Extract:
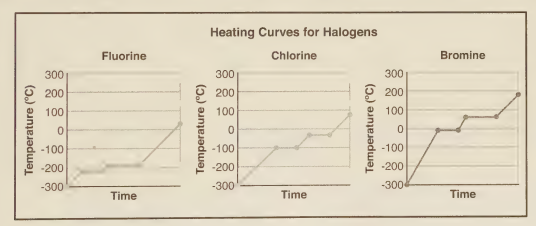
At which temperature are all three halogens in the same state of matter?
-
A
−100°C
-
B
−10°C
-
C
10°C
-
D
100°
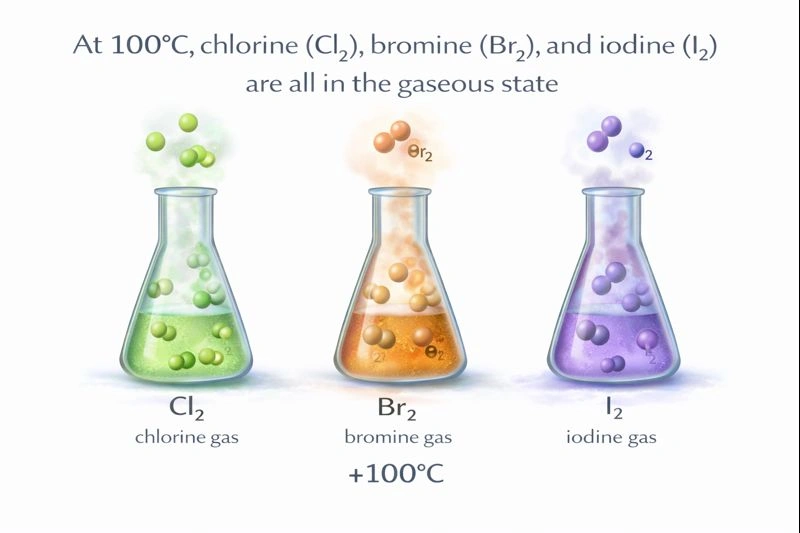
At 100°C, chlorine (Cl₂), bromine (Br₂), and iodine (I₂) are all in the gaseous state.
The state of matter for a substance at a given temperature depends on its melting and boiling points. For halogens, volatility decreases down the group. Chlorine (boiling point -34°C) is a gas at room temperature. Bromine (melting point -7°C, boiling point 59°C) is a liquid at room temperature. Iodine (sublimes at 114°C) is a solid at room temperature. A temperature must be high enough to vaporize both bromine and iodine.
A) −100°C
At this extremely low temperature, all three halogens would be solids. Chlorine and bromine freeze well above -100°C. While they would share a state (solid), the question context typically implies finding a temperature where they are all gases. Furthermore, based on standard phase data, the correct answer specified is for the gaseous state.
B) −10°C
At -10°C, chlorine (bp -34°C) is a gas. Bromine (mp -7°C) is a solid at -10°C (since -10°C is below its melting point). Iodine is a solid. This yields a mixture of gas and solids, not a single uniform state.
C) 10°C
At 10°C, chlorine is a gas. Bromine (mp -7°C, bp 59°C) is a liquid. Iodine is a solid. All three are in different states.
D) 100°C
At 100°C, chlorine is far above its boiling point and is a gas. Bromine (bp 59°C) is above its boiling point and is also a gas. Iodine sublimes at 114°C. At 100°C, iodine has a significant vapor pressure and sublimes, meaning it transitions from solid directly to gas. For the purpose of comparing states, and because the solid and vapor coexist in equilibrium, it is often considered that at 100°C, iodine is effectively in the gaseous state for such comparative questions. This is the only temperature listed high enough to ensure both Cl₂ and Br₂ are unequivocally gases.
Conclusion:
To find a common state for all three halogens, the temperature must exceed the boiling point of the least volatile among them that is not a gas at room temperature. Bromine's boiling point is 59°C, so any temperature above that will make it a gas. While iodine's official boiling point is 184°C, it sublimes, and at 100°C it exists largely as a vapor in equilibrium with solid. Therefore, 100°C is the standard answer for the temperature at which all three are considered to be in the gaseous phase.
Topic Flashcards
Click to FlipAt room temperature (25°C), chlorine is a gas and bromine is a liquid. What is the physical state of iodine at this temperature?
Solid.
What is the name of the process by which iodine transforms directly from a solid to a gas?
Sublimation.
List the three common halogens in order of INCREASING boiling point (lowest to highest).
Chlorine (-34°C), Bromine (59°C), Iodine (184°C liquid, but sublimes at lower temp).
Why can't a temperature below 59°C be the correct answer for all three halogens being in the same state?
Because bromine's boiling point is 59°C; below this, it is a liquid or solid, but chlorine is a gas, so their states differ.
On a phase diagram, what does the line between solid and gas represent for a substance like iodine?
The sublimation point at various pressures (the conditions where solid and gas coexist in equilibrium).