Energy levels, electron configuration, groups
-
A
The oxygen atoms share electrons equally.
-
B
One oxygen atom pulls harder on shared electrons than the other.
-
C
One atom of oxygen loses two electrons, while the other gains two electrons.
-
D
There is not enough information to describe the bond in a molecule of oxygen
The oxygen atoms share electrons equally.
The bond in a diatomic oxygen molecule (O₂) is a covalent bond formed between two identical atoms. The nature of this bond is determined by the electronegativity of the bonding atoms.
A) The oxygen atoms share electrons equally.
This is correct. Oxygen atoms have identical electronegativity—the ability to attract bonding electrons. Because there is no difference in electronegativity, the electron pair in the covalent bond is shared equally between the two oxygen nuclei. This type of bond is specifically called a nonpolar covalent bond. The molecule is symmetrical, and the electron cloud is evenly distributed.
B) One oxygen atom pulls harder on shared electrons than the other.
This would imply a difference in electronegativity, leading to a polar covalent bond where the electron cloud is skewed toward one atom. In a molecule composed of two atoms of the same element, their electronegativities are by definition identical. Neither atom can exert a greater pull on the shared electrons than the other. This statement is false for any homonuclear diatomic molecule like O₂, N₂, H₂, etc.
C) One atom of oxygen loses two electrons, while the other gains two electrons.
This describes the formation of an ionic bond, which involves the complete transfer of one or more electrons from one atom to another. This occurs between atoms with a very large difference in electronegativity, typically a metal and a nonmetal. For two oxygen atoms, such a transfer is energetically highly unfavorable. Both atoms need to gain electrons to achieve stability; having one oxygen atom lose electrons would leave it with a highly unstable electron configuration. Oxygen atoms achieve stability by sharing electrons, not transferring them.
D) There is not enough information to describe the bond in a molecule of oxygen.
This is false. The information given—that the molecule is O₂—is sufficient. Knowing that the molecule consists of two atoms of the same element allows us to conclude definitively that the bond must be a nonpolar covalent bond formed by equal sharing of electrons. This is a fundamental principle in chemistry.
Conclusion:
In any molecule composed of two identical atoms, the bond is a nonpolar covalent bond due to the equal electronegativity of the atoms. Therefore, the two oxygen atoms in O₂ share their bonding electrons equally.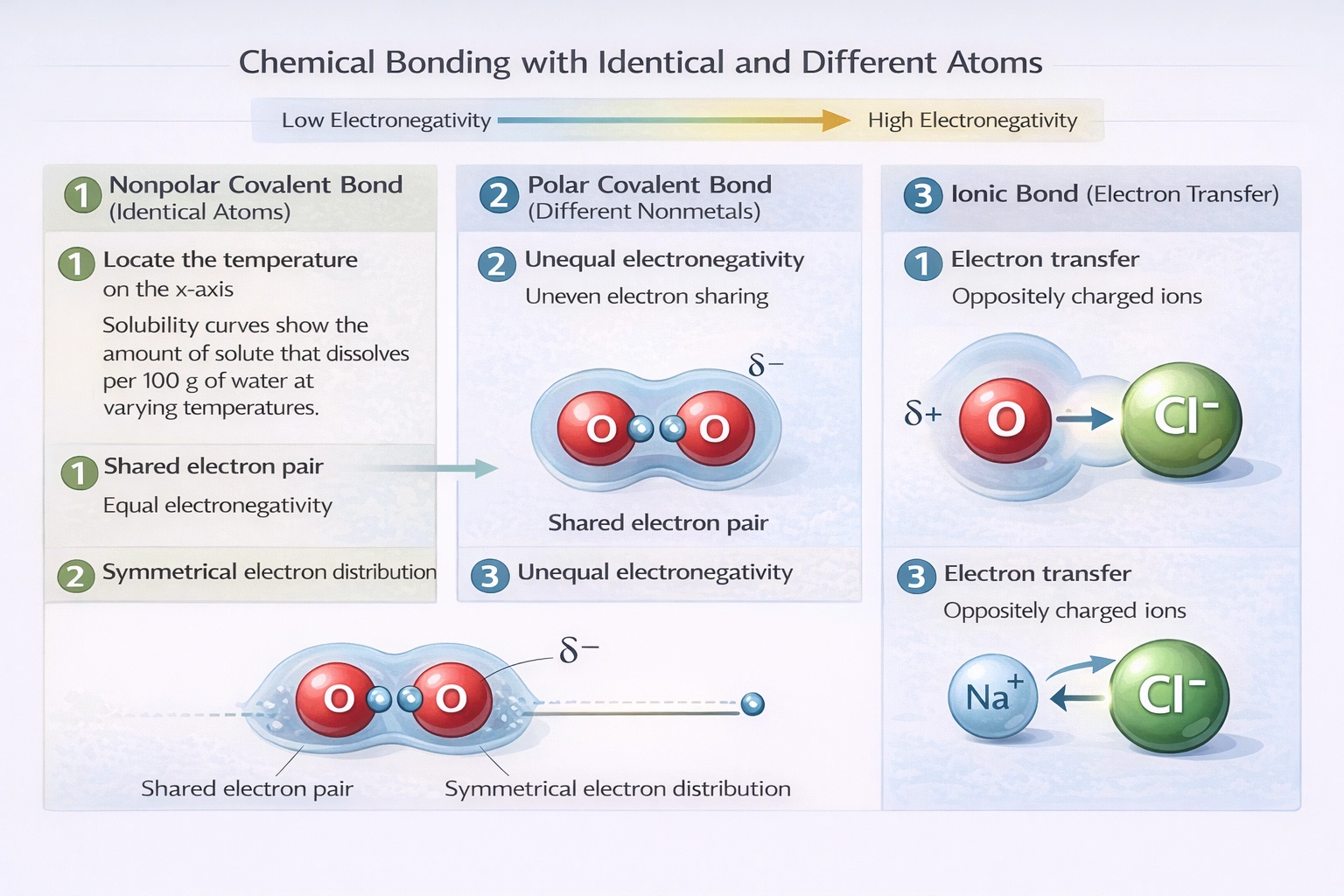
Topic Flashcards
Click to FlipWhat specific type of bond is found in a molecule of O₂ (two oxygen atoms), and why?
A nonpolar covalent bond. Because the two identical oxygen atoms have equal electronegativity, they share the bonding electrons equally.
What property of two bonding atoms determines whether a covalent bond is polar or nonpolar?
The difference in their electronegativity. If the atoms are the same (difference = 0), the bond is nonpolar. If they are different, the bond is polar.
Why is it impossible for one oxygen atom to lose electrons to another to form an ionic O₂ molecule?
Both oxygen atoms are high-energy, nonmetals that strongly attract electrons (high electronegativity). Neither wants to lose electrons; they both achieve stable configurations by sharing.
In a diatomic molecule like O₂, N₂, or H₂, how is the bonding electron cloud distributed between the two atoms?
It is distributed equally or symmetrically. Since the atoms are identical, the shared electrons spend equal time around each nucleus.
How does the bond in an O₂ molecule differ from the bond in an H₂O molecule?
O₂ has a nonpolar covalent bond (O-O). H₂O has polar covalent bonds (O-H) because oxygen is much more electronegative than hydrogen, pulling the shared electrons closer to itself.