The specific heat capacity of tin is 0.217 J/(g°C). Which material would require about twice as much heat as tin to raise a sample by 1°C?
-
A
Copper [0.3844 J/(g°C)]
-
B
Iron [0.449 J/(g°C)]
-
C
Gold [0.1291 J/(g°C)]
-
D
Aluminum [0.904 J/(g°C)]
The material that would require about twice as much heat as tin to raise a sample by 1 °C is aluminum.
Specific heat capacity measures how much heat energy is required to raise the temperature of one gram of a substance by 1 °C. Tin has a relatively low specific heat capacity (0.217 J/(g·°C)), so a material with a significantly higher value would require considerably more heat for the same temperature change. Comparing the given values shows which substance best fits the idea of requiring roughly twice (or more) the heat compared with tin.
A) Copper [0.3844 J/(g·°C)]
Copper’s specific heat is higher than that of tin and would require more heat for the same temperature increase. However, its value is only about 1.8 times that of tin, which is somewhat below the idea of “about twice” when compared with the other options.
B) Iron [0.449 J/(g·°C)]
Iron requires a little more than twice as much heat as tin for the same mass and temperature change. While this is close to double, it is still notably lower than aluminum’s capacity to absorb heat.
C) Gold [0.1291 J/(g·°C)]
Gold has a lower specific heat capacity than tin. This means it would require less heat, not more, to raise its temperature by 1 °C, making it unsuitable for the comparison.
D) Aluminum [0.904 J/(g·°C)]
Aluminum has a much higher specific heat capacity than tin. It requires well over twice the amount of heat needed by tin to raise the same mass by 1 °C. This makes aluminum the best choice when identifying a material that clearly requires about twice as much heat (or more) compared with tin.
Conclusion
Comparing specific heat capacities shows that aluminum absorbs far more heat per gram per degree than tin. Among the choices, aluminum best matches the requirement of needing about twice as much heat to raise a sample by 1 °C.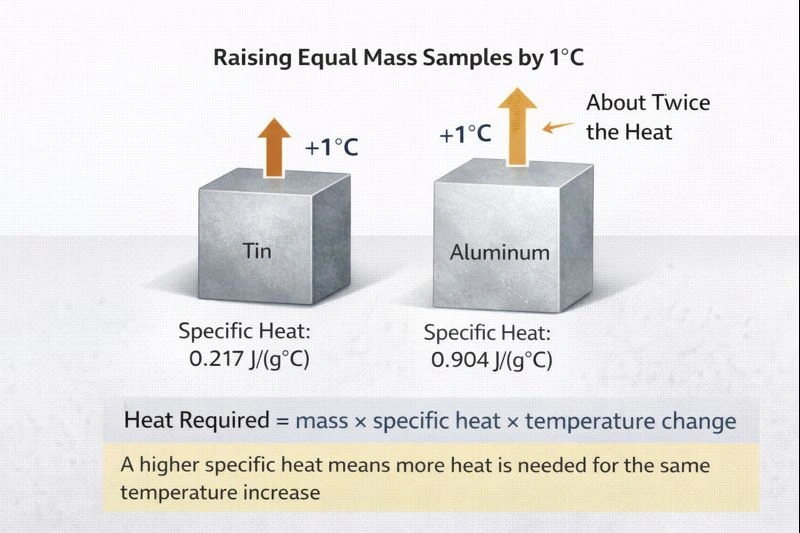
Topic Flashcards
Click to FlipDefine specific heat capacity and state its standard SI units.
Specific heat capacity (c) is the amount of heat energy required to raise the temperature of 1 gram of a substance by 1 degree Celsius (or 1 Kelvin). Its units are J/(g·°C) or equivalently J/(g·K).
How much heat energy is required to raise the temperature of a 50 g sample of tin by 10°C? (Use c_tin = 0.217 J/(g·°C))
Q = m * c * ΔT = 50 g * 0.217 J/(g·°C) * 10°C = 108.5 J.
If you have equal masses of tin and aluminum at the same temperature and add the same amount of heat to each, which material will have a higher final temperature, and why?
Tin will have a higher final temperature. Since tin has a lower specific heat capacity (0.217 vs. 0.904 J/(g·°C)), the same amount of heat will cause a larger temperature increase (ΔT = Q/(m*c)).
Based on the given values, the specific heat of aluminum (0.904 J/(g·°C)) is about 4.17 times that of tin. How many times more heat would aluminum require to raise an equal mass by the same temperature?
It requires about 4.17 times more heat. This is because Q ∝ c when m and ΔT are fixed. (0.904 / 0.217 ≈ 4.17).
Explain a practical consequence of aluminum's high specific heat capacity compared to iron, using the idea of heating or cooling rates.
An aluminum pot heats up more slowly than an iron pot of the same mass on the same stove because it requires more energy input per degree of temperature increase. Conversely, it also cools down more slowly, retaining heat longer.