Why is hydrogen bonding with water important?
-
A
It contributes to water's low boiling point.
-
B
It prevents water from being a universal solvent.
-
C
It creates weak interactions among water molecules.
-
D
It allows water in its solid form to float on liquid water.
The importance of hydrogen bonding in water stems from the collective effect of numerous, individually weak, intermolecular attractions that give water its unique properties.
A hydrogen bond in water forms between the partially positive hydrogen atom of one molecule and the lone pair electrons on the partially negative oxygen atom of a neighboring molecule. This extensive, dynamic network of bonds is responsible for the cohesive and adhesive properties that result in water's high surface tension, high specific heat capacity, high heat of vaporization, and solvent capabilities.
A) It contributes to water's low boiling point.
Hydrogen bonding has the opposite effect. The attractive forces between water molecules due to hydrogen bonding are relatively strong for an intermolecular force. More thermal energy is required to break these bonds and transition water from liquid to gas. Consequently, water has a high boiling point (100°C at sea level) compared to other molecules of similar size that lack extensive hydrogen bonding.
B) It prevents water from being a universal solvent.
Hydrogen bonding is a primary reason water is such an excellent and versatile solvent. Water's polarity and ability to form hydrogen bonds allow it to dissolve a wide range of ionic compounds and polar molecules. Water molecules surround solute particles, forming hydration shells through ion-dipole interactions or hydrogen bonding, effectively pulling them into solution. Hydrogen bonding thus enhances, not prevents, water's solvating power.
C) It creates weak interactions among water molecules.
Hydrogen bonds are intermolecular forces that are weaker than covalent or ionic bonds but strong in aggregate due to their sheer number in liquid water. This pervasive network of weak, dynamic bonds is the direct cause of water's emergent properties, its high surface tension, high specific heat, high heat of vaporization, and viscosity. These properties are fundamental to life and Earth's environment.
D) It allows water in its solid form to float on liquid water.
This is a specific and ecologically vital consequence of hydrogen bonding, but it is not the primary reason for hydrogen bonding's overarching importance. When water freezes, hydrogen bonds stabilize a crystalline lattice that is more spacious and less dense than liquid water. This lower density of ice causes it to float. While crucial, this property is one of many that stem from the underlying weak interactions described.
Conclusion:
Hydrogen bonding is paramount because it establishes an extensive network of interactions between water molecules. This network directly causes water's exceptional physical properties. The notion that it lowers water's boiling point or reduces its solvent ability is factually reversed. While the density anomaly of ice is a critical outcome, the fundamental importance lies in the cohesive network these weak interactions create, which governs countless biological and chemical processes.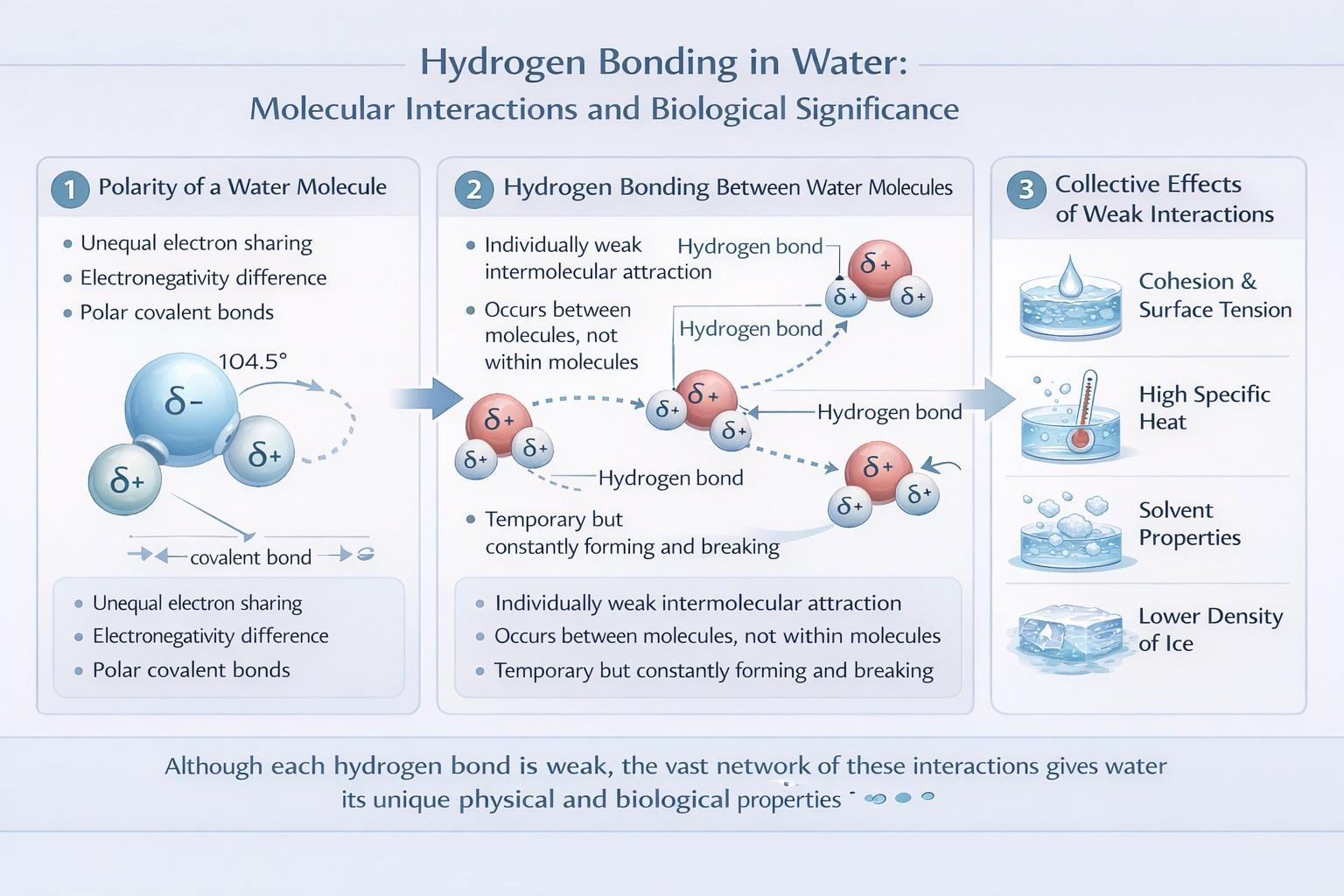
Topic Flashcards
Click to FlipWhat type of attractive force is a hydrogen bond between water molecules?
An intermolecular force (between molecules), specifically between a hydrogen (δ+) and an oxygen (δ-) with lone pair electrons.
What is the effect of hydrogen bonding on water's boiling point?
It raises it. Water has a high boiling point because significant energy is needed to break the hydrogen bonds.
Why does water have an unusually high boiling point compared to other small molecules like methane (CH₄)?
Because hydrogen bonds between water molecules are relatively strong intermolecular forces. It takes a lot of heat energy to break this network and turn water into a gas.
True or False: The hydrogen bonds in liquid water are permanent, static connections that lock molecules in place.
False. Hydrogen bonds in liquid water are dynamic—they constantly break and reform, allowing water to flow while maintaining cohesion.
What property of water, enabled by hydrogen bonding, allows it to dissolve ionic compounds like sodium chloride (table salt) so effectively?
Water's polarity and hydrogen-bonding capability. The partial charges on water molecules surround and pull apart the salt's ions in a process called hydration.