Which names the approximate freezing point of water?
-
A
0 K
-
B
100 K
-
C
273 K
-
D
373 K
The approximate freezing point of water is 273 K.
The Kelvin temperature scale is based on absolute zero, and temperature values on this scale are offset from the Celsius scale by 273 degrees. Water freezes at 0 °C, which corresponds to 273 K on the Kelvin scale.
A) 0 K
Zero kelvin represents absolute zero, the lowest possible temperature, at which molecular motion is minimized. This is far colder than the freezing point of water.
B) 100 K
A temperature of 100 K corresponds to about −173 °C, which is well below the freezing point of water.
C) 273 K
A temperature of 273 K is equivalent to 0 °C, the temperature at which pure water freezes under standard atmospheric pressure.
D) 373 K
A temperature of 373 K corresponds to 100 °C, which is the boiling point of water, not the freezing point.
Conclusion
On the Kelvin scale, the freezing point of water occurs at approximately 273 K.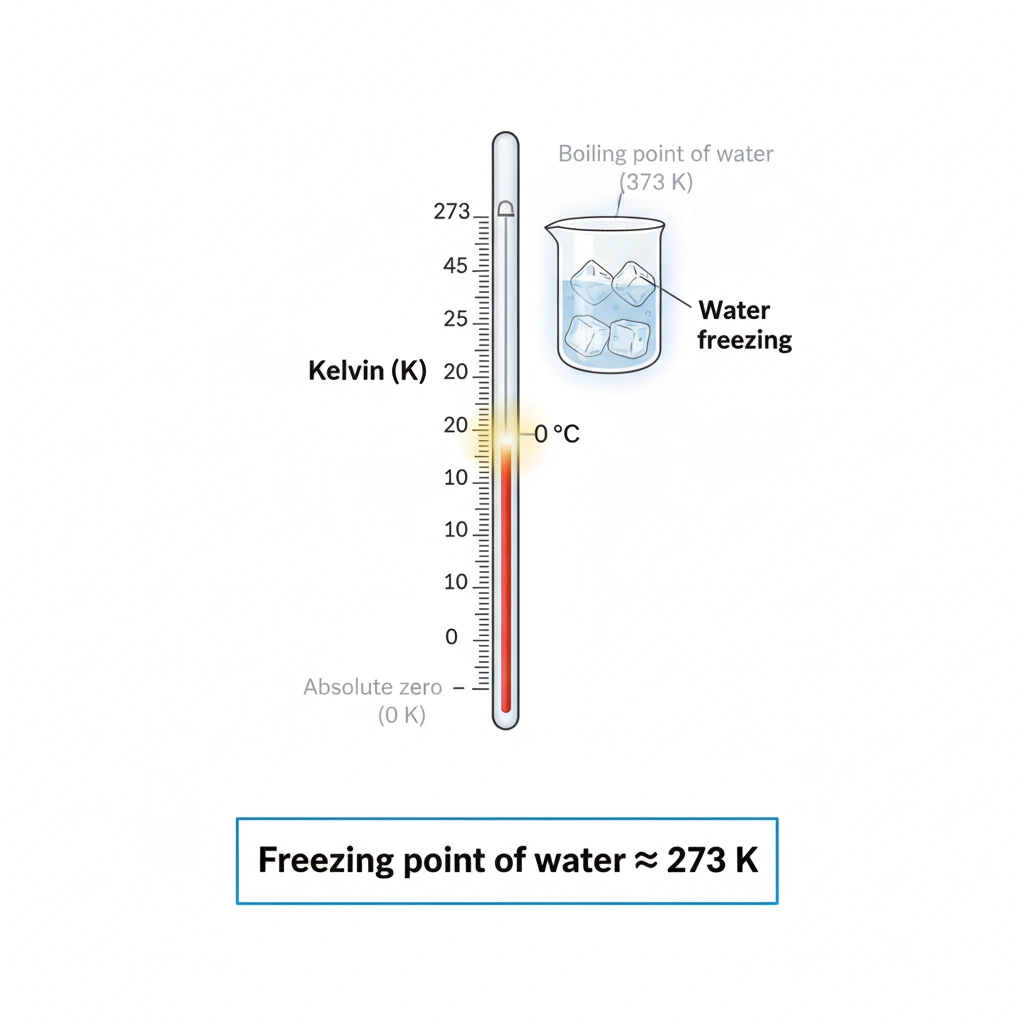
Topic Flashcards
Click to FlipWhat is the exact formula to convert a temperature from degrees Celsius (°C) to Kelvin (K)?
K = °C + 273. (Therefore, 0 °C = 273 K).
What does a temperature of 0 Kelvin (0 K) represent, and what is its equivalent in degrees Celsius?
0 K represents absolute zero, the theoretical point where all molecular motion ceases. It is equal to -273 °C.
On the Kelvin scale, what are the approximate temperatures for the freezing point and boiling point of water?
Freezing point: 273 K. Boiling point: 373 K.
Why is the Kelvin scale considered an "absolute" temperature scale, unlike Celsius or Fahrenheit?
Its zero point (0 K) is based on a fundamental physical minimum (absolute zero), not an arbitrary point like the freezing point of water.
If the temperature of a substance is 300 K, is it above or below the freezing point of water? By how many degrees Celsius?
It is above freezing. 300 K - 273 = 27 °C.