When the heat of a reaction is negative, which statement is true?
-
A
The products have less energy and are less stable.
-
B
The products have more energy and are more stable.
-
C
The products have less energy and are more stable.
-
D
The products have more energy and are less stable.
When reaction heat is negative, the products possess less energy and greater stability than the reactants because exothermic processes release thermal energy to the surroundings. This energy reduction corresponds to stronger bonding or more favorable molecular configurations that resist disruption, defining enhanced thermodynamic stability.
A) The products have less energy and are less stable.
Lower energy states correlate with greater stability, not lesser stability. Systems naturally evolve toward minimum energy configurations because these states resist perturbation. Claiming lower-energy products are less stable contradicts fundamental thermodynamic principles governing molecular stability.
B) The products have more energy and are more stable.
Exothermic reactions release energy, meaning products must possess less energy than reactants, not more. Higher-energy states are inherently less stable because they can spontaneously release energy to reach lower configurations. This option incorrectly associates energy increase with both exothermicity and stability.
C) The products have less energy and are more stable.
Negative ΔH signifies energy release during reaction, proving products possess less internal energy than reactants. Lower energy states exhibit greater stability because stronger bonds or more favorable arrangements resist disruption. This combination, reduced energy with enhanced stability, defines exothermic reaction outcomes.
D) The products have more energy and are less stable.
This describes endothermic reactions (ΔH > 0), not exothermic ones. Energy absorption increases product energy relative to reactants, creating less stable configurations that may spontaneously revert. Negative reaction heat cannot accompany energy gain, it fundamentally requires energy loss from the system.
Conclusion
Negative reaction heat identifies exothermic processes where products occupy lower energy states than reactants, resulting in enhanced molecular stability through stronger bonding or more favorable configurations. This energy-stability relationship explains why exothermic reactions often proceed spontaneously, systems naturally evolve toward minimum energy, maximum stability conditions.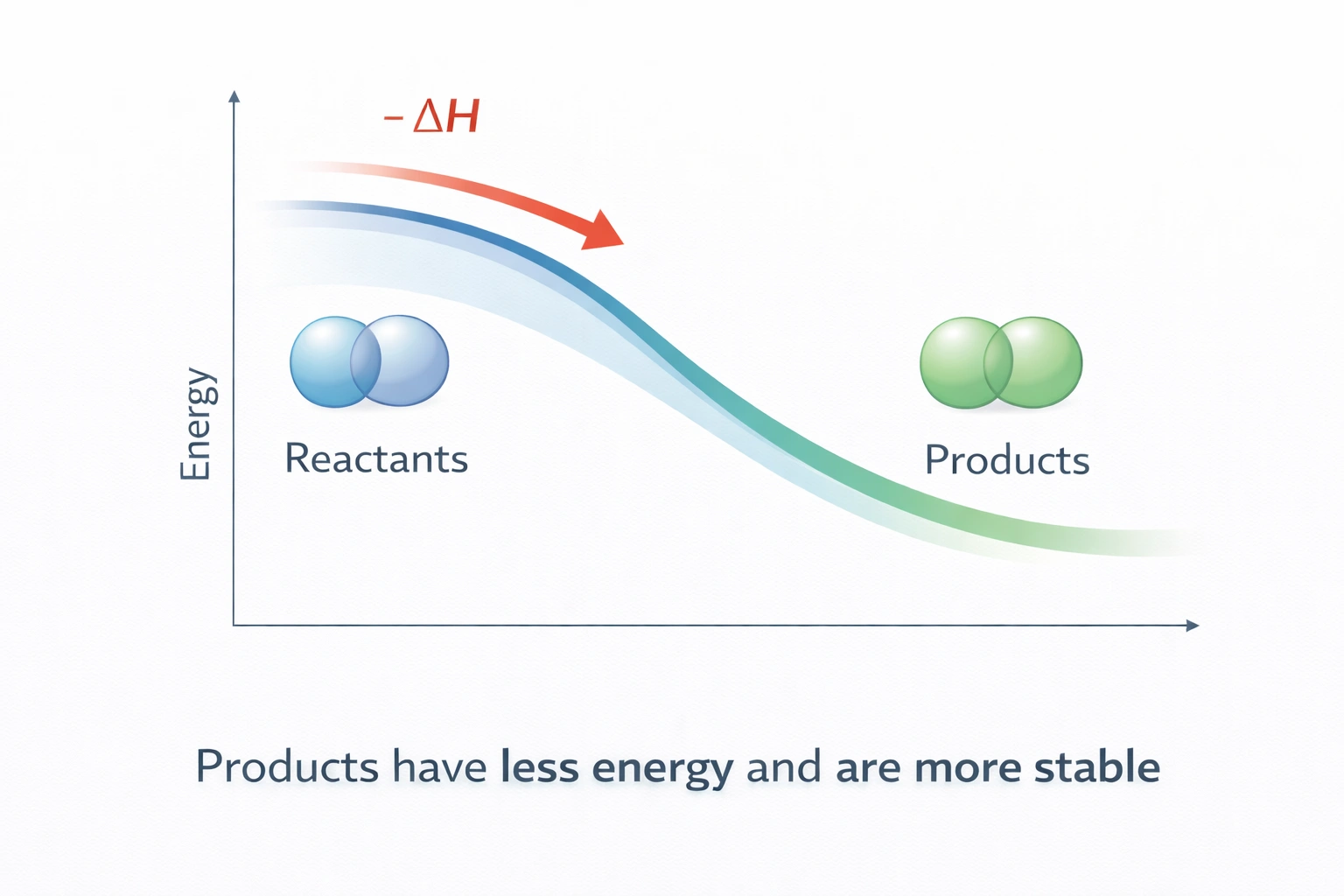
Topic Flashcards
Click to FlipWhat does a negative value for the heat of a reaction (ΔH < 0) indicate about the flow of energy?
It indicates an exothermic reaction, meaning energy (usually as heat) is released from the reacting system to the surroundings.
In an exothermic reaction (ΔH < 0), how does the total potential energy of the products compare to that of the reactants?
The products have a lower total potential energy than the reactants. The difference in energy is released to the surroundings.
How is the stability of a chemical species generally related to its potential energy?
Lower potential energy corresponds to greater thermodynamic stability. A more stable substance has stronger bonds and is less likely to undergo change.
For the reaction Reactants → Products with ΔH = -100 kJ, which is more stable: the reactants or the products?
The products are more stable. The reaction released 100 kJ of energy to form the lower-energy, more stable products.
What is typically true about the strength of the bonds in the products versus the reactants in an exothermic reaction?
The bonds in the products are, on average, stronger than the bonds in the reactants. More energy is released forming the new bonds than was required to break the old ones.