Why are boats more buoyant in salt water than in fresh water?
-
A
Salt decreases boat mass.
-
B
Salt increases water volume.
-
C
Salt affects boat density.
-
D
Salt increases water density.
Boats are more buoyant in salt water because salt increases the density of the water.
Buoyancy is explained by Archimedes’ principle, which states that an object immersed in a fluid experiences an upward buoyant force equal to the weight of the fluid it displaces. When the density of the fluid is higher, a given volume of displaced fluid has more mass and therefore more weight. This produces a greater buoyant force, allowing boats to float higher.
A) Salt decreases boat mass.
The mass of the boat does not change when it is placed in salt water. Salt dissolves in the water, not in the boat, so this option does not explain increased buoyancy.
B) Salt increases water volume.
Dissolving salt in water does not significantly increase the volume of the water. Any small volume change is negligible and does not account for the increased buoyant force.
C) Salt affects boat density.
The density of the boat remains the same regardless of whether it is in fresh water or salt water. Buoyancy depends on the density of the fluid, not the object.
D) Salt increases water density.
When salt dissolves in water, it adds mass without greatly increasing volume, increasing the water’s density. According to Archimedes’ principle, a denser fluid exerts a greater buoyant force on an object, allowing boats to float more easily and higher in the water.
Conclusion
Salt water is denser than fresh water, and this higher density produces a greater buoyant force. As a result, boats are more buoyant in salt water than in fresh water.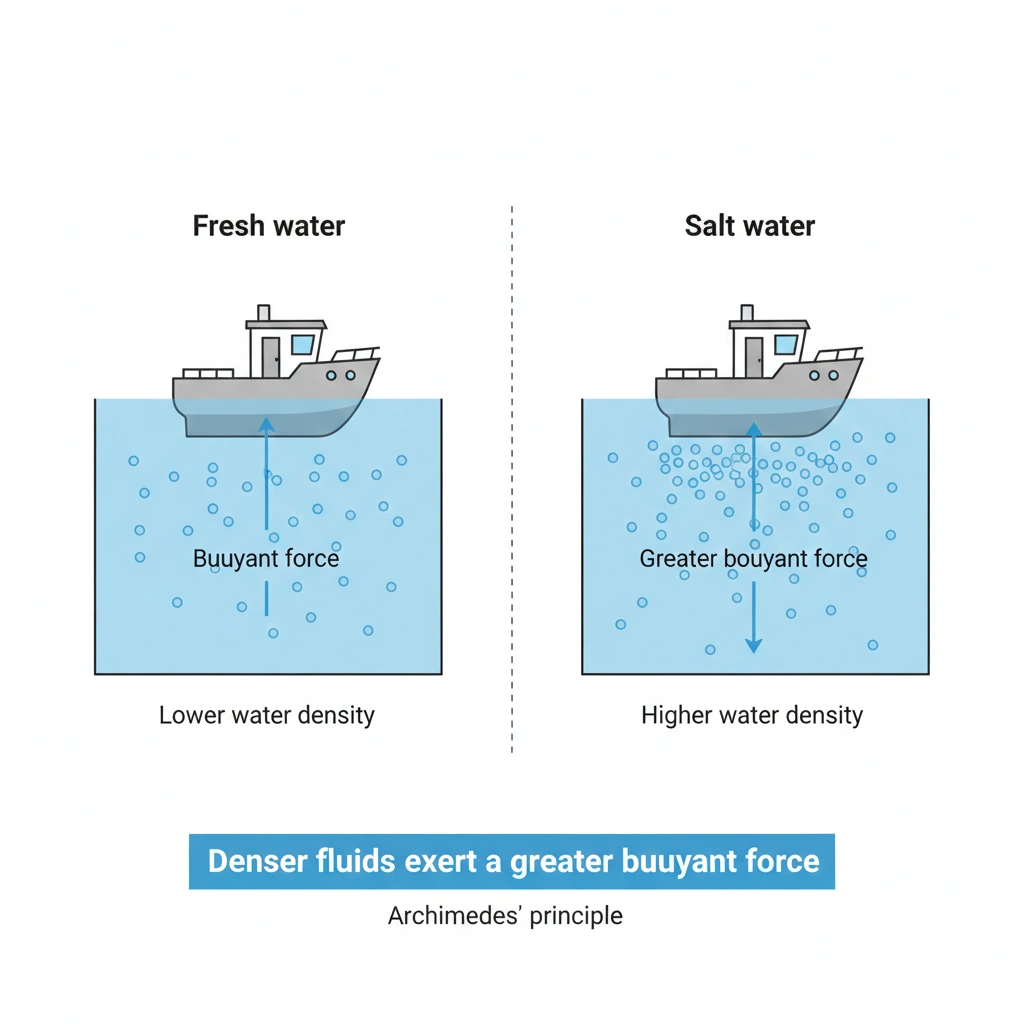
Topic Flashcards
Click to FlipState Archimedes' Principle. What determines the magnitude of the buoyant force on an object?
An object immersed in a fluid experiences an upward buoyant force equal to the weight of the fluid it displaces. The magnitude is determined by the density of the fluid and the volume of fluid displaced (Buoyant Force = ρ_fluid * V_displaced * g).
How does adding salt to water change its density? Explain in terms of mass and volume.
Dissolving salt adds mass to the water without significantly increasing its volume. Since density = mass/volume, adding mass while volume remains nearly constant increases the density.
For a boat floating in both fresh and salt water, which property of the boat (its weight, volume, density) remains constant, and why is this important?
The boat's weight (and therefore its mass) remains constant. To float, the buoyant force must equal the boat's weight. In denser salt water, it needs to displace less volume to achieve this same buoyant force, so it floats higher.
The Dead Sea is extremely salty. Explain why people float so easily in it compared to a freshwater lake
The Dead Sea's water has a very high density due to its high salt concentration. A person displaces a volume of water. The weight of that displaced, very dense water is much greater than the person's weight, resulting in a large buoyant force that makes them float very high.
If a loaded ship moves from the ocean (salt water) into a freshwater river, what must happen to its displacement (the volume of its hull below the waterline) to keep it afloat?
The displacement (submerged volume) must increase. Fresh water is less dense, so the ship must displace a larger volume of it to generate a buoyant force equal to the ship's unchanged weight.