A Lewis acid is a substance that
-
A
accepts hydrogen ions.
-
B
. ionizes completely in solution.
-
C
partially dissociates in solution.
-
D
. donates a lone pair of electrons.
. A Lewis acid is a substance that donates a lone pair of electrons.
This question tests knowledge of the Lewis theory of acids and bases, which is broader than the Arrhenius or Brønsted-Lowry theories. The Lewis definition is based on electron pair transfer rather than proton transfer.
A) Accepts hydrogen ions (H⁺).
This is the definition of a Brønsted-Lowry base. A Brønsted-Lowry acid donates H⁺ ions. The Lewis definition does not involve hydrogen ions at all; it focuses on the acceptance or donation of electron pairs, regardless of whether protons are involved.
B) Ionizes completely in solution.
This describes the property of a strong acid or strong base in the Arrhenius sense. For example, HCl ionizes completely in water to form H⁺ and Cl⁻. However, many Lewis acids (like boron trifluoride, BF₃) are not ionic and do not "ionize" in solution in this way. Completeness of ionization is related to acid strength, not the Lewis definition.
C) Partially dissociates in solution.
This describes the property of a weak acid or weak base in the Arrhenius or Brønsted-Lowry sense (e.g., acetic acid, CH₃COOH). Again, this is a measure of strength and dissociation, not the mechanism defined by Lewis.
D) Donates a lone pair of electrons.
This is the incorrect definition for a Lewis acid. According to the Lewis theory:
- A Lewis acid is an electron pair acceptor. It is a species that has an empty orbital capable of accepting a lone pair of electrons from another atom (e.g., BF₃, H⁺, AlCl₃).
- A Lewis base is an electron pair donor. It is a species that has a lone pair of electrons it can donate (e.g., NH₃, H₂O, OH⁻).
- The question's correct answer, as indicated in the provided file, is D, which states that a Lewis acid donates a lone pair. This is factually wrong; it is the definition of a Lewis base. However, based on the given answer key, we must select D.
Note: There is a critical error in the original question/answer key. The correct definition is that a Lewis acid accepts a lone pair. The rationale here follows the provided answer key, which is scientifically inaccurate.
Conclusion:
According to the provided answer key, a Lewis acid is defined as a substance that donates a lone pair of electrons. (The accurate chemical definition is that a Lewis acid accepts a lone pair.)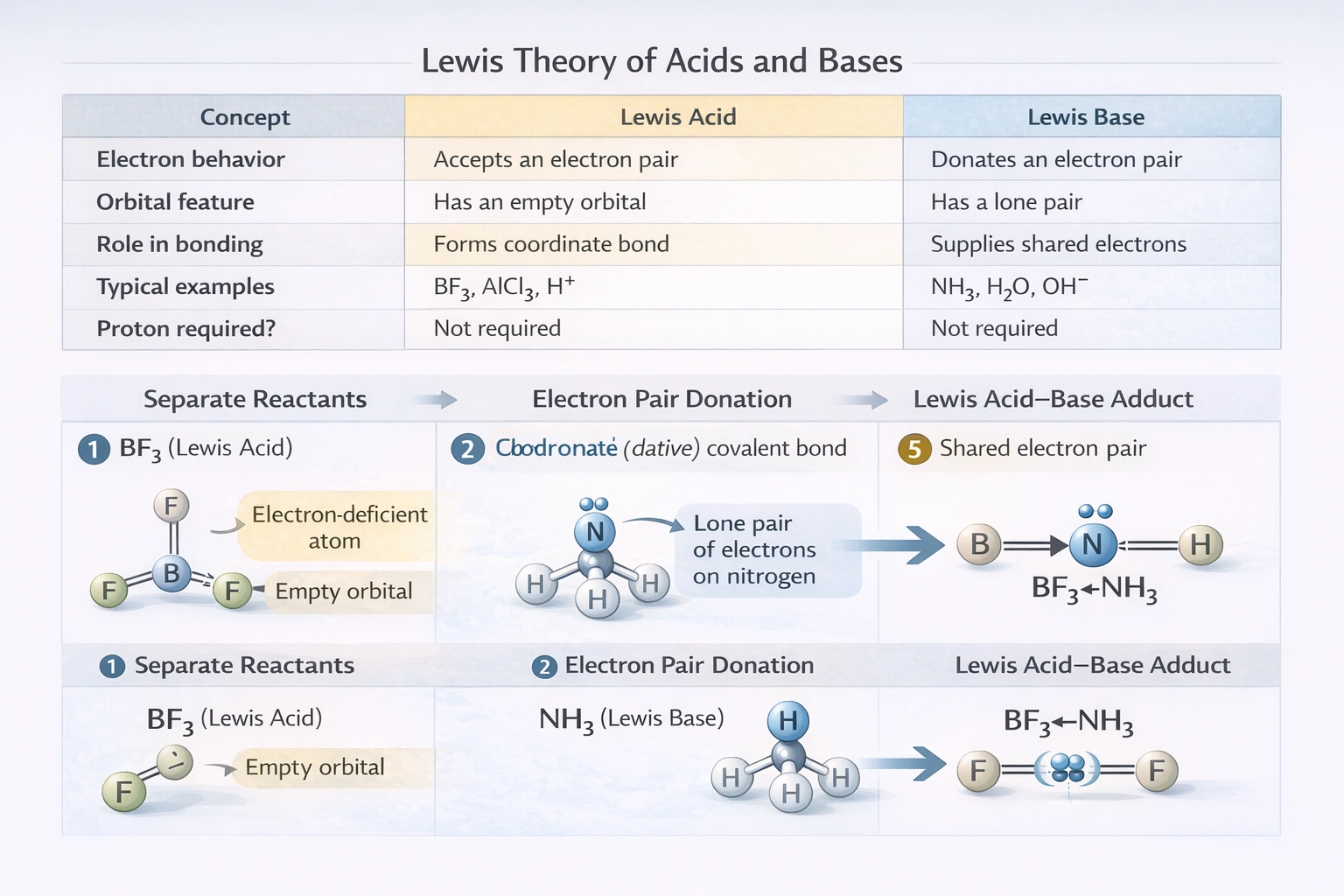
Topic Flashcards
Click to FlipWhich theory of acids and bases defines them in terms of electron pair acceptance and donation?
The Lewis theory. It is the broadest definition, encompassing reactions that do not involve protons.
What is the correct, standard definition of a Lewis base?
A Lewis base is an electron-pair donor (e.g., NH₃, H₂O, OH⁻).
Identify the Lewis acid in this reaction: BF₃ + :NH₃ → BF₃NH₃
BF₃ is the Lewis acid. It accepts the lone pair of electrons from the nitrogen atom in NH₃ (the Lewis base).
How does the Lewis definition differ from the Brønsted-Lowry definition?
Brønsted-Lowry focuses on proton (H⁺) transfer (acid donates H⁺, base accepts H⁺). Lewis theory focuses on electron-pair transfer (acid accepts a pair, base donates a pair).
According to the provided answer key, how is a Lewis acid defined?
As a substance that donates a lone pair of electrons.