Which of the following atoms will have an overall negative charge?
-
A
9 protons, 10 neutrons, 9 electrons
-
B
12 protons, 13 neutrons, 10 electrons
-
C
14 protons, 14 neutrons, 10 electrons
-
D
15 protons, 16 neutrons, 18 electrons
The atom with 15 protons, 16 neutrons, and 18 electrons will have an overall negative charge.
The net electrical charge of an atom or ion is determined by the balance between the number of positively charged protons in the nucleus and the number of negatively charged electrons in the surrounding cloud. The formula is: Net Charge = (Number of Protons) - (Number of Electrons). Neutrons are neutral and do not affect charge.
A) 9 protons, 10 neutrons, 9 electrons
Number of protons: 9. Number of electrons: 9. The charges are balanced (9+ vs. 9-). Net charge = 9 - 9 = 0. This atom is electrically neutral.
B) 12 protons, 13 neutrons, 10 electrons
Number of protons: 12. Number of electrons: 10. There are more protons than electrons. Net charge = 12 - 10 = +2. This atom has a net positive charge and is a cation.
C) 14 protons, 14 neutrons, 10 electrons
Number of protons: 14. Number of electrons: 10. There is a significant excess of protons. Net charge = 14 - 10 = +4. This atom has a net positive charge.
D) 15 protons, 16 neutrons, 18 electrons
Number of protons: 15. Number of electrons: 18. There are more electrons than protons. Net charge = 15 - 18 = -3. This atom has a net negative charge and is an anion.
Conclusion:
An atom carries a negative charge only when it possesses more electrons than protons. Evaluating each option using the fundamental charge calculation reveals that only the atom with 15 protons and 18 electrons has an electron excess, resulting in a net negative charge.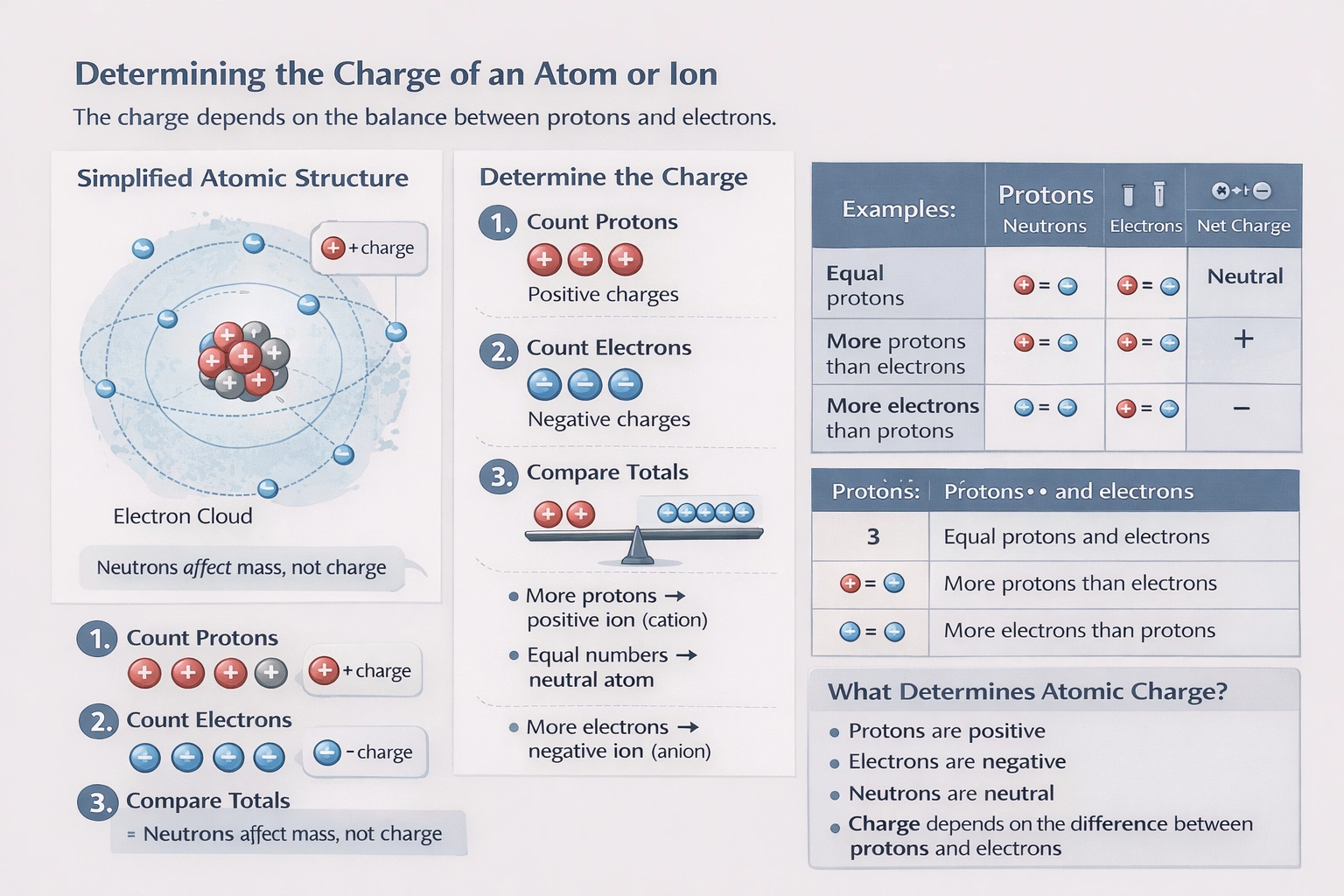
Topic Flashcards
Click to FlipWhat simple formula determines the net electrical charge of an atom or ion?
Net Charge = (Number of Protons) – (Number of Electrons).
An atom has 17 protons and 18 electrons. What is its net charge, and what type of ion is it?
Net charge = -1. It is an anion (negatively charged ion).
Which subatomic particle (proton, neutron, or electron) does NOT influence the overall charge of an atom?
The neutron (it is electrically neutral).
If an atom is electrically neutral, what must be true about the number of protons and electrons?
The number of protons equals the number of electrons.
An ion has a charge of -2. If it has 16 protons, how many electrons does it have?
18 electrons. (Charge = Protons – Electrons; -2 = 16 – Electrons; Electrons = 16 + 2 = 18).