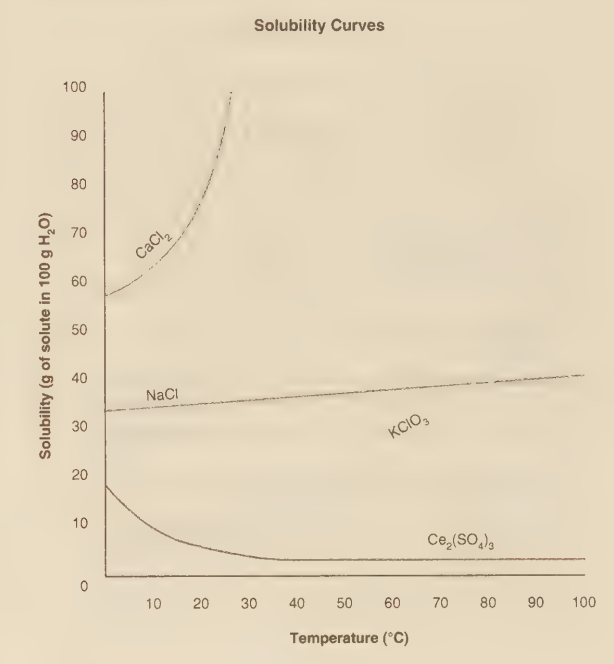
What is the maximum amount of KClO₃ that can be dissolved in 50 grams of water at 30°C?
-
A
5 grams
-
B
10 grams
-
C
20 grams
-
D
70 grams
The maximum amount of KClO₃ that can be dissolved in 50 grams of water at 30°C is 5 grams.
Solubility is defined as the maximum mass of solute that can dissolve in a given mass of solvent at a specified temperature to form a saturated solution. Solubility values are often given per a standard solvent mass (usually 100 g). A proportional calculation is needed when the solvent mass differs from this standard.
A) 5 grams
The graph indicates the solubility of KClO₃ at 30°C is 10 g per 100 g of water. This is a concentration: 10g KClO₃ / 100g H₂O. To find how much dissolves in 50 g of water (half the standard amount), we take half the solute mass: (10 g / 2) = 5 g. Mathematically, it's a ratio: (10 g / 100 g) = (X g / 50 g) -> X = (10 * 50) / 100 = 5 g.
B) 10 grams
This is the solubility in 100 g of water, not 50 g. Selecting this answer ignores the specified mass of the solvent and mistakenly uses the solubility value as the final answer without proportionally scaling it down.
C) 20 grams
This would be the amount for 200 g of water (double the standard amount). It results from mistakenly doubling the solubility value or from a misreading of the graph or question.
D) 70 grams
This value is implausibly high for KClO₃ at 30°C. It might correspond to the solubility of a very soluble salt like sodium nitrate (NaNO₃) at a high temperature, or it could stem from a severe misreading of the graph's scale.
Conclusion:
Solubility is an intensive property, but the total amount dissolved depends on the amount of solvent. Using the given solubility data (10g/100g H₂O) and applying it proportionally to 50 g of solvent yields a maximum dissolved mass of 5 grams of KClO₃.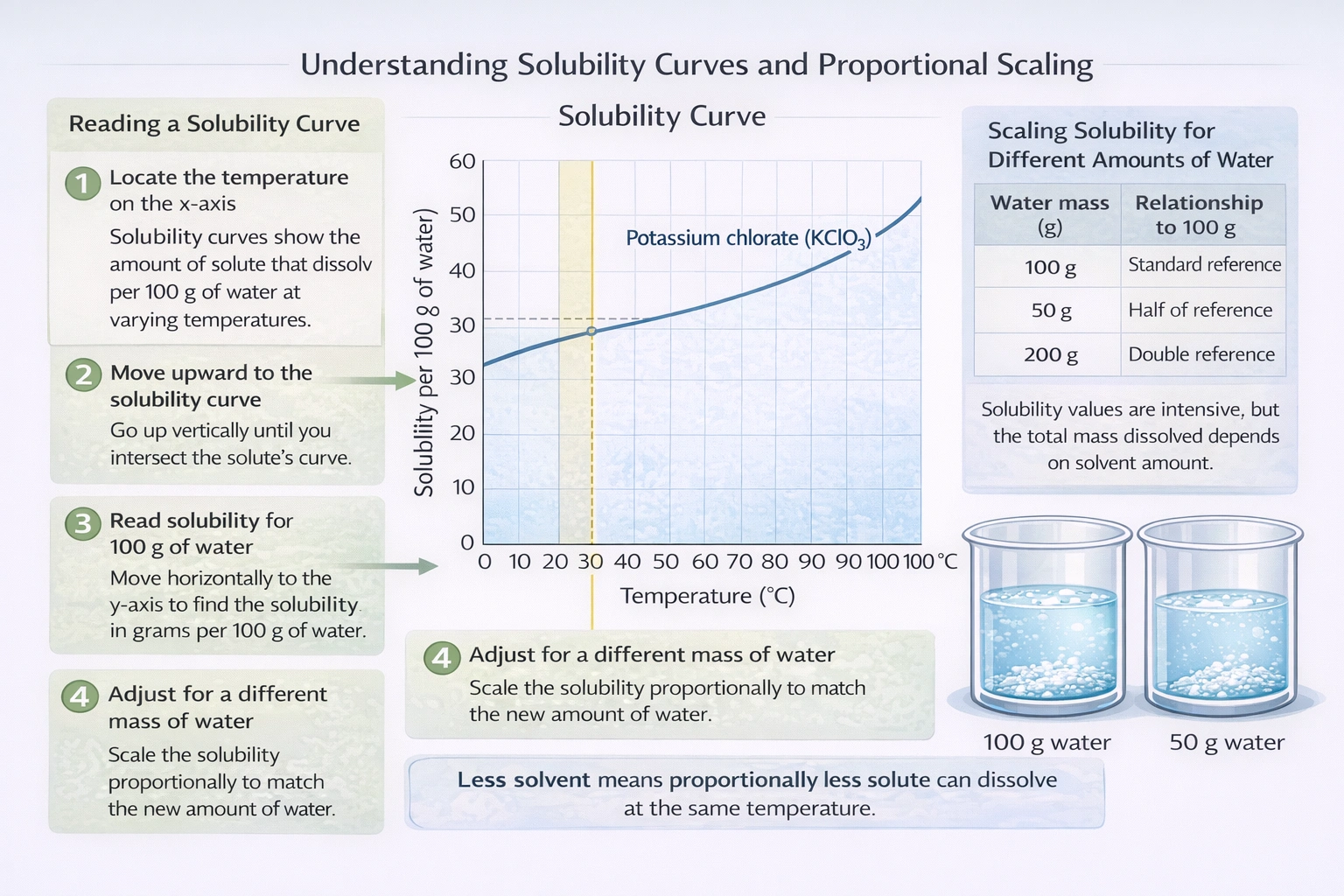
Topic Flashcards
Click to FlipWhat is the definition of solubility for a solid in water?
The maximum mass of solute that can dissolve in a given mass of solvent (usually 100 g) at a specified temperature to form a saturated solution.
If the solubility of KClO₃ is 10 g per 100 g of water at 30°C, how much can dissolve in 50 g of water at the same temperature?
5 grams. Calculation: (10 g / 100 g) × 50 g = 5 g.
How do you calculate the mass of solute that can dissolve in a different mass of solvent when you know the solubility per 100 g?
Use a proportion: (Solubility in g / 100 g solvent) = (X g / Given solvent mass). Then solve for X.
Using the same solubility data (10 g/100 g H₂O at 30°C), what is the maximum mass of KClO₃ that can dissolve in 200 g of water?
20 grams. (10 g/100 g) × 200 g = 20 g.