A good illustration to differentiate between the two types of properties is that mass and volume are _____ properties, but their ratio (density) is a(n) _____ property of matter.
-
A
chemical, physical
-
B
physical, chemical
-
C
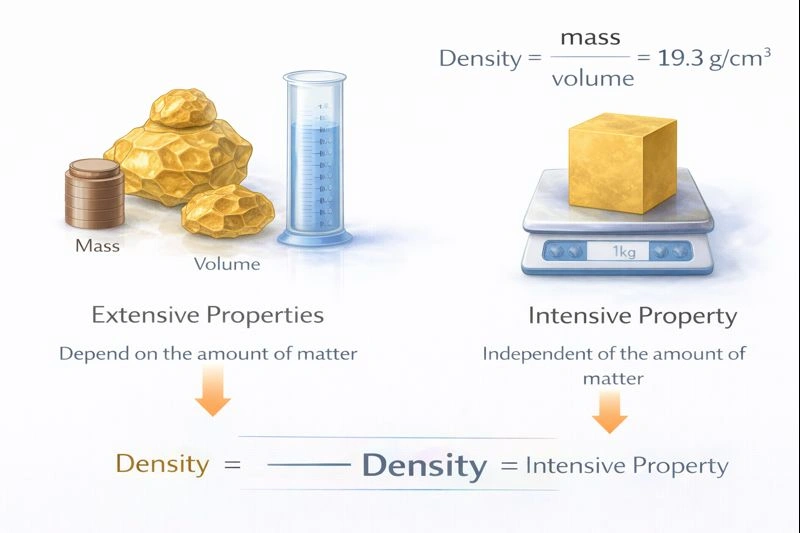
extensive, intensive
-
D
intensive, extensive
Mass and volume are extensive properties, but their ratio (density) is an intensive property of matter.
Physical properties are categorized based on their dependence on the amount of substance present. Extensive properties, such as mass, volume, and length, depend on the quantity or size of the sample. If you double the amount of matter, you double its mass and volume. Intensive properties, such as density, color, melting point, and boiling point, do not depend on the amount of substance. They are inherent characteristics of the material itself. The density of pure gold is the same whether you have a 1-gram nugget or a 1-kilogram bar.
A) chemical, physical
Both mass and volume are physical properties, not chemical properties. Chemical properties describe a substance's potential to undergo chemical change (e.g., reactivity, flammability). Density is also a physical property. This option misapplies the categories.
B) physical, chemical
Again, all properties mentioned—mass, volume, and density—are physical properties. They can be observed or measured without changing the substance's chemical identity. Density is not classified as a chemical property.
C) extensive, intensive
Extensive properties scale with the amount of matter. Mass and volume are classic examples. Intensive properties are independent of amount. Density, defined as mass per unit volume (an extensive property divided by another extensive property), always yields an intensive property. This ratio remains constant for a pure substance at a given temperature and pressure, regardless of sample size.
D) intensive, extensive
This reverses the correct relationship. Intensive properties, like density, do not depend on amount. Extensive properties, like mass, do depend on amount. Mass and volume are clearly extensive, not intensive.
Conclusion:
The distinction between extensive and intensive properties is fundamental in chemistry. Extensive properties change when the sample size changes, while intensive properties are invariant and serve as identifying characteristics of a material. Since mass and volume change proportionally with sample size, they are extensive. Their ratio, density, cancels out this dependence on size, making it an intensive property.
Topic Flashcards
Click to FlipDefine an "extensive property" of matter and give two examples other than mass or volume.
An extensive property depends on the amount of matter present. Examples: length, weight, heat capacity.
Define an "intensive property" of matter and give two examples other than density.
An intensive property is independent of the amount of matter. Examples: color, melting point, conductivity.
Why is the density of a pure substance considered an intensive property?
It is a ratio of two extensive properties (mass/volume). Changing the sample size changes both mass and volume proportionally, leaving the ratio (density) constant.
A chemist has two samples of the same pure metal. One is large and one is small. Which property will be the same for both, helping to identify the metal?
Intensive properties (like density, melting point, or color).
If you cut a gold bar in half, which of these changes (is extensive): Mass, Density, Color?
Only the mass changes (decreases by half). Density and color remain the same (they are intensive).