Which would turn litmus paper blue?
-
A
HF
-
B
KOH
-
C
HNO₃
-
D
C₅H₆O₃
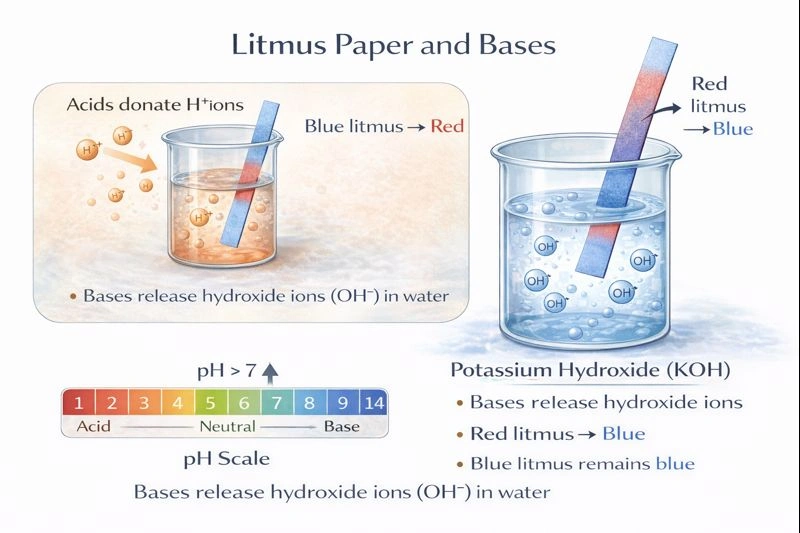
Potassium hydroxide (KOH) would turn litmus paper blue.
Litmus paper is a common pH indicator. Blue litmus paper turns red under acidic conditions (pH < 7), and red litmus paper turns blue under basic (alkaline) conditions (pH > 7). A substance that turns litmus blue must be basic, meaning it increases the concentration of hydroxide ions (OH⁻) in aqueous solution.
A) HF (Hydrofluoric acid)
HF is an acid, albeit a weak one. In water, it partially dissociates to produce H⁺ (or H₃O⁺) ions, lowering the pH. Acids turn blue litmus paper red, not blue.
B) KOH (Potassium hydroxide)
KOH is a strong base. It dissociates completely in water into K⁺ ions and OH⁻ ions. The high concentration of OH⁻ raises the pH significantly above 7. A base will turn red litmus paper blue. If blue litmus paper is introduced, it will remain blue.
C) HNO₃ (Nitric acid)
HNO₃ is a strong, highly corrosive acid. It dissociates completely to produce H⁺ and NO₃⁻ ions, creating a very low pH. It would turn blue litmus paper red.
D) C₅H₆O₃ (Levulinic acid)
The molecular formula C₅H₆O₃ corresponds to levulinic acid (4-oxopentanoic acid), an organic acid containing a carboxylic acid group (-COOH). Like other acids, it donates a proton in solution, resulting in an acidic pH. It would also turn blue litmus paper red.
Conclusion:
The behavior of litmus paper is a classic test for acidity or basicity. Only substances with basic properties (pH > 7) will cause litmus paper to appear blue. Potassium hydroxide, as a strong, ionic base, is a definitive example of such a substance.
Topic Flashcards
Click to FlipWhat color will a strip of RED litmus paper turn when dipped into a basic solution like sodium hydroxide (NaOH)?
Blue.
In water, potassium hydroxide (KOH) dissociates completely. What ion is responsible for its ability to turn litmus paper blue?
The hydroxide ion (OH⁻).
You have two clear solutions: one is nitric acid (HNO₃) and one is potassium hydroxide (KOH). Which one would cause BLUE litmus paper to change color, and to what color?
Nitric acid (HNO₃) would change blue litmus paper to red.
Hydrofluoric acid (HF) is classified as a weak acid. Will it still turn blue litmus paper red?
Yes. Both strong and weak acids turn blue litmus paper red because they increase the concentration of H⁺ ions (lower pH).
Levulinic acid (C₅H₆O₃) contains a carboxylic acid group. What is the expected result when it is tested with red litmus paper?
The red litmus paper will stay red (because it's an acid; only a base would turn it blue).